Case Report
Successful Management of Hyaluronic Acid Infiltration Embolism in Face: A Case Report
Hector Cesar Durán Vega* and Alba Regina Camargo Goñi
*Corresponding author: Hector Cesar Durán Vega, Department of Plastic surgery, Regional General Hospital T1. Mexican Institute of Social Security (IMSS), Merida, Yucatan, Mexico,
Published: 16 Jan 2018
Cite this article as: Vega HCD, Goñi ARC. Successful
Management of Hyaluronic Acid
Infiltration Embolism in Face: A Case
Report. Clin Surg. 2018; 3: 1860.
Abstract
The use of Injectable Hyaluronic Acid (HA) in wrinkle filling and facial volumization is one of
the most commonly performed procedures in cosmetic dermatology practice. While vascular
complications are rare, techniques for their management and treatment should be known by any
physician performing the procedure. We present a case of a 39-year-old male who experienced
vascular occlusion of nasal alar region following HA injection. He experienced persistent pain,
tenderness and progressive color alteration of the skin on the injected area that extended to the
maxilar region of the same side. This case highlights the importance of immediate recognition
and treatment of vascular occlusion to improve patient outcomes and reduce risk of permanent
complications.
Keywords: Hyaluronic acid; Vascular complications; Hyaluronidase; Nasal alar necrosis;
Injectable fillers
Introduction
Hyaluronic acid based dermal fillers have been the most frequently used filling substances [1] over the past several years; its injection has become increasingly popular and is now the preferred treatment for physicians performing soft tissue augmentations, facial contouring [2] and correction of fat pad loss due to aging [3]. HA fillers have advantages such as longer lasting, less immunogenic reactions [4], and can be hydrolyzed by hyaluronidase enzyme [4,5]. According to recent data published by the American Society of Plastic Surgeons (ASPS) in 2014, HA fillers constituted 7.3% of all injectable dermal fillers. As the usage of these fillers is expanding, complications will likely increase [6]. Although all commercially available options for HA voluminization present good tolerance, there is no filling substance available that is totally devoid of risk, and even experimented professionals can come across reactions or complications [3]. Most complications associated with the use of HA fillers are rare and benign. The most severe and early occurring is tissue necrosis due to embolization of a specific vessel or obstruction by the filler material, which can cause ischemia and necrosis [4]. The management of complications must be well known to the specialist physician [3]. Maximizing injection technique and thorough understanding of potential complications and their management can help avoid, identify, and treat them if they occur [6].
Case Presentation
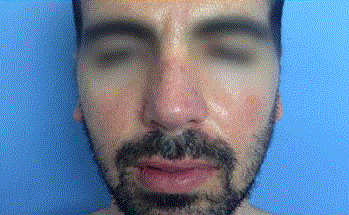
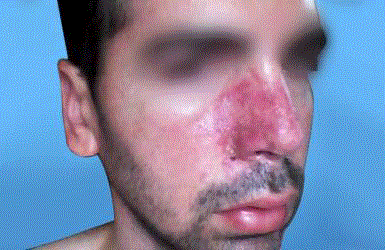
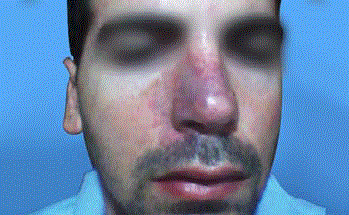
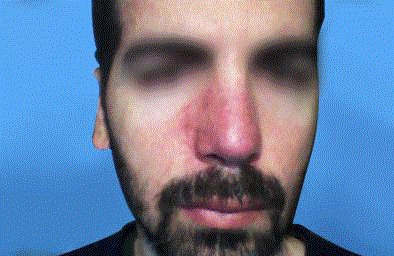
A thirty-nine year-old Israeli male, who had undergone two different nose reconstructive surgical operations, presents for a filling procedure with HA aimed to volumize the right alar region of the nose, which appeared slightly depressed compared to the left alar region, due to previous procedures. After informed consent was obtained and the patient was photographed, (Figure 1) a subcutaneous injection with 0.5 ml of HA (Belotero Volume, Merz Pharmaceuticals, Mexico) was performed on the right alar region of the nose. An hipoperfused area was observed in the injected zone that subsided within the next seconds of the application, which was suspended immediately. After applying massage to the affected area, the patient was released. Twelve hours after injection of HA, the patient experienced persistent pain and tenderness, and noticed progressive color alteration of the skin on the right nasal area which extended to maxilar region of the same side. (Figure 2) The diagnostic hypothesis a vascular complication due to intravascular infiltration of HA. HA aspiration was performed using a 25g cannula and a dose of 0.5 ml of lyophilized hyaluronidase (PBSerum, Mexico) was administered on the right nasal alar region, (Figure 3) presenting immediate pain relief and flattening of the area. A single dose of 8mg intravenous dexamethasone was indicated, salicylic acid 300 mg orally in one dose, as well as oral serratiopeptidase (Danzen, Hormona hyperbaric oxygen therapy once a day for ten sessions to improve blood perfusion and oxygenation. Twenty days after, the patient was reevaluated presenting a skin appearance improvement, completely pain free, without signs of necrosis (Figure 4).
Figure 1
Figure 2
Figure 3
Figure 4
Discussion
With HA dermal fillers, vascular complications (VCs) have been
a concern. Such complications can result from either intravascular
injection or the compressive effect of the filler on local vessels
[7,8]. In severe cases, VCs can cause extensive skin necrosis [5,8].
Facial regions prone to vascular side effects following dermal HA
filler injections correlate with the anatomy of superficial arteries
[2]. The main high risk areas for skin necrosis and embolization
are the glabella, nasal ala and dorsum of the nose [2,4,6]; as these
regions have limited collateral blood supply [5,7]. The most feared
and potentially serious complications are vascular in nature [7].
The patophysiology of vascular occlusion begins with immediate
changes visible in the vascular system: initial blanching, followed by
mottled discoloration called livedo reticularis (macular, violaceous,
net-like skin discoloration) [9], accompanied by pain, unless there
is a nerve block or local anesthetic blocking the pain pathways. The
resulting ischemia produces a dusky discoloration associated with
absent capillary refill after digital compressions [2,5,9]. The final stage
includes pus, scabs, scar tissue [10], and necrosis [11].
Vascular compromise as a result of hyaluronic acid filler injection
should be treated immediately. Signs of impending necrosis include
pain, prolonged blanching and coolness of the skin. Hyaluronidase
should be administered as soon as this complication occurs. There is
good evidence that tissue necrosis will be prevented or less severe the
sooner the hyaluronidase is injected [1]. In this context, the title of a
review by Almeida-Balassiano et al. [12] “Hyaluronidase: a necessity
for any dermatologist applying injectable hyaluronic acid” as well as
“Hyaluronidase in the office: a necessity for every dermatosurgeon
that injects hyaluronic acid” by Hirsch et al. [13] underscores the
importance of hyaluronidase in aesthetic practice [2].
Appropriate treatment should be initiated immediately upon
suspicion of vascular compromise [7,9]. Injection should be stopped,
and the injected area should be massaged. Immediate hyaluronidase
enzyme injection (every 150u of hyaluronidase can dissolve 1 ml
HA) [9] is crucial for cases that present early in order to minimize
the amount of tissue necrosis [6]. Systemic or topical steroids must
be administered to reduce associated inflammation, mitigating
the degree of injury [5,11]. Additionally start the patient on 300
mg of aspirin to prevent clot formation, and consider hyperbaric
oxygen therapy in cases where there is still necrosis after the initial
treatment [6,10]. Hyperbaric oxygen may prove helpful in treating
ischemic injuries, delivering oxygen deep into the skin to keep
tissues viable [7]; patients treated with hyperbaric oxygen along with
other methods described appear to do better than patients who had
not been so treated [9]. Serratiopeptidase has been found useful in
patients suffering from acute or chronic inflammatory disorders by
hydrolyzing bradikinin, histamine and serotonin responsible for the
edematic status. It reduces swelling and improves microcirculation
[14]. For remote vascular events emergency treatment is of high
importance [15]. The aim for treatment is dissolving the product
(HA), facilitating blood flow and promoting vasodilatation. Dayan et
al. [11] have suggested the use of hyaluronidase in all cases of vascular
compromise because of its edema-reducing benefits and theoretical
advantage in reducing the occluding vessel pressure when performed
in the first 24 hours after the isquemic event [12]. Its use may
reduce the size of the necrotic area [16] promoting the degradation
of injected HA improving the healing process [3]. A thorough
individual assessment and treatment plan should be instituted for
each patient [7]. Early diagnosis of vascular compromise and necrosis
after filler injections may improve the outcome of wound healing [4].
Prevention is essential. Practitioners are to be equipped with sufficient
anatomy knowledge, especially mapping face vessel distribution [10].
It is important to be cautious when injecting high risk anatomic areas [7].
Conclusion
When working with dermal HA fillers, hyaluronidase should always be available; severe complications of vascular necrosis following accidental intravascular HA filler injection must be immediately treated. Any patients suspected to experience VCs should be given early management trying to salvage the skin. Treatment begins with diagnosis of the event and continues with administration of hyaluronidase, along with warm compresses and massage of the area, aspirin, and steroids. If ischemia is still present, evidence suggests the use of hyperbaric oxygen therapy. Although there is not enough evidence, Serratiopeptidase has been successfully used for its antiinflammatory and antiedemic activity in a number of tissues. Finally, it’s important to remember that an early identification and a prompt intervention can significantly decrease the risk of long term sequeleae due to VCs.
References
- King M. The Use of Hyaluronidase in Aesthetic Practice. Aesthetic Complications Expert Group. 2016;2:1-9.
- Buhren BA, Schrumpf H, Hoff NP, Bölke E, Hilton S, Gerber PA. Hyaluronidase: from clinical applications to molecular and cellular mechanisms. Eur J Med Res. 2016;21:5.
- Ramos Nogueira Guerra Neri S, Sant´Anna Addor FA, Brasil Parada M, Schalka S. The use of hyaluronidase in complications caused by hyaluronic acid for volumization of the face: a case report. Surg Cosmet Dermatol. 2013;5(4):364-6.
- Manafi A, Barikbin B, Manafi A, Hamedi ZS, Ahmadi Moghadam S. Nasal alar necrosis following hyaluronic Acid injection into nasolabial folds: a case report. World J Plast Surg. 2015;4(1):74-8.
- Cohen BE, Bashey S, Wysong A. The Use of Hyaluronidase in Cosmetic Dermatology: A Review of the Literature. J Clin Investigat Dermatol. 2015;3(2):7.
- Abduljabbar MH, Basendwh MA. Complications of hyaluronic acid fillers and their managements. J Dermatology and Dermatologic Surg. 2016;20:100-6.
- Beleznay K, Humphrey S, Carruthers J, Carruthers A. Vascular Compromise from soft Tissue Augmentation; Experience with 12 Cases and Recomendations for Optimal Outcomes. J Clin Aesthet dermatol. 2014;7(9):37-43.
- Chen Q, Liu Y, Fan D. Serious Vascular Complications after Nonsurgical Rhynoplasty: A Case Report. Plast Reconstr Surg Glob Open. 2016;4:683.
- DeLorenzi C. Complications of injectable fillers, part 2: vascular complications. Aesthet Surg J. 2014;34(4):584-600.
- Fan X, Dong M, Li T, Ma Q, Yin Y. Two Cases of Adverse Reactions of Hyaluronic Acid-based Filler Injections. Plast Reconstr Surg Glob Open. 2016;4:1112.
- Kim JH, Ahn DK, Jeong HS, Suck Suh I. Treatment Algorithm of complications after Filler Injection: Base don Wound Healing Process. J Korean Med Sci. 2014;29:176-82.
- Almeida Balassiano LK, Feliz Bravo BS. Hyaluronidase: a necessity for any dermatologist applying injectable hyaluronic acid. Surg Cosmet Dermatol. 2014;6(4):338-43.
- Hirsch RJ, Brody HJ, Carruthers JD. Hyaluronidase in the office: a necessity for every dermatosurgeon that injects hyaluronic acid. J Cosmet Laser Ther. 2007;9(3):182-5.
- UmaMaheswari T, Hemalatha T, Sankaranarayanan P, Puvanakrishnan R. Enzyme Therapy: Current Perspectives. Indian J Exp Biol. 2016;54(1):7-16.
- Landau M1. Hyaluronidase Caveats in Treating Filler Complications. Dermatol Surg. 2015;41 Suppl 1:S347-53.
- Cavallini M, Gazzola R, Metalla M, Vaienti L. The Role of Hyaluronidase in the Treatment of Complications from Hyaluronic Acid Dermal Fillers. Aesthet Surg J. 2013;33(8):1167-74.