Research Article
Management of Abdominal Wound Dehiscence Following Cesarean Section: The Case for Debridement and Immediate Primary Re-Closure
Reuben A Falola, Alexandra Tilt, Alexandria M Carroll, Madison J Kim, Gloria Bowles-Johnson,
Christopher E Attinger and Karen K Evans*
Department of Plastic Surgery, MedStar Georgetown University Hospital, Washington, DC, USA
*Corresponding author: Karen K Evans, Department of Plastic Surgery, MedStar Georgetown University Hospital, Washington, DC, USA
Published: 24 Jan, 2018
Cite this article as: Falola RA, Tilt A, Carroll AM, Kim MJ,
Bowles-Johnson G, Attinger CE, et
al. Management of Abdominal Wound
Dehiscence Following Cesarean
Section: The Case for Debridement and
Immediate Primary Re-Closure. Clin
Surg. 2018; 3: 1881.
Abstract
Abdominal wound dehiscence occurs in 3.6% to 29% of all obstetrics and gynecology laparotomies,
producing a higher morbidity and mortality rates, increased cost of care, and lower Quality Of
Life (QOL) in this population. Evidence-based guidelines for management of post-operative wound
dehiscence after cesarean laparotomy are limited, and thus has led to wide variation in management
strategies. The traditional method of wet to dry dressing changes can prolong care and the period
of disability. Debridement and immediate re-closure is showing promise as the optimal means
management of these patients. In this article, we review and assess the factors that contribute
to patient outcomes after cesarean laparotomy, and present our experience with this operative
technique.
Keywords: Cesarean section; Debridement and Re-closure; Wound dehiscence
Abbreviations
QOL: Quality of Life; BMI: Body Mass Index; COPD: Chronic Obstructive Pulmonary Disorder; SFS: Superficial Fascial System
Introduction
Abdominal wound dehiscence occurs in 3.6% to 29% of all obstetrics and gynecology
laparotomies [1,2], and frequently presents with prolonged wound healing and continued
drainage. Multiple host-related factors, including obesity, smoking, surgical site infection, which
occurs in roughly 3% to 20% of cesarean births [3,4], have been shown to inhibit wound healing.
Other technical factors, such as inadequate hemostasis leading to the accumulation of seroma or
hematoma, can also impede wound healing. Similarly, post-operative increases in intra-abdominal
pressure due to ileus, vomiting, or coughing can place tension on the wound, and thus increase the
probability of separation [5].
Obstetric patients tend to be young and healthy, however, abdominal wound dehiscence after
cesarean section has been linked to increases the cost of care, lower Quality Of Life (QOL), and a
higher morbidity and mortality in this population [6,7]. In fact, the mortality rate for abdominal
laparotomy dehiscence has been reported as high as 45% [8]. Prolonged disability as a result of the
abdominal wound can also interfere with post-partum mother-child bonding, and the emotional
toll, although difficult to quantify, can be significant. Evidence based guidelines for management of
post-operative wound dehiscence after cesarean laparotomy; however, are limited, and thus has led
to wide variation in management strategies. Because of this large gap in knowledge, clinical studies
are necessary to assess optimal management strategies for dehisced cesarean laparotomies.
The traditional method of wet to dry dressing changes, which allows for healing by secondary
intention, is costly, prolonging care and the period of disability [9,10]. Secondary closure
techniques that utilize wound debridement and a period of granulation before primary re-closure,
are increasingly being described in the literature as yielding superior results when compared to
non-operative management [9,11]. Debridement and immediate re-closure without a period of
granulation; however, is showing promise as the optimal means management of patients with
dehisced cesarean laparotomies, as has the potential to further reduce the period of disability,
financial costs, and emotional toll. We present our experience with this operative technique and assess the factors that contribute to patient outcomes.
Materials and Methods
After IRB approval was obtained, a retrospective chart review was conducted at a single academic medical center, of obstetric patients referred to plastic surgery for surgical debridement, irrigation, and immediate primary re-closure of suprafascially dehisced cesarean laparotomies, from 2011 to 2015. Out of 13 patients, 7 met inclusion criteria for the purposes of this study. Risk factors for dehiscence including increasing Body Mass Index (BMI), nutrition status, diabetes, Chronic Obstructive Pulmonary Disorder (COPD), chronic kidney disease, ethnicity, hypertension, peripheral vascular disease, time to plastic surgery consultation, smoking history, subsequent wound dehiscence, and culture positive presence of infection were identified as risk factors influencing healing. The relative risk for wound healing and the median time to wound healing were assessed using Cox proportional hazard model for time until event analysis. Healing was defined as a Georgetown University Hospital Center for Wound Healing clinic note stating that the “wound is healed.”
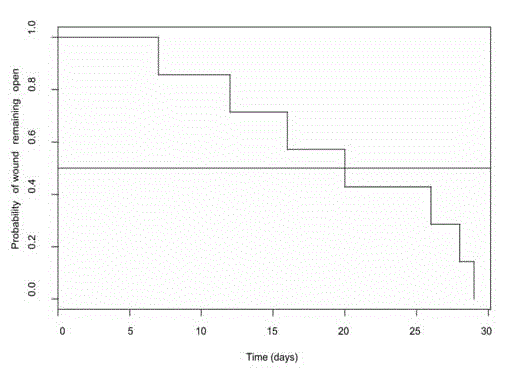
Figure 1
Results
Prior to debridement and re-closure, all patients had undergone cesarean section with a pfannenstiel incision. The average age was 34 and average BMI was 45. Complete epithelialization occurred in 100% of these patients. The mean number of debridements in all patients was 1.71 (SD 0.76). The median time to wound healing was 20 days. (Figure 1) Using the univariate Cox hazard model, no demographic factors were found to significantly decrease healing rates in this population. Postoperative complications included wound infection (n=2), incisional pain (n=2) and subsequent wound dehiscence after debridement and re-closure (n=1). Four out of the 7 total patients were reached for long-term follow up. All patients had remained healed, with a mean follow up time of 2.9 years (SD=1.6). The results are shown in Table 1.
Discussion
Both patient-related factors and non-patient factors contribute to surgical wound dehiscence after abdominal laparotomy. Unfortunately, there is wide variability in which patient and nonpatient factors have been included in the various studies, examining laparotomy dehiscence. A retrospective review of a patient registry conducted by van Ramshorst et al. compared 363 all-cause cases of abdominal wound dehiscence to matched controls. They found that the major independent risk factors predictive of abdominal wound dehiscence included age, gender, chronic pulmonary disease, ascites, jaundice, anemia, and emergency surgery, type of surgery, postoperative coughing, and wound infection [8]. Cesarean wounds, specifically, are also more likely to dehisce in the presence of obesity, hematoma and seroma, and in African Americans patients [12] (OR 3.9, 95% CI 1.1-13.8). Other factors such as post-operative increase in intra-abdominal pressure secondary to ileus, vomiting, and coughing have also been implicated [5].
Preventive Measures
Infection occurs in 3% to 20% of cesarean laparotomies,3 and
has been shown to inhibit wound healing, thus leading to wound
disruption. This number can approach 40% after obstetrics and
gynecology laparotomy in patients with greater than 3 cm of
subcutaneous abdominal fat [13]. Prior to incision, much can be done
to prevent the development of a postoperative infection. This should
include hair trimming at the surgical site just priorto incision, skin
preparation with chlorhexidine-alcohol scrub or povidone-iodine
scrub, whichcan reduce the microbial load and significantly decrease
incidence of superficial surgical site infections (p=0.008) [14]. The
use of preoperative antibiotics prior to incision, instead of after the
umbilical cord has been clamped, is also beneficial [15-17].
In conjunction with adequate skin preparation, the type of
incisions used contributes to wound dehiscence. The literature has
demonstrated the superiority of the pfannenstiel incision over the
vertical incision, in preventing post-operative wound disruption
[5,18,19]. Although, there are early reports suggesting that the use of
vertical incisions in women with morbid obesity may actually lower
wound complication rates after cesarean section [20] (OR=0.32;
CI=0.17-0.62; P< 0.001), the current standard is the transverse
incision. A study by Mowat et al. found that vertical incisions are, in
fact, 8 times more likely to dehisce than pfannenstiel’s incision [18].
Hematoma and infection are also present nine times more frequently
in vertical incisions compared with the pfannenstiel incision [21].
There is much variability in the type of closure technique utilized
after cesarean section, and reports on the best means for preventing
wound dehiscence are often conflicting. Closure of all layers of
the abdominal wall, is not routinely practiced following cesarean
section. The abdominal wall includes the of skin, subcutaneous
tissue, superficial fascia, muscle, deep fascia, extraperitoneal fat and
peritoneum, and exactly which layers should be closed following
cesarean laparotomy is cause for much debate.
Song et al. demonstrated that dermal closure in conjunction with
repair of the Superficial Fascial System (SFS) achieved a higher failure
load, and thus demonstrates a greater biomechanical strength, when
compared to dermal sutures alone, in an ex vivo study of human
abdominal tissue [22]. Because the SFS is integral in providing
structural support and minimizes skin tension, approximation of this
layer during closure of the cesarean laparotomy can reduce wound
complications, including early dehiscence [22,23].
Other studies have examined whether closure of the peritoneum
can improve outcomes in this population. While some data suggests
that there is no postoperative difference in the incidence of dehiscence,
wound infection, and length of hospital stay after cesarean section
between patients in which the parietal peritoneum was closed, and
those in whom the parietal peritoneum was left open [24,25], other studies have demonstrated improved outcomes when the parietal
peritoneum is left open after cesarean section. For instance Grundsell
et al. reported that non-closure of the peritoneum not only had an
average operating time that was significantly shorter by 7.9 min
(p< 0.01), but that non-closure had comparative cost-savings, and
correlated with a significant decrease in postoperative complications
such as infection (p< 0.05) [26].
A few other studies have analyzed outcomes after closure versus
non-closure, at other levels within the abdominal wall. Obese
patients traditionally have a higher incidence of difficult wound
healing and dehiscence, compared to those with normal BMI values.
Surgical trauma to fatty subcutaneous layer (Camper’s fascia)
can leave large areas of fat necrosis, providing a dead space within
which seromas and hematomas can form and disrupt local wound
healing. In patients with a subcutaneous tissue layer thicker than 2
cm, the literature supports a reduced risk for hematoma, seroma,
and resultant superficial wound dehiscence after closure of the deep
subcutaneous layer [27-29]. Other techniques that have successfully
decreased cesarean wound dehiscence rates due to hematoma and
seroma, include the prophylactic placement of a negative pressure
drainage device such as the Jackson-Pratt or J-VAC drain during
closure [4]; however, there is conflicting data suggesting that this
technique may be not always be effective [30]. Prophylactic incisional
negative pressure therapy has also been shown to decrease wound
complication in obese women, following cesarean section [31].
Like other aspects of cesarean closure, there is variability with
regard to the materials used for skin closure. Although staples
have been used to minimized skin trauma during closure and may
demonstrated better scar cosmesis [32], closure with suture is
associated with a decrease in wound complications up to 57%, when
compared to staples [33]. Conversely, other studies suggest that
closure with suture (OR 5.4, 95% CI 1.8-16.1) was associated with
a significantly higher incidence of wound disruption after cesarean
laparotomy [12].
Irrespective of the closure technique employed, emergency
procedures or cesareans combined with another simultaneous
procedure such as an abdominoplasty, tend to produce worse
outcomes [34]. Emergency procedures may not allow time for
optimal surgical prep, and may the performance of the surgical team
during emergency cesareans can also be affected. In such instances
there is a potential for poor wound closure, contamination, and sub-optimal patient nutritional status, all increasing the probability of
wound dehiscence.
Table 1
Traditional Management
Conservative management of dehisced cesarean wounds is accomplished by packing the wound with a moistened sterile gauze, covered by a semi-occlusive dressings that may be impregnated with petrolatum or silicone, which allows small amounts of exudates to pass through. These dressings have the added benefit of a moist environment that provides a medium through which cells important to healing can migrate [35]. Wet to dry dressing changes work as a form of physical debridement, which over time, allow for the periodic removal of inflammatory exudates, infections organisms, and debris while the wound bed granulates in. However, healing by secondary intention is a lengthy process, often prolonging the period of disability, and requiring multiple follow-up clinic visits. In conjunction with gauze packing, other adjunctive treatment methods such as the use of topical crude honey or the combination of zinc chloride spray and magnesium hydroxide ointment, have been shown to decrease the size of wounds, healing time, instance of infection, dehiscence and pain compared to placebo, however the data is limited, and the prospect of a lengthy recovery period still remains [36-38].
Debridement and Re-closure
Current multidisciplinary management strategies for dehisced
cesarean laparotomies, are trending away from traditional packing
techniques, and towards debridement and secondary re-closure.
Secondary re-closure employs both sharp wound debridement and
a minimum period of granulation, often around four days, and prior
to primary closure. A few studies have demonstrated the superiority
of secondary re-closure over secondary intention healing. Dodson
et al, in a prospective study of 33 similar obstetrics and gynecology
patients, found that the patients who underwent secondary closure
had a significantly shorter mean healing time (n=15) (17.6 +/- 7.2
days) compared to patients who were allowed to heal by secondary
intention (n=18) (61.2 +/- 35.8 days) (p< 0.001) [9]. The secondary
closure group also required a significantly fewer amount of followup
clinic visits (2.0 +/- 0.7) compared to the secondary intention
group (8.4 +/- 6.2) (p< 0.001) [9]. Similarly, study by Walter et al,
demonstrated a shorter healing time in patients who underwent
secondary closure (n=35) (3.2 +/- 3.7 days) compared to patients
allowed to heal by secondary intention healing (n=6) (71.8 days +/-
7.6) [11].
Debridement and immediate re-closure further improves upon
secondary re-closure in management of dehisced cesarean laparotomy
wounds. Our series of 7 patients who underwent debridement and
immediate re-closer, demonstrated a median healing time of 20 days
in the Cox Proportional Model for time to event analysis, which was
comparable to the simple mean healing time of the former studies,
and a more accurate assessment for reporting time to healing.
Further, immediate re-closure eliminates all together the period
of secondary intention healing, which may prolong the period of
disability, increase postoperative clinic visits, and take an emotional
toll on affected patients. Both secondary closure and immediate reclosure
do carry the risks associated with anesthesia and surgical
intervention, which is not present with wound packing, however, it
is within the purview of the study authors, that the benefit of surgical
management, namely debridement and immediate re-closure, far
outweigh the surgical risk. The risks and benefits of all methods of
managements should be discussed with patients, when deciding the
means of management that is best for them.
Limitations
There are a few significant limitations to this study. While one can observe a trend of complete healing in all 7 patients after debridement and immediate re-closure, it may be difficult to interpret the final message due to the limited sample size. Further, the lack of a control group of patients undergoing secondary closure may be considered a limitation to this study. More studies are warranted to examine such trends in a larger population and compared to a standard control. Future studies may also assess the differences in length of hospital stays, postoperative clinic visits, health care costs, as well as the effect of BMI on these two approaches to wound closure.
Conclusion
Operative management, including debridement of all nonviable tissue and immediate re-closure, is successful with a median time to heal of 20 days in our patients who had a mean BMI of 45. Patient risk factors including DM and smoking were not predictive of longer healing times. If optimization of skin preparations, type of laparotomy incision, multi-layer abdominal wall closure and closure materials during perioperative period fail to prevent cesarean laparotomy dehiscence, post-operative debridement and immediate re-closure may serve as an excellent option for management.
References
- Ferres MA, Olivarez SA, Trinh V, Davidson C, Sangi-Haghpeykar H, Aagaard-Tillery KM. Rate of wound complications with enoxaparin use among women at high risk for postpartum thrombosis. Obstet Gynecol. 2011;117(1):119-24.
- Pitkin RM. Abdominal hysterectomy in obese women. Surg gynecol obstet. 1976;142(4):532-6.
- Schneid-Kofman N, Sheiner E, Levy A, Holcberg G. Risk factors for wound infection following cesarean deliveries. Int J Gynaecol Obstet. 2005;90(10:10-15.
- Kajiwara K, Eizo Kimur, Makoto Nakano, Hirokuni Takano, Aikou Okamoto. Clinical experience of J‐VAC drains for skin closure in the laparotomy of obstetrics and gynecology. J Obstet Gynaecol Research. 2014;40(40):1089-97.
- Helmkamp FB. Abdominal wound dehiscence. Am J Obstet Gynecol. 1977;128 (7):803-7.
- Kirkland B, Briggs JP, Trivette SL, Wilkinson WE, Sexton DJ. The impact of surgical-site infections in the 1990s: attributable mortality, excess length of hospitalization, and extra costs. Infect Control Hosp Epidemiol. 1999;20(11):725-730.
- Zhan C, Miller MR. Excess length of stay, charges, and mortality attributable to medical injuries during hospitalization. JAMA. 2003;290(14):1868-74.
- van Ramshorst GH, Nieuwenhuizen J, Hop WC, Arends P, Boom J, Jeekel J, et al. Abdominal Wound Dehiscence in Adults: Development and Validation of a Risk Model. World J Surg. 2010;34.1:20-27.
- Dodson MK, Magann EF, Meeks GR. A randomized comparison of secondary closure and secondary intention in patients with superficial wound dehiscence. Obstet Gynecol. 1992;80(3):321-4.
- Sarsam SE, Elliott JP, Lam GK. Management of wound complications from cesarean delivery. Obstet Gynecol Survey. 2005;60(7):462-73.
- Walters MD, Dombroski RA, Davidson SA, Mandel PC, Gibbs RS. Reclosure of disrupted abdominal incisions. Obstet Gynecol. 1990;76(4):597-602.
- Subramaniam A, Jauk VC, Figueroa D, Biggio JR, Owen J, Tita AT, et al. Risk factors for wound disruption following cesarean delivery. J Matern-Fet Neonat Med. 2014;27(12):1237-1240.
- Soper DE, Bump RC, Hurt WG. Wound infection after abdominal hysterectomy: Effect of the depth of subcutaneous tissue. Am J Obstet Gynecol. 1995;173:465-71.
- Darouiche RO, Wall MJ, Itani KM, Ottersono MF, Webb AL, Carrick MM, et al. Chlorhexidine-alcohol versus povidone-iodine for surgical-site antisepsis. N Engl J Med. 2010;362:18-26.
- Sullivan SA, Smith T, Chang E, Hulsey T, Vandorsten JP, Soper D. Administration of cefazolin prior to skin incision is superior to cefazolin at cord clamping in preventing postcesarean infectious morbidity: a randomized controlled trial. Am J Obstet Gynecol. 2007;196(5):455.e1-5.
- Kaimal AJ, Zlatnik MG, Cheng YW, Thiet MP, Connatty E, Creedy P, et al. Effect of a change in policy regarding the timing of prophylactic antibiotics on the rate of postcesarean delivery surgical-site infections. Am J Obstet Gynecol. 2008;199(3):310.e1-5.
- Owens SM, Brozanski BS, Meyn LA, Wiesenfeld HC. Antimicrobial prophylaxis for cesarean delivery before skin incision. Obstet Gynecol. 2009;114(3):573-9.
- Mowat J, Bonnar J. Abdominal wound dehiscence after caesarean section. Br Med J. 1971;2(5756):256-7.
- Marrs CC, Moussa HN, Sibai BM, Blackwell SC. The relationship between primary cesarean delivery skin incision type and wound complications in women with morbid obesity. Am JObstet Gynecol. 2014;210(4):319.
- Biswas KK. Why not Pfannenstiel's incision? Obstet Gynecol. 1973;41(2):303-7.
- Song AY, Askari M, Azemi E, Alber S, Hurwitz DJ, Marra KG, et al. Biomechanical properties of the superficial fascial system. Aesthet Surg J. 2006;26(4):395-403.
- Al-Benna S, Al-Ajam Y, Tzakas E. Superficial fascial system repair: an abdominoplasty technique to reduce local complications after caesarean delivery. Arch Gynecol Obstet. 2009;279(5):673-5.
- Hull DB, Varner MW. A randomized study of closure of the peritoneum at cesarean delivery. Obstet Gynecol. 1991;77(6):818-20.
- Pietrantoni M, Parsons Mt, O'brien WF, Collins E, Knuppel RA , Spellacy Wn. Peritoneal closure or non-closure at cesarean. Obstet Gynecol. 1991;77(2):293-6.
- Grundsell HS, Rizk DE, Kumar RM. Randomized study of non-closure of peritoneum in lower segment cesarean section. Acta obstetricia et gynecologica Scandinavica. 2009.
- Del Valle GO, Combs P, Qualls C, Curet LB. Does closure of Camper fascia reduce the incidence of post-cesarean superficial wound disruption? Obstet Gynecol. 1992;80:1013-6.
- Bohman VR, Gilstrap LC, Ramin SM, Little BB, Santos-Ramos R, Goldaber KG, et al. Subcutaneous tissue: to suture or not to suture at cesarean section. Infect Dis Obstet Gynecol. 1994;1(6):259–64.
- Chelmow D, Rodriguez EJ, Sabatini MM. Suture closure of subcutaneous fat and wound disruption after cesarean delivery: a meta-analysis. Obstet Gynecol. 2004;103(5):974-80.
- Ramsey PS, White AM, Guinn DA, Lu GC, Ramin SM, Davies JK, et al. Subcutaneous tissue reapproximation, alone or in combination with drain, in obese women undergoing cesarean delivery. Obstet Gynecol. 2005; 105(5):967-73.
- Mark KS, Alger L, Terplan M. Incisional Negative Pressure Therapy to Prevent Wound Complications Following Cesarean Section in Morbidly Obese Women A Pilot Study. Surg Innov. 2014;21(4):345-9.
- Sharm C, Ashok V, Anjali S, Meghna T, V.K. Mahajan, Suresh V, et al. A randomized controlled trial comparing cosmetic outcome after skin closure with ‘staples’ or ‘subcuticular sutures’ in emergency cesarean section. Arch Gynecol Obstet.2014;290(4):655-9.
- Mackeen AD, Khalifeh A, Fleisher J, Vogell A, Han C, Sendecki J, et al. Suture compared with staple skin closure after cesarean delivery: a randomized controlled trial. Obstet Gynecol. 2014;123(6):1169-75.
- Ali A, Essam A. Abdominoplasty combined with cesarean delivery: evaluation of the practice. Aesthet Plast Surg. 2011;35(1):80-6.
- Okan D, Woo K, Ayello EA, Sibbald G. The role of moisture balance in wound healing. Adv Skin Wound Care. 2007;20(1):39-53.
- Pastorfide GB, Gorgonio NM, Ganzon AR, Alberto RM. Zinc chloride spray--magnesium hydroxide ointment dual topical regimen in the treatment of obstetric and gynecologic incisional wounds. Clin Therapeut. 1988;11(2): 258-63.
- Al-Waili NS, Saloom KY. Effects of topical honey on post-operative wound infections due to gram positive and gram negative bacteria following caesarean sections and hysterectomies. Europ J Med Research. 1999;4(3):126-30.
- Phuapradit W, Saropala N. Topical application of honey in treatment of abdominal wound disruption. Aus New Zeal J Obstet Gynaecol. 1992;32(4):381-4.