Case Report
Successful Surgical Treatment of a Necrotizing Fasciitis due to Klebsiella Pneumonia Following Open Prostatectomy
Şener Balas1, Fatma Yıldırım2*, Mücait Ünal1, Alper Gök3 and Emre İnözü4
1Department of General Surgery, Dışkapı Yıldırım Beyazıt Research and Education Hospital, Turkey
2Department of Pulmonary and Critical Care Medicine, Dışkapı Yıldırım Beyazıt Research and Education Hospital,
Turkey
3Department of Urology, Dışkapı Yıldırım Beyazıt Research and Education Hospital, Turkey
4Department of Plastic and Reconstructive Surgery, Dışkapı Yıldırım Beyazıt Research and Education Hospital,
Turkey
*Corresponding author: Fatma Yıldırım, Department of Pulmonary and Critical Care Medicine, Dışkapı Yıldırım Beyazıt Research and Education Hospital, 06500 Besevler, Ankara ,Turkey
Published: 09 Jan, 2018
Cite this article as: Balas Ş, Yıldırım F, Ünal M, Gök A,
İnözü E. Successful Surgical Treatment
of a Necrotizing Fasciitis due to
Klebsiella Pneumonia Following Open
Prostatectomy. Clin Surg. 2018; 3:
1850.
Abstract
We report a case of early NF associated with Klebsiella pneumoniae infection in a 61-yearold man who underwent to open prostatectomy 10 days ago. The patient presented with acute, painful, erythematous, and edematous skin lesions of his left flank. The patient underwent surgical exploration, and a diagnosis of necrotizing fasciitis was confirmed by pathological evidence of necrosis of the fascia and neutrophil infiltration in tissue biopsies. Cultures of fascial tissue biopsies and blood samples were positive for Klebsiella pneumoniae. Following extensive debridement, a VAC system was applied to the large open wound and successfully contributed to wound bed cleansing, which was followed by surgery for skin grafting.
Introduction
Necrotizing Fasciitis (NF) is an aggressive soft tissue infection that involves the deep fascia and is characterized by the extensive deterioration of the surrounding tissue. It can be seen postoperatively as a complication. Immediate recognition and aggressive treatment, including debridement and systemic antibiotics, are mandatory for the successful management of NF.
Case Presentation
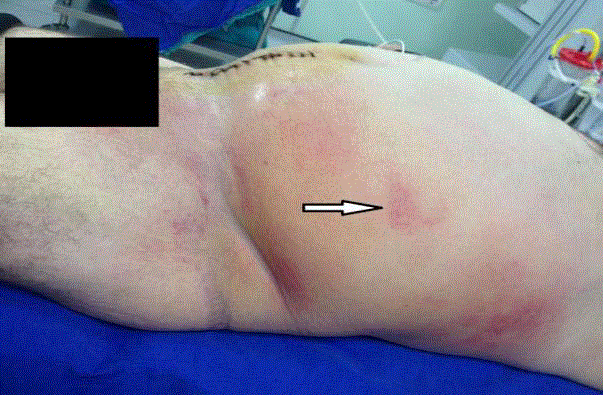
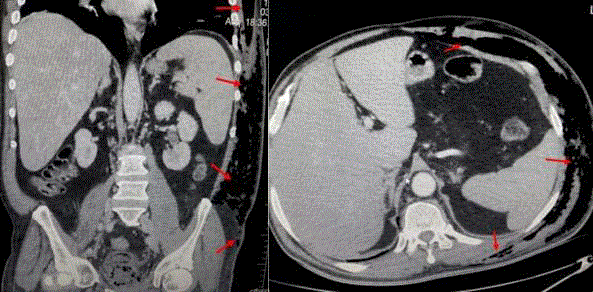
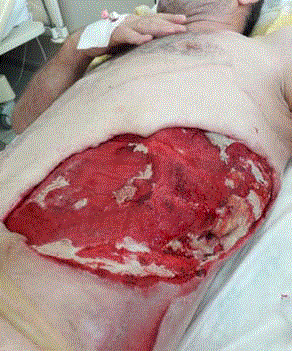
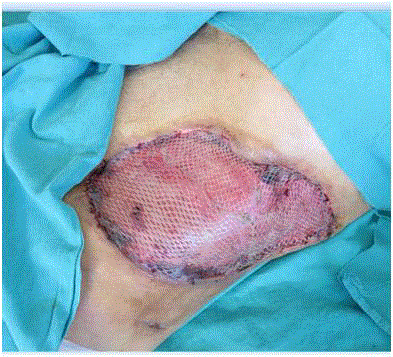
A 61-year-old male with a prior history of prostatic hyperplasia and bladder calculi underwent open prostatectomy. His past medical history included poorly controlled type II diabetes mellitus and femoral neck fracture following a traffic accident 10 years previously. On postoperative day 7 there were no complications relating to his surgery seen and the patient was discharged. On the tenth day after surgery he presented to the emergency department with acute, painful, erythematous, and edematous skin lesions over his left flank which rapidly progressed to the lower abdominal and pelvic region and upper part of left leg. He had been experiencing these symptoms for three days, with the left flank and abdominal swelling becoming progressively worse. He had also been pyrexial for three days. On physical examination the patient was systemically unwell with a heart rate of 139/min, blood pressure of 87/36 mmHg and temperature 38.8°C indicating septic shock. Physical examination revealed a large fluctuant mass over the left flank, abdominal and inguinal region. On the flank region this mass exhibited blackish discoloration and skin necrosis. Swelling and crepitus extended to the abdominal and chest wall (Figure 1). The initial blood investigations revealed a white cell count (WBC) of 19.0 × 109/L, C - reactive protein (CRP) of 147 mg/L, procalcitonin 100 ng/mL, hemoglobin (Hb) of 10.9 g/dL and platelets 259 x 103/mcL. Biochemistry results were as follows: sodium 129 mmol/L, potassium 4.2 mmol/L, chloride 109 mmol/L, blood urea nitrogen 47 mg/dL, creatinine 1.2 mg/dL, glucose 310 mg/dL, and lactate 3.5 mmol/L. CT of the abdomen and chest revealed extensive subcutaneous emphysema throughout the left flank, left abdominal and chest wall, and throughout the superficial spaces left side of the neck (Figure 2)|. One hour later, the patient was taken to the operating theatre for debridement and was discovered to have necrotic deep muscle tissue and fascia. Intra operative biopsies confirmed the diagnosis of necrotizing fasciitis, with necrotic and purulent material found in the dermis, subcutaneous tissues, and fascia. He received vancomycin before surgical debridement; fluconazole and carbapenem were added after debridement. Then the patient was transferred to Intensive Care Unit (ICU) for hemodynamic monitoring and ongoing management. Central venous catheterization was performed and fluid resuscitation commenced. Despite adequate (30 mL/kg) intravenous fluid replacement, the patient remained hypotensive (mean arterial pressure < 60 mmHg) and thus vasopressor therapy and invasive blood pressure monitoring were started. After a second surgical debridement, vasopressors were stopped. The APACHE II score of the patient at this stage was 24. Following the second debridement the patient was left with a significant soft tissue defect, and negative pressure was applied at 125 mm Hg with Vacuum Assisted Closure (VAC) to facilitate wound management. The VAC system had to be reapplied in ICU twice weekly. Cultures of blood, urine, and surgical specimens all grew Klebsiella pneumonia resistant to ampicillin but sensitive to carbapenem (imipenem). Fluconazole and Linezolid treatments were discontinued and the patient’s condition improved sufficiently to allow cessation of sedation and extubation. Sequential debridement and wound assessment demonstrated marked improvements with development of visible granulation tissue following the application of the VAC system (Figure 3). Total parenteral nutrition was started to support patient's nutritional status. After 16 days on ICU the patient was transferred to the general ward to complete his course of parenteral antibiotic therapy. Application of VAC system also continued on the ward. The patient underwent skin grafting after 21 days of hospitalization with grafts obtained from the anterolateral aspect of the left thigh resulting in successful and complete coverage of the previous soft tissue defect (Figure 4).
Figure 1
Figure 2
Figure 2
Computed tomography scan demonstrated soft tissue inflammation
and edema with multiple gas bubble formations.
Figure 3
Figure 3
Wound appearance during the Vacuum-Assisted Closure (VAC)
exchange (post- operative day 21). Healthy granulation tissue is present
throughout the wound.
Figure 4
Figure 4
Appearance of wound after the skin grafting (post- operative day
13). All authors declare that they have no conflict of interests.
Discussion
Necrotizing Fasciitis (NF) caused by Klebsiella pneumonia after open prostatectomy in our case was treated successfully, in accordance with current therapeutic concepts, with aggressive hemodynamic stabilization, broad spectrum parenteral antibiotic administration, and urgent wide surgical debridement to remove devitalized tissue as well as the application of a VAC dressing. Necrotizing fasciitis is an acute, progressive, invasive, and severe infection that spreads along fascial planes, producing inflammation and necrosis of the adjacent muscle, subcutaneous fat, and skin. The incidence of NF is about 4 per million world population, and mortality rates as high as 73% are reported [1]. NF may be classified according to microbiological aetiology (polymicrobial or monomicrobial), anatomy, and depth of infection. Polymicrobial NF (a mix of aerobic and anaerobic bacteria) mostly occurs in immune compromised individuals. Monomicrobial NF (caused mainly by Streptococcus pyogenes but also by Klebsiella pneumonia) is less common and may affect healthy individuals who often have a history of trauma, or recent surgical intervention as seen in our case [2]. Initial empiric broad-spectrum antibiotic coverage is needed in cases of NF, directed against aerobic Gram-positive cocci, Gram-negative rods, and a variety of anaerobes as recommended by the Infectious Diseases Society of America [3]. We started with carbapenem as an initial treatment given our patients' previous open prostatectomy operation. Our patient had a history of open prostatectomy operation and he had been hospital sated for one week prior to becoming unwell with NF. We isolated Klebsiella pneumonia as a causative agent in blood, urine and as well as tissue cultures in our patient. Klebsiella pneumonia is an enteric gram-negative rod that is frequently implicated in hospital-acquired infections such as urinary tract infections, pneumonia, septicemia, and intra-abdominal infections. Given the recent hospitalisation of our patient it is highly likely that his Klebsiella pneumonia was hospital-acquired. Monie et al. [4] reported a case of a 92-year-old-woman with NF of the leg caused by Klebsiella pneumonia. The patient had been hospitalized in a geriatric unit but had no risk factors for the development of NF. In this particular case the Klebsiella pneumonia strain was resistant to carbapenems and the patient unfortunately died due to septic shock 9 hr after the onset of symptoms [4]. Rana et al. [5] reported a case series of 6 liver transplant recipients with NF also caused by carbapenemase producing Klebsiellapneumonia. In this series half of the patients died, and the author’s emphasized the need for heightened suspicion of Klebsiellapneumonia NF, as early recognition and debridement may improve survival. It is likely that our patient did not have a carbapenemase producing Klebsiellapneumonia, which combined with early surgical debridement within one hour, may have contributed to an improved likelihood of survival [5]. Commonly reported risk factors for NF with Klebsiella pneumonia include diabetes mellitus and chronic liver disease. Our patient had a past history of type II diabetes mellitus that had been treated with oral hypoglycemic agents but had poor blood sugar control. It is thought that diabetes mellitus could cause susceptibility to NF via tissue hypoxia resulting from diabetic vascular disease, and also the underlying immunodeficiency associated with poorly controlled disease [6]. After open prostatectomy complications such asanastomotic leakage and anastomotic stricture are well recognized. In a study by Jacobsen et al. [7] diabetes mellitus, advanced age and large prostate volume are all recognized risk factors for anastomotic leakage. Additionally the end pelvic fascia and neurovascular bundle are peeled off during prostatectomy which may leave a residual fascial defect postoperatively [7,8]. Both anastomotic complications and fascial opening may contribute to the development of NF in these patients although there are few reported NF cases after urological interventions in the literature [9]. There is a growing body of evidence to indicate that application of negative-pressure wound therapy is useful in the treatment of patients with NF. The cyclical application of negative pressure can accelerate wound healing as optimized blood flow improves local oxygenation, promotes fibroblast stimulation and granulation formation, decreases local tissue edema, and accelerates removal of excessive fluid from the wound bed. This in turn facilitates the removal of bacteria from the wound [10,11]. In our case, no adverse events occurred in association with VAC usage, indicating that VAC facilitated primary closure of a large wound. Several studies have suggested that the most important factors influencing mortality from NF are the time to first surgical intervention and adequacy of surgical debridement [12]. Therefore early recognition and diagnosis of NF is essential to facilitate early surgical intervention and thus decrease morbidity and mortality. CT scan and MRI are sensitive in the diagnosis of NF and the differential diagnosis of other causes of soft tissue infection, such as abscesses [13]. In our case, the presence of bullae and gas on CT scan was diagnostic. In summary we feel that our case report highlights an unusual presentation of NF caused by Klebsiella pneumonia. Although it is a rare condition and it should be considered in the differential diagnosis of infection after urological operations, such prostatectomy as in our case, or any procedure where risk factors may be present. We also feel that our report reinforces the need for early and decisive surgical intervention once the diagnosis has been made to improve mortality and patient outcomes.
References
- Trent JT, Kirsner RS. Necrotizing fasciitis. Wounds. 2002;14(8):284-92.
- Giuliano A, Lewis F Jr, Hadley K, Blaisdell FW. Bacteriology of necrotizing fasciitis. Am J Surg. 1977;134(1):52-7.
- Stevens DL, Bisno AL, Chambers HF, Dellinger EP, Goldstein EJ, Gorbach SL, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the infectious diseases society of America. Clin Infect Dis. 2014;59(2):147-59.
- Monié M, Drieux L, Nzili B, Dicko M, Goursot C, Greffard S, et al. Klebsiellapneumoniae necrotizing fasciitis of the leg in an elderly French woman. Clin Interv Aging. 2014;9:1171-4.
- Rana MM, Sturdevant M, Patel G, Huprikar S. Klebsiella necrotizing soft tissue infections in liver transplant recipients: a case series. Transpl Infect Dis. 2013;15(4):E157-63.
- Mc Ardle P, Gallen I. Necrotizing fasciitis in diabetics. Lancet. 1996;348(9026):552.
- Jacobsen A, Berg KD, Iversen P, Brasso K, Røder MA. Anastomotic complications after robot-assisted laparoscopic and open radical prostatectomy. Scand J Urol. 2016;50(4):274-9.
- Ferretti M, Phillips J. Prostatectomy for benign prostate disease: open, laparoscopic and robotic techniques. Can J Urol. 2015;22(1):60-6.
- Koukouras D, Kallidonis P, Panagopoulos C. Fournier's gangrene, a urologic and surgical emergency: presentation of a multi-institutional experience with 45 cases. Urologia Internationalis. 2011;86(2):167-72.
- Phelps JR, Fagan R, Pirela-Cruz MA. A case study of negative pressure wound therapy to manage acute necrotizing fasciitis. Ostomy Wound Manage. 2006;52(3):54-9.
- Huang C, Leavitt T, Bayer LR, Orgill DP. Effect of negative pressure wound therapy on wound healing. Curr Probl Surg. 2014;51(7):301-31.
- Bilton BD, Zibari GB, McMillan RW, Aultman DF, Dunn G, McDonald JC. Aggressive surgical management of Necrotizing fasciitis serves to decrease mortality; A retrospective study. Am Surg. 1998;64(5):397-401.
- Young MH, Aronoff DM, Engleberg NC. Necrotizing fasciitis: pathogenesis and treatment. Expert Rev Anti Infect Ther. 2005;3(2):279-94.