Research Article
Coronary Artery Bypass Grafting in a Bioresorbable Vascular Scaffold Implanted Porcine Coronary Artery
Benham JJ1*, Perkins L1, Sun B2, Gross C3 and Rapoza R1
1Abbott Vascular, 3200 Lakeside Drive, Santa Clara, USA
2Minneapolis Heart Institute, 800 E 28th St., Minneapolis, USA
3American Preclinical Services, 8945 Evergreen Blvd., Minneapolis, USA
*Corresponding author: James J. Benham, Abbott Vascular, 3200 Lakeside Drive, Santa Clara, USA
Published: 06 Dec, 2017
Cite this article as: Benham JJ, Perkins L, Sun B, Gross
C, Rapoza R. Coronary Artery Bypass
Grafting in a Bioresorbable Vascular
Scaffold Implanted Porcine Coronary
Artery. Clin Surg. 2017; 2: 1809.
Abstract
It is not well understood how Percutaneous Coronary Intervention (PCI) with permanent metallic stents complicates an impending Coronary Artery Bypass Grafting (CABG) procedure. However, previously implanted metallic stents often limit the graft landing zone options. Due to their transient properties, Bioresorbable Vascular Scaffolds (BVS) may address this issue and allow for the implanted site to serve as a suitable grafting location. The aim of this study was to acutely evaluate this hypothesis. The Left Anterior Descending (LAD) coronary arteries of three healthy Yucatan miniswine were implanted with 3.0 mm × 12 mm BVS. At one year post implant, a CABG procedure was performed within the BVS implanted segment. Procedural feedback was obtained from the cardiothoracic surgeon. Patency of the graft and artery was assessed with standard angiography and intravascular with Optical Coherence Tomography (OCT). The animals were sacrificed immediately post procedure and the BVS implanted segment with graft and the downstream myocardium were assessed histologically. The procedure was similar to performing an incision in a mildly-moderately diseased vessel but was not like a calcified vessel. Post-graft imaging revealed patent arteries with no evidence of thrombosis, embolization, or luminal obstruction due to the scaffold struts. Histology confirmed that the graft was within the BVS implanted region, confirmed the imaging results, and demonstrated there were no adverse effects from cutting through the scaffold.
Introduction
Despite advances in Percutaneous Coronary Intervention (PCI), Coronary Artery Bypass Grafting (CABG) still plays a significant role in the treatment of complex cardiovascular disease due to good long-term survival and low major adverse events rates [1-3]. Due to the severity and complexity of cardiovascular disease in patients requiring CABG, it is not uncommon for them to have a history of PCI. How those previous interventions may impact the CABG procedure is not completely understood. Therefore, the potential for complications due to pre-existing permanent metallic stents is an important consideration when deciding if a patient is a candidate for CABG. Patients that have undergone PCI prior to CABG and have developed significant lesions in other areas of the same artery may leave the surgeon with limited options for a graft landing zone. Polymeric Bioresorbable Vascular Scaffolds (BVS) may provide an attractive alternative to metallic stents with regards to this issue. The scaffold polymer is designed to maintain radial strength like a metallic device for at least six months, but through hydrolysis, the scaffold becomes more compliant and completely resorbs by approximately 3 years post implant [4]. This may allow for grafting into a previously scaffolded vessel segment, effectively overcoming the complications and limitations for CABG presented by metallic stents as early as 1-2 years post PCI. The aim of this study was to acutely evaluate the feasibility of performing CABG to a coronary artery segment implanted with a BVS at 1 year post implantation. The cardiothoracic surgeon provided feedback on the clinical relevance of the incision and graft attachment into a BVS implanted artery as well as clinical perspectives on the anastomosis creation, grafting procedure, and overall procedural outcomes. A concern about performing an incision into a BVS implanted segment is the potential for scaffold strut disruption or fragmentation that may lead to thrombosis or embolization. To evaluate the site for strut disruption and vessel patency, post procedural angiography and OCT imaging of the grafted BVS artery was performed. The imaging was also performed to confirm the graft was attached within the BVS implanted segment and to assess the procedural success. In addition, arterial histopathology was performed to further confirm the graft location within the BVS implanted segment and evaluate the site for thrombosis and scaffold disruption. Furthermore, myocardial histopathology was performed to evaluate the downstream myocardium for thrombo-embolization.
Figure 1
Figure 1
Incision and Grafting Procedural Images.
Top left: Incision into BVS implanted artery using a standard surgical blade
(black circle)
Top right: Completed arterial incision (black circle)
Bottom left: LITA graft (black arrow) being sutured to the BVS implanted graft
site (yellow arrow) with standard needle and 7-0 prolene suture material
Figure 2
Figure 2
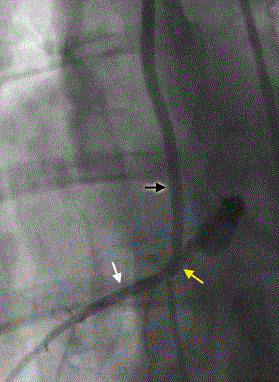
Fluoroscopy of the grafted BVS implanted segment. The post
procedural angiographic image shows a patent coronary artery (white arrow),
BVS implanted vessel segment (yellow arrow), and LITA graft (black arrow).
No filling defects were observed.
Figure 3
Figure 3
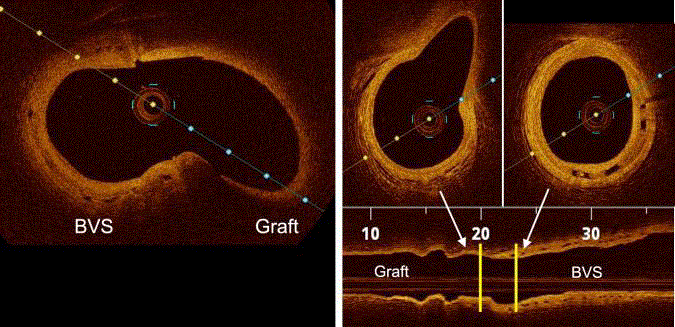
Post procedural OCT pullbacks. Left: Cross sectional OCT image
of the BVS and graft interface (yellow double arrow) obtained by pullback
through the coronary artery. The image demonstrates the procedural success
and absence of strut disruption or fragmentation. Right: Cross sectional and
longitudinal section of the BVS implanted artery and graft interface obtained
by pullback through the graft. Yellow lines represent an approximation of the
cross-section locations. The image demonstrates the continuity between the
coronary artery and graft.
Figure 4
Figure 4
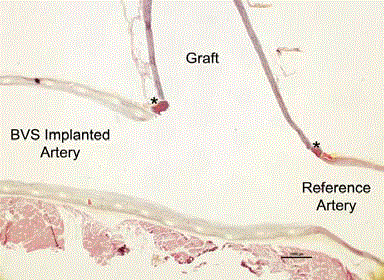
Photomicrograph of the longitudinal section of the coronary artery
and graft. Histological evaluation demonstrates the procedural success
with intact anastomosis sites (asterisks), benign response to the grafting
procedure, and patency of the lumens, confirming that grafting into the BVS
implanted artery does not disrupt the BVS struts and is free of thrombus.
Materials and Methods
One year prior to the grafting procedure, the Left Anterior Descending (LAD) coronary arteries of three normal Yucatan miniswine were implanted with 3.0 mm × 12 mm BVS (Abbott Vascular, Inc.) using standard, sterile implant procedures. At one year post implant, the animals were sedated and anesthetized. A sternotomy was performed and a standard off-pump grafting procedure using an Urchin heart positioner and Octopus tissue stabilizer (Medtronic, Inc.) was performed into the BVS-implanted segment of the LAD by a cardiothoracic surgeon. The Left Internal Thoracic Artery (LITA) was used as the bypass graft. The incision into the BVS implanted artery was performed using a standard fine blade. The LITA was prepared as a graft and sutured onto the targeted LAD site using a standard needle and 7-0 prolene (Figure 1). Procedural feedback regarding the ease of the procedure and clinical application was collected from the surgeon. Angiography and OCT were performed post-procedure to assess patency and overall lumen condition and to confirm a suitable anastomosis without leakage. OCT pullbacks were performed through the LAD and through the graft. The animals were sacrificed and the arteries and downstream myocardium were evaluated grossly and histologically to assess local and downstream effects from grafting in the BVS implanted region.
Results and Discussion
Procedurally, the incision into the BVS- implanted LAD was
performed without difficulty, felt fibrotic with mild grittiness, but
was easier than entering a hard, calcified lesion per the cardiothoracic
surgeon. The implanted tissue was moderately thickened compared
to naïve arterial tissue but was otherwise unremarkable upon gross
evaluation. The LITA graft was sutured onto the targeted LAD
site without difficulty or the need for specialized instruments. The
procedure was uncomplicated and clinically like grafting into a mildly
to moderately diseased coronary artery.
Post-grafting angiography revealed normal blood flow, a patent
and well-sealed anastomosis, and a patent internal thoracic artery graft
and LAD lumen (Figure 2). OCT confirmed the graft placement and
revealed patent lumens free from thrombosis or luminal obstruction
by BVS struts (Figure 3). Additionally, the gross assessment at
necropsy and histological evaluation confirmed that the graft was
successfully placed in the BVS region and confirmed the patency
of the graft, anastomosis, and BVS implanted artery (Figure 4).
Histological evaluation of the downstream myocardium showed no evidence of thrombo-embolization related to the grafting procedure.
While grafting into a non-diseased vessel is ideal, it is common for
a graft site to have mild to moderate disease. Therefore, due to the
similarity to the clinical condition, the BVS implanted porcine artery
provides a clinically relevant model and demonstrates that a BVS
implanted artery can serve as a suitable implant site. Furthermore,
the post procedural imaging shows that cutting through the BVS
does not cause strut disruption or fragmentation, which could lead
to thrombosis or embolization. This is likely due to the combination
of the unique mechanical properties of BVS during resorption and
the stability provided by the neointima. Histopathology confirms
this observation and demonstrates the acute safety to the grafting
procedure in a BVS-implanted segment.
Conclusion
In a porcine coronary artery model, CABG was successfully performed in BVS implanted porcine coronary arteries one year postimplant. The grafting procedure could be performed in accordance with standard off-pump procedures without complication and using standard equipment. Angiography, OCT, and histology revealed a successful graft and patency of the graft, anastomosis, and LAD without evidence of thrombosis in the treated artery or thromboembolization in the downstream myocardium.
References
- Sipahi I, Akay MH, Dagdelen S, Blitz A, Alhan C. Coronary Artery Bypass Grafting vs Percutaneous Coronary Intervention and Long-term Mortality and Morbidity in Multivessel Disease: Meta-analysis of Randomized Clinical Trials of the Arterial Grafting and Stenting Era. JAMA Intern Med. 2014;174(2):223-30.
- Papadopoulos K, Lekakis I, Nicolaides E. Outcomes of coronary artery bypass grafting versus percutaneous coronary intervention with second-generation drug-eluting stents for patients with multivessel and unprotected left main coronary artery disease. SAGE Open Medicine. 2017;5:1-5.
- Bangalore S, Guo Y, Samadashvili Z, Blecker S, Xu J, Hannan EL. Everolimus-Eluting Stents or Bypass Surgery for Multivessel Coronary Disease. N Engl J Med. 2015;372:1213-22.
- Onuma Y, Serruys PW. Bioresorbable scaffold: the advent of a new era in percutaneous coronary and peripheral revascularization? Circulation. 2011;123(7):779-97.