Case Report
A Missed Case of a Strangulated Right-Sided Diaphragmatic Hernia Containing Perforated Small Bowelin an Elderly Patient
Caitlin T Yeo* and Wiley Chung
Department of Surgery, Queen’s University, Kingston, Canada
*Corresponding author: Caitlin T Yeo, Department of Surgery, Queen’s University, Kingston, Canada
Published: 16 Jan 2018
Cite this article as: Yeo CT, Chung W. A Missed Case
of a Strangulated Right-Sided
Diaphragmatic Hernia Containing
Perforated Small Bowelin an Elderly
Patient. Clin Surg. 2018; 3: 1858.
Abstract
We present a patient who was admitted with hypoxemia and was thought to have a right-sided
empyema secondary to a parapneumonic effusion. She was treated for two weeks prior to being
transferred for a Thoracic Surgery consultation. On review of imaging she actually had a right
diaphragmatic hernia with strangulated, perforated small bowel. She was initially stable but during
transfer developed increasing respiratory requirements requiring emergent surgery. Atraumatic
adult right-sided incarcerated diaphragmatic hernia with bowel perforation is extremely rare with
only two other cases reports involving colon and none involving small bowel. We describe her
presentation, management, and post-operative considerations.
Keywords: Diaphragmatic hernia; Perforated small bowel
Introduction
Diaphragmatic hernias are either congenital or acquired from traumatic diaphragmatic injury. Congenital Diaphragmatic Hernias (CDH) occurs in approximately 1 in 3000 live births, with 85% found on the left side [1-3]. They are usually diagnosed in the neonatal period, as early at 16 weeks gestational age, and require early surgical intervention [4]. Right-sided CDH are extremely rare due to the earlier fusion of the right diaphragm and the protective location of the liver [5]. We present a case report of a missed right-sided CDH involving strangulated small bowel with perforation resulting in a fecopneumothorax.
Case Presentation
An 81-year-old female with a past medical history of hypertension, hypothyroid and reflux
disease is presented to the emergency department at a community hospital with nausea, vomiting,
right-sided chest pain, and dyspnea. Her past surgical history included a hysterectomy and partial
mastectomy. She had no known past high-impact blunt or penetrating trauma that would predispose
her to a missed right diaphragmatic injury giving rise to an acquired diaphragmatic hernia. Her
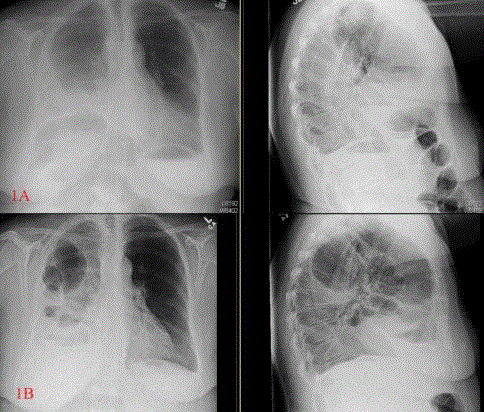
initial chestx-rays showed a right lower lobe consolidation with an associated effusion (Figure 1A).
A CT was done at the time of presentation for a concurrent bowel obstruction, which was thought to
be an ileus secondary to a thickened and inflamed right hemi-diaphragm adjacent to loops of small
bowel. She was managed in the community for two weeks, where they attempted a thoracentesis
that drained minimal fluid. She continued to decompensatewith an increasing white count despite
antibiotic therapy. Repeat imaging (Figure 1B) showed a worsening hemopneumothorax and it
was felt that the patient would benefit from a thoracic surgery referral for tube thoracotomy and
possible decortication. Vital signs on transfer were the following: blood pressure 143/65, heart rate
86, respiratory rate 20, and 93% oxygen saturation on 50% Ventimask. The images were reviewed
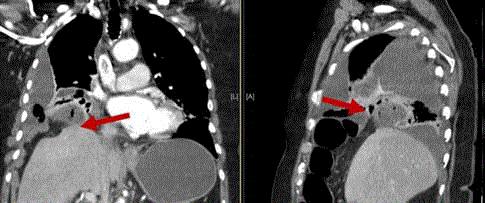
by our thoracic surgeon and interventional radiologist, and it became apparent that this was not an
empyema secondary to a parapneumonic effusion, but rather a segment of strangulated small bowel
within a diaphragmatic hernia that had perforated (Figure 2). The only prior chest imaging available
was a chest x-ray from 2013 which did not show evidence of the diaphragmatic hernia.
The decision was made to take the patient urgently to the operating room for an exploratory
laparotomy and right thoracotomy. Exploratory laparotomy revealed an approximately 3 cm x2cm
diaphragmatic defect, posterior and lateral to the falciform ligament. A loop of small bowel was
incarcerated through the diaphragmatic defect. On our attempt to reduce the small bowel, the
proximal small bowel was necrotic and disintegrated. We quickly tied off the proximal small bowel to prevent further spillage of stool. We measured the small bowel within the abdominal cavity. There was more than 200cm of small
bowel proximal to the hernia, so we made the decision to divide the
distal small bowel at the level of the hernia as it could not be reduced
into the abdomen. We reduced the small bowel remnant up into the
chest cavity, and proceeded to suction over 2L of feculent material
from her chest via the hernia defect. During this time, she developed
ventricular tachycardia and required electrical cardioversion. Once
spillage of feculent material was controlled, a sponge was placed
within the chest, with the intention of removing the small bowel
and sponge during the thoracotomy. The diaphragmatic hernia was
closed with 1.0 Ethibond sutures in an interrupted fashion. A side to
side anastomosis of the small bowel was created with healthy bowel.
The abdomen was irrigated and a Jackson-Pratt drain left in the right
subdiaphragmatic space.
We then repositioned the patient in the left lateral decubitus
position and performed a right serratus-sparing posterolateral
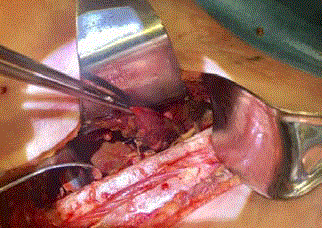
thoracotomy. Irrigation and decortication was carried out along with
removal of the sponge and necrotic piece of small bowel (Figure 3).
Our primary repair of the diaphragm was intact and there was no
residual defect. There was good lung re-expansion post-decortication
and multiple chest tubes were placed. Our patient was kept intubated
and admitted to ICU post-operatively. After a prolonged hospital stay
and rehabilitation, the patient was discharged home.
Figure 1
Figure 1
(A) Initial X-rays showing possible right lower lobe consolidation
with pleural effusion. (B) Repeat X-rays showing air fluid levels in the right
chest.
Figure 2
Figure 3
Figure 3
Intraoperative photograph of right serratus-sparing posterolateral
thoracotomy with small bowel evident within thoracic cavity.
Discussion
As our patient had no historyto support a traumatic diaphragmatic
hernia, this was most likelycongenital in etiology.The location of the
diaphragmatic hernia was consistent with a Morgagni hernia. The lack
of visualization on prior imaging was possibly due to the small defect
size and protective effect of theliver precluding abdominal contents
from herniating into the right thoracic cavity. Although respiratory
symptoms in a patient presenting with a bowel obstruction are usually
secondary to aspiration, this case demonstrates the importance
of considering an incarcerated diaphragmatic hernia within the
differential. Signs and symptoms that should have prompted an earlier
referral to Thoracic Surgery include her lack of response to antibiotic
treatment, inability to perform a successful thoracentesis, complex
and loculated appearance of her hydropneumothorax, and prolonged
GI symptoms. In this particular case, the loops of bowel were sitting
anterior to the liver, which is unusual, and can be seen in the initial
lateral films (Figure 1A). There was also evidence of a transition point
with dilated loops leading up to the diaphragm with collapsed bowel
distally, along with thickening of the bowel wall and stranding which
would be contradictory to a diagnosis of an ileus. These were some
radiographic features that should have prompted a closer review of the
case. We decided to start with a laparotomy approach initially because
we wanted to control the source of sepsis. As in any strangulated or
perforated hernia, proximal control of the bowel and its contents is
desired. If we chose to start with a thoracotomy first, the abdomen
would have been filling with stool and grossly contaminated while we
completed the decortication. If the hernia was merely incarcerated
and not perforated, a thoracotomy as an initial approach would not
be unreasonable. The posterolateral thoracotomy was required in
order to reach and appropriately decorticate the apical and posterior
surfaces of the lung. A thoracoabdominal incision would have been
too anterior and inferior. There was no role for a VATS procedure in
an unstable, grossly contaminated patient.
The post-operative recovery requires a multidisciplinary
approach. As with our patient, ICU care and ventilation are pertinent.
These patients require chest physiotherapy, nebulized saline to
mobilize secretions, and intense bronchopulmonary toilet. There is
a low threshold for performing a tracheostomy if there is prolonged
ventilation or difficulty managing secretions. Management of GI
symptoms with a nasogastric tube, electrolyte imbalance from GI
losses, total parenteral nutrition for prolonged nil per os, and broad
spectrum antibiotics should be considered. Interval CT is important
in re-evaluating the empyema and the need or position of each chest
tube.
Conclusion
Strangulated right-sided diaphragmatic hernia containing perforated small bowel is extremely uncommon. One needs to include this on the differential when patients present with symptoms of a small bowel obstruction in conjunction with respiratory distress and an effusion on imaging.Unusual anatomic position should prompt closer evaluation rather than assumption of congenital deviance.
References
- Granier V, Coche E, Hantson P, Thoma M. Intrathoracic caecal perforation presenting as dyspnea. Case Rep Med. 2010;2010:296730.
- Slesser AA, Ribbans H, Blunt D, Stanbridge R, Buchanan GN. A spontaneous adult right-sided Bochdalek hernia containing perforated colon. JRSM Short Reports. 2011;2(7):54.
- Langham Jr MR, Kays DW, Ledbetter DJ, Frentzen B, Sanford LL, Richards DS. Congenital diaphragmatic hernia. Epidemiology and outcome. ClinPerinatol. 1996;23(4):671-88.
- Bohn D. Congenital diaphragmatic hernia. Am J Respir Crit Care Med. 2002;166(7):911-5.
- Bianchi E, Mancini P, De Vito S, Pompili E, Taurone S, Guerrisi I, et al. Congenital asymptomatic diaphragmatic hernias in adults: a case series. J Med Case Rep. 2013;7(1):125.