Research Article
Complications of Minimal Invasive Surgery for Rectal Cancer - A Systematic Review
Benjamin Sejr Thinggaard* and Sharaf Karim Perdawood
Department of Surgery, Slagelse Hospital, Denmark
*Corresponding author: Benjamin Thinggaard, Department of Surgery, Slagelse Hospital, Denmark
Published: 18 Dec 2017
Cite this article as: Thinggaard BS, Perdawood SK.
Complications of Minimal Invasive
Surgery for Rectal Cancer - A
Systematic Review. Clin Surg. 2017; 2:
1818.
Abstract
Background: Total Mesorectal Excision (TME) is widely accepted as the standard surgical treatment
for mid and low rectal cancer. The Robotic (RoTME) and Laparoscopic (LaTME) approaches to treat
rectal cancer are shown to be feasible. Transanal TME (TaTME) is the most recent minimal invasive
approach with promising results. We aimed to review the peri and postoperative complications
associated with the three approaches.
Methods: A systematic search in the PubMed and Embase databases was performed. Both authors
assessed the studies for eligibility. Clinical randomized as well as non-randomized studies published
during the last six years were included.
Results: In total 39 studies (8094 patients) met the inclusion criteria. The LaTME had low rates of
urinary complications, high rates of wound infection and intraabdominal abscesses. The RoTME
had high rates of anastomotic leakages, but low rates of several other complications like; ureter and
bladder injuries, bleeding and thirty-day mortality. The anastomosis performed more efficiently
after TaTME with lower rates of anastomotic leakages, but higher rates of bleeding and 30-days
mortality.
Conclusion: The procedures each performed well in relation to the different complications however
further research especially concerning TaTME and RoTME is needed. Focus on which procedure
that best treats a specific tumor stage, tumor location and type of patient could possibly reduce
complications and postoperative mortality in the future.
Keywords: Rectal cancer; Complications; Total mesorectal excision; Transanal; Robot-assisted
surgery; Laparoscopic surgery
Introduction
Rectal cancer is the fifth most common type of cancer in Denmark and less than 2 out of 3 will survive more than 5 years after diagnosis [1]. Significant advances have been achieved in the treatment of rectal cancer over the past three decades. The introduction of TME [2], and improved preoperative diagnostic work-up have improved the short- and long-term results [3,4]. TME is widely accepted as the standard surgical treatment for mid and low rectal cancer [3-5]. It is general acknowledged that the laparoscopic approach for the treatment of rectal cancer has more advantages than open surgery with faster recovery, shorter hospital stays, decreased blood loss, infection rates and multi-organ failure as well as comparable long-term outcomes [6-11]. However, LaTME is a technically difficult procedure in obese patients, males and patients with a narrow pelvis [12,13]. RoTME is a feasible approach with potential advantages compared with laparoscopy and open surgery [14-17], and is reported to be equivalent to laparoscopic surgery with respect to the shortterm perioperative and oncologic outcomes [15,18,19]. However, the true cost-benefit advantages still are questionable [12,20,21], and results of a randomized trial are awaited [22]|. As the newest approach, TaTME shows promising results as a safe and effective technique with acceptable shortterm outcomes [23,24]. Perioperative complications play an important role in the adoption of the appropriate approach, like both the pathological outcomes and the complication rates do. The aim of this review is to highlight common and severe reported complications associated with LaTME, RoTME and TaTME.
Materials and Methods
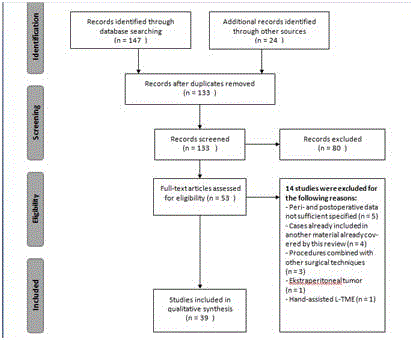
A systematic search was conducted in PubMed and Embase databases on October 7, 2016 using the following words separately and in combination: “complication”, “surgery”, “rectal”, “cancer”, “total mesorectal excision”, “transanal”, “TaTME”, “laparoscopic” and “robotic”. Only full text articles in English were included. The search and the review processes were undertaken by the two authors using the PRISMA guidelines [25]. The search identified 147 clinical trials. After crosschecking the reference lists 24 additional articles were retrieved. After removing duplicates 133 articles remained and abstracts were read systematically for eligibility. Only articles published during the last six years were included, as all these three approaches have been practiced during this period. All prospective and retrospective randomized and non-randomized studies concerning TME were included. Articles not sufficiently specifying data about complications and studies combining the procedure with other surgical techniques were excluded. We focused on the following intraoperative and postoperative complications; bleeding, Estimated Blood Loss (EBL), need for blood transfusion, anastomosed leakage, urine retention and complication, bladder and urethral injury, urinary tract infection, wound infection, bowel injury, intraabdominal and pelvic abscess and 30-days mortality. In total, we included 39 studies corresponding to 8094 patients. See the PRISMA flow diagram for detail (Figure 1).
Figure 1
Results
The included studies were single center as well as multicenter
clinical studies, prospective or retrospective, including several
randomized studies. Numerous studies were comparative or casematched
studies, where data originally were compared with open
surgery [7-10,13,26-30]. In six studies, LaTME was compared with
RoTME while four studies compared LaTME and TaTME [5,12,15,31-
37]. The rest focused on a single procedure.
The extracted data are presented systematically in Table 1.
Indications
The pathology was cancer in all studies except Penna et al. [23],
who included 86 (out of 720) benign cases. All cancer stages were
included though Yamamoto et al. [38] and Fujii et al. [39] only
included stage Tis-T2 tumors, Fleshman et al. [29] included T2-T3
and Baik et al. [28] included T1-T3. Rouanet et al. [40] selected men
with narrow pelvis, fatty mesorectum, high BMI, large prostate and
presence of fibrosis. Neither if the operation had to be converted into
open surgery nor the experience of the surgeon effected if a study
was included or not. Five studies only reported patients treated with
neoadjuvant therapy while several studies included both cases with
and without neoadjuvant treatment [7,28,29,31,35,36,41-43].
Peri and postoperative data
Bleeding and estimated blood loss (EBL): Fernandez-Hevia et
al. [37] reported one readmission in the LaTME group because of
hemorrhage, while Cho et al. [12] reported two conversions to open
surgery in the LaTME group due to intractable major vessel bleeding.
Van der Pas et al. [9] reported conversion in six out of 22 cases
because of bleeding. Hellan et al. [16] reported three cases of bleeding
after RoTME. Chen et al. [35] subdivided bleeding into presacral
bleedings and other bleedings. Another two studies subdivided as
intraabdominal or anastomotic bleeding [24,38]. Two studies found
no significant difference in EBL between TaTME and LaTME [35,36]
and three studies found no significant difference between RoTME
and LaTME in bleeding or EBL [31,32,44]. Perdawood reported
a lower blood loss for TaTME than LaTME [5]. Penna et al.[23]
reported an intraoperative blood loss during TaTME of less than
100ml in 61% of the cases, and furthermore reported six cases (1%) of
bleeding more than one liter. Serra-Aracil et al. [45] reported 158 ml
in EBL after TaTME, and proclaimed it lower than the laparoscopic
group of the COLOR II trail. Ielpo et al. [33] reported the highest
EBL=280 ml for RoTME (range 0 ml to 4000 ml) while Park et al. [34]
reported the lowest EBL=77.6 ml (range 0 ml to 700 ml). Two LaTME
studies reported EBL of 28 ml and 20 ml respectively [38,39]. Overall,
2.9% of the patients had perioperative bleeding during TaTME and
a general average EBL at 107.9 ml for every case 1.1% of the patients
had perioperative bleeding during RoTME and an average EBL at
143.7 ml. The LaTME studies reported 1.4% of patients with bleeding
and an average EBL at 115.1 ml. See Table 2 for calculations of the
percentage and an overview.
Need for blood transfusion: Approximately 20% required blood
transfusion after TaTME in Rouanet et al. [40]. Lacy et al. [24]
reported three blood transfusions because of anemia and one due
to hemorrhage after TaTME. Another study reported 1.6 % of cases
requiring blood transfusion after LaTME [38]. Overall, 5% of patients
undergoing TaTME, 2% of patients after RoTME and 2.3% after
LaTME had peri or postoperative blood transfusion.
Anastomotic leakage: Five studies reported the diagnosis as a
clinical suspicion where different criteria were reported; fever and
pain, pus, gas or fecal discharge from the drain, pelvic abscess and
peritonitis [9,10,31,33,46]. After TaTME, Penna et al. [23] reported
32 cases identified early and eight cases identified after 30 days.
Perdawood reported four patients readmitted after TaTME and one
after LaTME because of anastomotic leakage [5]. Penninckx et al.
[26] subdivided leakage as: minor (14 cases) and major (30 cases)
leaks after LaTME. Schiphorst et al. [30] reported four out of 46 cases
leading to reoperation or fistula as Hu et al. [7] reported 3.1% of
leakage and concluded that LaTME after neoadjuvant CRT is a safe
procedure. Cho et al. [12]| reported no early postoperative difference
between LaTME vs. RoTME while Chen et al. [47] reported no
statistically significant difference in leakage rates between the TaTME
and LaTME groups. As for postoperative pelvic abscess formation
overall 5.8% of the patients after TaTME, 8.0% after RoTME and 6.9%
after LaTME had anastomotic leakage.
Urine retention and complications: Kang et al. [48] defined voiding difficulty as urinary retention or urinary incontinence
requiring urological medication or reinsertion of a Foley catheter.
Tuech et al. [46] reported five patients (8.9%) with postoperative
urinary retention, all treated by temporary urethral catheterization.
After three months, all patients reported normal urinary function.
Kang et al. [49] reported two urinary retentions during TaTME and
one reported three cases of urinary retention after removing the
Foley catheter after LaTME and none after TaTME [47]. Five cases
of neurogenic urinary retention were reported after LaTME in one
study [38]. In summary, 4.7% after TaTME, 4.9% after RoTME and
2.5% after LaTME had urinary retention or complications.
Bladder and urethral injury: Penna et al. [23] reported five cases
of urethral injuries (0.7%) and two bladder injuries (0.3%) during
perineal dissection after TaTME while Kang et al. [49] reported one
urethral injury as a result of dissecting too anteriorly into an enlarged
prostate. Rasulov et al. [43] reported one bladder injury during
TaTME, which was closed laparoscopically and the patient was
discharged with a urinary catheter, removed one week later. Park et
al. [34] reported one case of bladder injury because of tumor adhesion
to the bladder as well responsible for conversion from RoTME to
LaTME. One study reported five genitourinary injuries after RoTME
as just one out of two RoTME studies mentioning ureter or bladder
injuries [16]. Gong et al. [13] reported one conversation from LaTME
to open surgery due to ureteric injury. Overall, five bladder injuries
and seven urethral injuries (1.5%) among 806 patients after TaTME
were reported. 1.1% of the patients undergoing RoTME and 1.1%
of the patients undergoing LaTME had intraoperative urethral or
bladder injuries.
Urinary tract infection: Chen et al. [35] reported as only TaTMEstudy
one case among 50 patients and Levic et al. [17] as the only
RoTME-study none cases of urinary infection among 52 patients.
Baik et al. [28] reported two cases of grade three and one case of
grade two urinary tract infections after LaTME. Concerning LaTME,
2.3% among 2814 patients suffered from postoperative urinary tract
infection.
Wound infection: Several studies emphasized that the TaTME
procedure frequently allows specimen exteriorization without an
abdominal incision, eliminating abdominal site morbidity as wound
infection and hernia during specimen extraction [37,42,50,51].
Yamamoto et al. [38] reported that wound infection was diagnosed
within 30 days of the operation according to the criteria of the
Centers for Disease Control and Prevention. 1.6% of the patients
after TaTME, 1.9% after RoTME and 5.1% after LaTME suffered from
wound infection.
Bowel injury: Perdawood reported none bowel injury after
TaTME [5]. Two studies reported eight cases (1.1%) after RoTME,
besides accentuated one patient that required conversation from
RoTME to LaTME because of bowel perforation [12,16]. Three
studies reported 0.8% of patients among 1002 patients after LaTME
[5,9,12].
Intraabdominal abscess: Fifteen cases of pelvic or abdominal
abscess without evidence of anastomotic leakage were reported by
Penna et al. [23], listed under intraabdominal abscess in our tables.
Van der Pas [9] reported just abscess, listed as intraabdominal.
Saklani et al. [31] categorized intraabdominal abscess as a severe
complication together with anastomotic leakage. Two studies
reported no significant difference between RoTME and LaTME
[32,34]. Overall 2.3% of patients after TaTME, 1.4% after RoTME and
4.3% after LaTME had intra-abdominal abscesses.
Pelvic abscess: Chen et al. [35] reported three cases of pelvic
abscess (8%) in the TaTME group and four cases (6%) in the LaTME
group, all treated with antibiotics. Burke et al. [42] reported four cases
among 50 patients and two readmissions because of pelvic collection
after TaTME. All leaks and pelvic collections were managed
conservatively with antibiotics and prolonged drainage by Leong et al.
[52]. Baik et al. [28] graded it as a serious condition since one patient
was hospitalized for 167 days because of pelvic abscess. Kang et al.
[48] reported no significant difference neither RoTME nor LaTME
groups regarding pelvic abscess. Overall, 9.2% of the patients after
TaTME, 1.9% after RoTME and 3.1% after LaTME had pelvic abscess.
30-days mortality: TaTME: Four studies concluded no
postoperative mortality and two studies reported no surgeryassociated
deaths [36,42,43,50,51]. Penna et al. [23] reported 0.5% 30-days mortality and concluded to be similar according to previous
rectal surgery trials. Muratore et al. [53] reported a patient that
died of myocardial infarction three days after surgery. Three studies
did not mention mortality while Serra-Aracil reported no 30-days
mortality but a six percent median score on the CROPOSSUM scale
[5,35,37,45]. Overall, 2.1% patients died during the first 30 days after
surgery.
RoTME: Two studies reported none cases of postoperative death
without mentioning the follow-up period, while ten studies reported
no cases in a follow-up period between 30 days and 3 years [34,44].
Overall, one patient among 1414 (0.1%) died within the first 30 days.
LaTME: Van der Pas reported eight cases of death within 28
days [9]. Penninckx et al. [26] reported 11 cases among 764 patients
(1.44%) while Yamamoto et al. [38] reported the lowest mortality;
none cases among 490. Overall 0.8% of the patients died during the
first month after surgery.
Table 1
Table 2
Discussion
Complications and mortality due to surgery are of major importance and widespread adoption of new surgical methods depends among other things on complication rates and risk of postoperative death. Consequently, the studies management of data is important. One source of potential reporting bias is the retrospective nature of twelve studies in this review. Here the choice of outcomes reported can be influenced by the results, potentially making published results misleading [54]. Performance bias due to the experience among surgeons is another limiting factor as well as the number of surgeons performing the procedures vary from one to 65 different surgeons [13,29]. Several studies reported complications using the Clavien-Dindo classification, which impeded the use of data in relation to our study design [12,23,24,29,32,33,43,49,55]. Different diagnostic criteria and treatment options for the anastomotic leakage have been used throughout the studies [9,10,31,33,46]. However, more cases of leakage in the RoTME group compared to the TaTME and LaTME were reported. The anastomosis seemed to perform more efficiently after TaTME with lower number of leaks. The seriousness of the cases was however unclear. Unfortunately, only readmission or not after anastomotic leakage have been reported, while the length of hospitalization and death has not [5,23,30,42]. The LaTME-group advanced with well experienced surgeons because of the longer use of laparoscopic approach, though no superior data found as compared with TaTME [56]. Opposite, the LaTME performed better in relation to urine retention than RoTME and TaTME. Urine retention is a complication due to the operative procedure with iatrogenic injuries on the autonomic nerve in the narrow pelvic area [49], as well as the epidural analgesia which often is used during abdominal surgery [57]. Nevertheless, patients mostly recovered and gained previously bladder function during weeks after surgery and hospitalization, and it was further more deemed to constitute a mild complication [31,46]. Regarding wound infection, LaTME constituted a higher risk of post-operative wound infection, while TaTME possibly advantage from the fact that the specimen extraction were done transanal reducing intraabdominal wound related complications [37,42,50,51]. An almost equal percentage of urethral injuries among the three approaches were noted, as well as no higher percentage of injuries on nearby organs like bowel and bladder. Nevertheless, previous studies reported the TaTME procedure to cause more cases of urethral injuries [58]. Like injuries on organs, pelvic and intraabdominal abscess is a severe complication, which can lead to reoperation [23,37,42]. The reported cases in the TaTME varied from 2.4% to 20% what clearly underline the need for further research including big randomized trials [23,35,59]. As well the indication for blood transfusion varied among the studies, well-illustrated by Liang et al. [8] who transfused four patients out of 169 while Schiphorst et al. [30] transfused ten out of 86. The need for blood transfusion generally followed the bleedings and blood loss, but uniformed international evidence-based guidelines according to indication for blood transfusion might reduce bias. More bleeding and need for blood transfusion were reported among TaTME and presents a potential serious condition, with a need for acute conversion to open surgery [9,12,37]. We calculated an average EBL for each one of the approaches, which possible distorted the accuracy, though gave us an opportunity to make a comparison. The average EBL was higher among RoTME than TaTME and LaTME. Baik et al. [14] pointed, that prompt open conversion is impossible during RoTME because removing the robotic system is a time-consuming procedure, which is sometimes necessary for immediate control of serious bleeding. A fact that possible influences a higher blood loss during especially the most difficult RoTME operations ending up with conversion. The TaTME had a higher 30-days mortality rate than RoTME and LaTME, with poor evidence concerning cause of death. A recently published TaTME-study concluded no higher mortality according to previous rectal surgery trials [23]. However, research with longer follow-up period is needed since TaTME is a new technique meaning that surgeons improved in the studies and possibly reduced the complication rate in step with gaining higher level of experience [5,23,36,49].
Conclusion
We exposed an extract of complications in minimal invasive surgery for rectal cancer. The TaTME, as the newest procedure, had equal percentage of urethral injuries compared to LaTME and RoTME besides a low rate of anastomotic leakage. On the other hand, RoTME presented a lower percentage of bleeding and 30-days mortality in compare to especially TaTME. Further research is required, ideally randomized studies, to compare the three procedures regarding rates of serious intra and postoperative complications besides comparing their pathological results. Also, focus on which procedure that best treats a specific tumor stage, location and type of patient could possibly reduce complications and postoperative mortality in the future.
References
- NORDCAN. Cancer Statistics: Key Figures and Figures Denmark - rectal and anus. 2014.
- Heald RJ, Moran BJ, Ryall RD, Sexton R, MacFarlane JK. Rectal cancer: the Basingstoke experience of total mesorectal excision, 1978-1997. Arch Surg. 1998;133(8):894-9.
- Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery-the clue to pelvic recurrence? Br J Surg. 1982;69(10):613-6.
- Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet. 1986;1(8496):1479-82.
- Perdawood SK, Al Khefagie GA. Transanal vs. laparoscopic total mesorectal excision for rectal cancer: initial experience from Denmark. Colorectal Dis. 2016;18(1):51-8.
- Jayne DG, Guillou PJ, Thorpe H, Quirke P, Copeland J, Smith AM, et al. Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3-year results of the UK MRC CLASICC Trial Group. J Clin Oncol. 2007;25(21):3061-8.
- Hu JJ, Liang JW, Wang Z, Zhang XM, Zhou HT, Hou HR, et al. Short-term outcomes of laparoscopically assisted surgery for rectal cancer following neoadjuvant chemoradiotherapy: a single-center experience. J Surg Res. 2014;187(2):438-44.
- Liang X, Hou S, Liu H, Li Y, Jiang B, Bai W, et al. Effectiveness and safety of laparoscopic resection versus open surgery in patients with rectal cancer: a randomized, controlled trial from China. J Laparoendosc Adv Surg Tech A. 2011;21(5):381-5.
- van der Pas MH, Haglind E, Cuesta MA, Fürst A, Lacy AM, Hop WC, et al. Laparoscopic versus open surgery for rectal cancer (COLOR II): short-term outcomes of a randomised, phase 3 trial. Lancet Oncol. 2013;14(3):210-8.
- Lujan J, Valero G, Biondo S, Espin E, Parrilla P, Ortiz H. Laparoscopic versus open surgery for rectal cancer: results of a prospective multicentre analysis of 4,970 patients. Surg Endosc. 2013;27(1):295-302.
- Clinical Outcomes of Surgical Therapy Study Group; Nelson H, Sargent DJ, Wieand HS, Fleshman J, Anvari M, et al. A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med. 2004;350(20):2050-9.
- Cho MS, Baek SJ, Hur H, Min BS, Baik SH, Lee KY, et al. Short and long-term outcomes of robotic versus laparoscopic total mesorectal excision for rectal cancer: a case-matched retrospective study. Medicine. 2015;94(11):e522.
- Gong J, Shi DB, Li XX, Cai SJ, Guan ZQ, Xu Y. Short-term outcomes of laparoscopic total mesorectal excision compared to open surgery. World J Gastroenterol. 2012;18(48):7308-13.
- Baik SH, Kwon HY, Kim JS, Hur H, Sohn SK, Cho CH, et al. Robotic versus laparoscopic low anterior resection of rectal cancer: short-term outcome of a prospective comparative study. Ann Surg Oncol. 2009;16(6):1480-7.
- D'Annibale A, Pernazza G, Monsellato I, Pende V, Lucandri G, Mazzocchi P, et al. Total mesorectal excision: a comparison of oncological and functional outcomes between robotic and laparoscopic surgery for rectal cancer. Surg Endosc. 2013;27(6):1887-95.
- Hellan M, Ouellette J, Lagares-Garcia JA, Rauh SM, Kennedy HL, Nicholson JD, et al. Robotic Rectal Cancer Resection: A Retrospective Multicenter Analysis. Ann Surg Oncol. 2015;22(7):2151-8.
- Levic K, Donatsky AM, Bulut O, Rosenberg J. A Comparative Study of Single-Port Laparoscopic Surgery Versus Robotic-Assisted Laparoscopic Surgery for Rectal Cancer. Surg Innov. 2015;22(4):368-75.
- Baek JH, Pastor C, Pigazzi A. Robotic and laparoscopic total mesorectal excision for rectal cancer: a case-matched study. Surg Endosc. 2011;25(2):521-5.
- Bianchi PP, Ceriani C, Locatelli A, Spinoglio G, Zampino MG, Sonzogni A, et al. Robotic versus laparoscopic total mesorectal excision for rectal cancer: a comparative analysis of oncological safety and short-term outcomes. Surg Endosc. 2010;24(11):2888-94.
- Schootman M, Hendren S, Ratnapradipa K, Stringer L, Davidson NO. Adoption of Robotic Technology for Treating Colorectal Cancer. Dis Colon Rectum. 2016;59(11):1011-8.
- Health Quality Ontario. Robotic-assisted minimally invasive surgery for gynecologic and urologic oncology: an evidence-based analysis. Ontario health technology assessment series. 2010;10(27):1-118.
- Collinson FJ, Jayne DG, Pigazzi A, Tsang C, Barrie JM, Edlin R, et al. An international, multicentre, prospective, randomised, controlled, unblinded, parallel-group trial of robotic-assisted versus standard laparoscopic surgery for the curative treatment of rectal cancer. International journal of colorectal disease. 2012;27(2):233-41.
- Penna M, Hompes R, Arnold S, Wynn G, Austin R, Warusavitarne J, et al. Transanal Total Mesorectal Excision: International Registry Results of the First 720 Cases. Ann Surg. 2017;266(1):111-7.
- Lacy AM, Tasende MM, Delgado S, Fernandez-Hevia M, Jimenez M, De Lacy B, et al. Transanal Total Mesorectal Excision for Rectal Cancer: Outcomes after 140 Patients. J Am Coll Surg. 2015;221(2):415-23.
- Mother D, Liberati A, Tetzlaff J, Altman DG. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. 2009;151(4):264-9.
- Penninckx F, Kartheuser A, Van de Stadt J, Pattyn P, Mansvelt B, Bertrand C, et al. Outcome following laparoscopic and open total mesorectal excision for rectal cancer. Br J Surg. 2013;100(10):1368-75.
- Wu WX, Sun YM, Hua YB, Shen LZ. Laparoscopic versus conventional open resection of rectal carcinoma: A clinical comparative study. World J Gastroenterol. 2004;10(8):1167-70.
- Baik SH, Gincherman M, Mutch MG, Birnbaum EH, Fleshman JW. Laparoscopic vs open resection for patients with rectal cancer: comparison of perioperative outcomes and long-term survival. Dis Colon Rectum. 2011;54(1):6-14.
- Fleshman J, Branda M, Sargent DJ, Boller AM, George V, Abbas M, et al. Effect of Laparoscopic-Assisted Resection vs Open Resection of Stage II or III Rectal Cancer on Pathologic Outcomes: The ACOSOG Z6051 Randomized Clinical Trial. JAMA. 2015;314(13):1346-55.
- Schiphorst AH, Doeksen A, Hamaker ME, Zimmerman DD, Pronk A. Short-term follow-up after laparoscopic vs. conventional total mesorectal excision for low rectal cancer in a large teaching hospital. Int J Colorectal Dis. 2014;29(1):117-25.
- Saklani AP, Lim DR, Hur H, Min BS, Baik SH, Lee KY, et al. Robotic versus laparoscopic surgery for mid-low rectal cancer after neoadjuvant chemoradiation therapy: comparison of oncologic outcomes. Int J Colorectal Dis. 2013;28(12):1689-98.
- Yoo BE, Cho JS, Shin JW, Lee DW, Kwak JM, Kim J, et al. Robotic versus laparoscopic intersphincteric resection for low rectal cancer: comparison of the operative, oncological, and functional outcomes. Ann Surg Oncol. 2015;22(4):1219-25.
- Ielpo B, Caruso R, Quijano Y, Duran H, Diaz E, Fabra I, et al. Robotic versus laparoscopic rectal resection: is there any real difference? A comparative single center study. Int J Med Robot. 2014;10(3):300-5.
- Park EJ, Cho MS, Baek SJ, Hur H, Min BS, Baik SH, et al. Long-term oncologic outcomes of robotic low anterior resection for rectal cancer: a comparative study with laparoscopic surgery. Ann Surg. 2015;261(1):129-37.
- Chen CC, Lai YL, Jiang JK, Chu CH, Huang IP, Chen WS, et al. Transanal Total Mesorectal Excision Versus Laparoscopic Surgery for Rectal Cancer Receiving Neoadjuvant Chemoradiation: A Matched Case-Control Study. Ann Surg Oncol. 2016;23(4):1169-76.
- Marks JH, Montenegro GA, Salem JF, Shields MV, Marks GJ. Transanal TATA/TME: a case-matched study of taTME versus laparoscopic TME surgery for rectal cancer. Techniques in coloproctology. 2016;20(7):467-73.
- Fernández-Hevia M, Delgado S, Castells A, Tasende M, Momblan D, Díaz del Gobbo G, et al. Transanal total mesorectal excision in rectal cancer: short-term outcomes in comparison with laparoscopic surgery. Ann Surg. 2015;261(2):221-7.
- Yamamoto S, Ito M, Okuda J, Fujii S, Yamaguchi S, Yoshimura K, et al. Laparoscopic surgery for stage 0/I rectal carcinoma: short-term outcomes of a single-arm phase II trial. Ann Surg. 2013;258(2):283-8.
- Fujii S, Yamamoto S, Ito M, Yamaguchi S, Sakamoto K, Kinugasa Y, et al. Short-term outcomes of laparoscopic intersphincteric resection from a phase II trial to evaluate laparoscopic surgery for stage 0/I rectal cancer: Japan Society of Laparoscopic Colorectal Surgery Lap RC. Surg Endosc. 2012;26(11):3067-76.
- Rouanet P, Mourregot A, Azar CC, Carrere S, Gutowski M, Quenet F, et al. Transanal endoscopic proctectomy: an innovative procedure for difficult resection of rectal tumors in men with narrow pelvis. Diseases of the colon and rectum. 2013;56(4):408-15.
- Araujo SE, da Silva eSousa AH, de Campos FG, Gama AH, Dumarco RB, de Paris Caravatto PP, et al. Conventional approach x laparoscopic abdominoperineal resection for rectal cancer treatment after neoadjuvant chemoradiation: results of a prospective randomized trial. Revista do Hospital das Clinicas. 2003;58(3):133-40.
- B Burke JP, Martin-Perez B, Khan A, Nassif G, de Beche-Adams T, Larach SW, et al. Transanal total mesorectal excision for rectal cancer: early outcomes in 50 consecutive patients. Colorectal Dis. 2016;18(6):570-7.
- Rasulov AO, Mamedli ZZ, Gordeyev SS, Kozlov NA, Dzhumabaev HE. Short-term outcomes after transanal and laparoscopic total mesorectal excision for rectal cancer. Techniques in coloproctology. 2016;20(4):227-34.
- Fernandez R, Anaya DA, Li LT, Orcutt ST, Balentine CJ, Awad SA, et al. Laparoscopic versus robotic rectal resection for rectal cancer in a veteran population. Am J Surg. 2013;206(4):509-17.
- Serra-Aracil X, Mora-Lopez L, Casalots A, Pericay C, Guerrero R, Navarro-Soto S. Hybrid NOTES: TEO for transanal total mesorectal excision: intracorporeal resection and anastomosis. Surg Endosc. 2016;30(1):346-54.
- Tuech JJ, Karoui M, Lelong B, De Chaisemartin C, Bridoux V, Manceau G, et al. A step toward NOTES total mesorectal excision for rectal cancer: endoscopic transanal proctectomy. Ann Surg. 2015;261(2):228-33.
- Chen Y, Guo R, Xie J, Liu Z, Shi P, Ming Q. Laparoscopy Combined With Transanal Endoscopic Microsurgery for Rectal Cancer: A Prospective, Single-blinded, Randomized Clinical Trial. Surgical laparoscopy, endoscopy & percutaneous techniques. 2015;25(5):399-402.
- Kang J, Min BS, Park YA, Hur H, Baik SH, Kim NK, et al. Risk factor analysis of postoperative complications after robotic rectal cancer surgery. World J Surg. 2011;35(11):2555-62.
- Kang L, Chen WH, Luo SL, Luo YX, Liu ZH, Huang MJ, et al. Transanal total mesorectal excision for rectal cancer: a preliminary report. Surg Endosc. 2016;30(6):2552-62.
- Wang Q, Wang C, Sun DH, Kharbuja P, Cao XY. Laparoscopic total mesorectal excision with natural orifice specimen extraction. World J Gastroenterol. 2013;19(5):750-4.
- de Lacy AM, Rattner DW, Adelsdorfer C, Tasende MM, Fernández M, Delgado S, et al. Transanal natural orifice transluminal endoscopic surgery (NOTES) rectal resection: "down-to-up" total mesorectal excision (TME)--short-term outcomes in the first 20 cases. Surg Endosc. 2013;27(9):3165-72.
- Leong QM, Son DN, Cho JS, Baek SJ, Kwak JM, Amar AH, et al. Robot-assisted intersphincteric resection for low rectal cancer: technique and short-term outcome for 29 consecutive patients. Surg Endosc. 2011;25(9):2987-92.
- Muratore A, Mellano A, Marsanic P, De Simone M. Transanal total mesorectal excision (taTME) for cancer located in the lower rectum: short- and mid-term results. Eur J Surg Oncol. 2015;41(4):478-83.
- Higgins JPT GSe. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration. 2011.
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240(2):205-13.
- Guillou PJ, Quirke P, Thorpe H, Walker J, Jayne DG, Smith AM, et al. Short-term endpoints of conventional versus laparoscopic-assisted surgery in patients with colorectal cancer (MRC CLASICC trial): multicentre, randomised controlled trial. The Lancet. 2005;365(9472):1718-26.
- Simmons SW, Cyna AM, Dennis AT, Hughes D, Taghizadeh N. Combined spinal-epidural versus epidural analgesia in labour. Cochrane Database Syst Rev. 2012;(3):CD003401.
- Bjorn MX, Perdawood SK. Transanal total mesorectal excision--a systematic review. Danish medical journal. 2015;62(7).
- Atallah S, Martin-Perez B, Albert M, deBeche-Adams T, Nassif G, Hunter L, et al. Transanal minimally invasive surgery for total mesorectal excision (TAMIS-TME): results and experience with the first 20 patients undergoing curative-intent rectal cancer surgery at a single institution. Techniques in coloproctology. 2014;18(5):473-80.