Editorial
Retinal Examınatıon for Plaque Burden
Umur Kayabasi*
Department of Ophthalmology, World Eye Hospital, Istanbul, Turkey
*Corresponding author: Umur Kayabasi, Department of Ophthalmology, World Eye Hospital, Istanbul, Turkey
Published: 16 Nov 2017
Cite this article as: Kayabasi U. Retinal Examınatıon for
Plaque Burden. Clin Surg. 2017; 2:
1736.
Editorial
A recent article authored by an experienced team of authors suggests that retinal amyloid burden
is correlated with brain amyloid burden in Alzheimer’s Disease (AD). Moreover, it is stated that
retinal amyloid plaques appear before they start to invade the hippocampus [1]. Screening Amyloid
Beta (AB) in the retina is definitely a major breakthrough in Alzheimer’s Disease (AD). Comparing
retinal amyloid burden with brain amyloid is even going one step further in the trials for the early
diagnosing of AD. Imaging the retina is easy, non-invasive, cheap and does not involve radiation.
But, there are two important questions that come to mind when considering the recent
developments in AD studies:
1. Mayo Clinic study of thousands of brains revealed Tau as driver of AD [2]. So, do we need
to show Tau protein along with AB in the retina to be able to make correct assumptions about the
disease process?
2. Postmortem studies have demonstrated that people with AD have far more AB plaques
in their brains than healthy people. But,roughly 30% of people without any signs of dementia have
brains “chock-full” of AB at autopsy [3]. So, the presence of AB may not be enough to prove that any
patient will develop the disease in the future.
Increased deposition of AB isoforms have been described on photoreceptor outer segments and
along the RPE–Bruch's membrane interface in the aging human and mouse retina [4]. Another
study using immunostaining revealed AB on photoreceptor outer segments all through the retina
in humans as an ageing process [5]. This deposition process may very well be only a sign of ageing
and not related to AD. Analyses of drusen components have shown deposition of AB within vesicles
in eyes of Age Related Macular Degeneration (AMD) patients. We also managed to show AB in
drusen of AMD patients [6]. A better approach may be to demonstrate and quantify both proteins
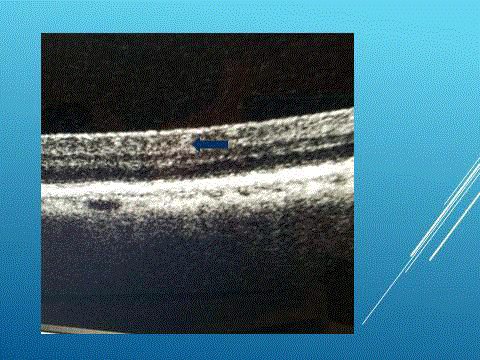
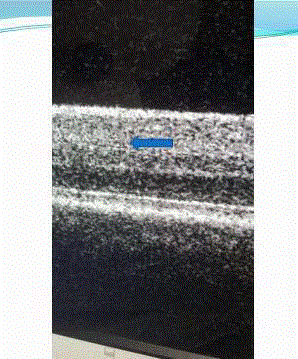
in the retina [7,8]. We have been able to image Tau aggregates in the retina of live patients (Figures
1 and 2).
Tau deposits have been found in the temporal lobe early in the disease which proves that Tau
PET imaging is also necessary to be able to correlate the abnormal protein burden with brain plaques.
Conclusion
Recent studies confirmed that combination of Tau and AB aggregates caused the disease. Another proof of this theory is the unsuccessful results of AB aimed therapies in AD. We believe more collaborative work is necessary for the definite answers to the remaining questions.
Figure 1
Figure 2
References
- Koronyo Y, Biggs D, Barron E, Boyer DS, Pearlman JA, Au WJ, et al. Retinal amyloid pathology and proof-of-concept imaging trial in Alzheimer's disease. JCI Insight. 2017;2(16).
- Murray M. Study of Thousands of Brains Reveals Tau as Driver of Alzheimer’s Disease. Mayo Clinic Alumni Association. 2015.
- Jones DT, Knopman DS, Gunter JL, Graff-Radford J, Vemuri P, Boeve BF, et al. Cascading network failure across the Alzheimer's disease spectrum. Brain. 2016;139(Pt 2):547-62.
- Shah TM, Gupta SM, Chatterjee P, Campbell M, Martins RN. Beta-amyloid sequelae in the eye: a critical review on its diagnostic significance and clinical relevance in Alzheimer's disease. Mol Psychiatry. 2017;22(3):353-63.
- Hoh Kam J, Lenassi E, Jeffery G. Viewing ageing eyes: diverse sites of amyloid Beta accumulation in the ageing mouse retina and the up-regulation of macrophages. PLoS One. 2010;5(10).
- Kayabasi U, Sahbaz I. Beta Amyloid in Age- Related Macular Degeneration Lesions in a Patient with Alzheimer’s Disease. EC Ophthalmology. 2017;7(1):18-21.
- Kayabasi U. Tau in the Retina. EC Neurology. 2016;3(5):493-9.
- Kayabasi U. Retinal Examination by OCT to Reveal Neurodegeneration in the Brain. EC Ophthalmology. 2017;6(5):139-140.