Short Communication
Partial Nephrectomy for Clinical T2 Renal Tumors: Current Evidence in Perspective
Mathew Yamoah Kyei* and James Edward Mensah
Department of Surgery and Urology, School of Medicine and Dentistry, College of Health Sciences, University of
Ghana, Ghana
*Corresponding author: Mathew Yamoah Kyei, Department of Surgery and Urology, School of Medicine and Dentistry, College of Health Sciences, University of Ghana, P. O. Box 4236, Accra, Ghana
Published: 02 Nov, 2017
Cite this article as: Kyei MY, Mensah JE. Partial
Nephrectomy for Clinical T2 Renal
Tumors: Current Evidence in
Perspective. Clin Surg. 2017; 2: 1709.
Abstract
Renal tumor is one of the most important urological malignancies with a rising worldwide incidence. There is advocacy for nephron sparing surgery (Partial nephrectomy (PN)) in the management of renal tumors because of studies that have demonstrated impaired renal function after radical nephrectomy with an associated increased risk of overall mortality. Renal tumors with size less than 4 cm, clinical stage T1a (cT1a), had been the focus for PN. However currently, clinical stage T2 (cT2) renal tumors which are renal tumors larger than 7 cm but confined to the kidneys are being managed with partial nephrectomy. This expanding use has been attributed to advances in tumor biology, radiological imaging and surgical technology. We explore the current evidence in support of this emerging practice.
Background
Renal tumor is one of the most important urological malignancies accounting for 2.4% of all
adult malignancies. Its incidence is noted to be rising with an early stage at detection due to current
wide spread use of cross sectional imaging in the evaluation of patients [1,2].
Currently, the diagnosis and staging of renal tumors involves the use of cross sectional imaging
such as ultrasound, CT scan and magnetic resonance imaging. These have been found to be
mostly accurate though some benign lesions have been inaccurately diagnosed as malignant [3].
Percutaneous renal biopsies have been found to be safe and able to different malignant lesions from
benign lesions and determine the potential for tumor growth and metastasis by determining the cell
subtype and the fuhrman grade [4-6].
However, it must be noted that the smaller the renal mass the more likely it would be benign [7]
and hence clinical stage T2 (cT2) renal tumors which are larger might have a higher percentage of
them being malignant as compared to the finding by some investigators that between 8% - 27% of
resected renal tumors tended to be benign [6,8].
The diagnosis of renal tumors at an early stage has contributed to the increasing call for nephron
sparing surgery for the management of these tumors. Though this observation is true of developed
countries, in developing countries renal tumors tend to be seen in a more advanced stage limiting
the use of nephron sparing procedures, thus radical nephrectomy (RN) tends to be the norm [9].
The advocacy for nephron sparing surgery (Partial nephrectomy (PN)) in the management of renal
tumors is because of studies that have demonstrated impaired renal function after RN with an
associated increased risk of overall mortality and a greater risk of cardiovascular events that arise
from loss of renal function [10].
The need to remove or ablate renal tumors stem from the fact that renal cell carcinoma, the
predominant histological type (70%), is not effectively treated with chemotherapy and radiation
therapy. The use of immunotherapy using cytokines like interferon alpha and interleukin 2 has
demonstrated variable results with IFN-α providing a response rate of 6% to 15% and a survival
benefit of 3–5 months and Interleukin-2 (IL-2) with documented response rates from 7% to 27%
[11]. Other agents such as vascular endothelial growth factor-targeting (VEGF) antiangiogenic
agents, mammalian target of rapamycin inhibitors, temsirolimus, and sorafenib have largely shown
no statistically significant improvement in overall survival which has led to the introduction of
Nivolumab, a programmed death-1 (PD-1) monoclonal antibody, which is under study [12].
Expanding Indications for partial Nephrectomy to include clinical T2 renal tumors
Partial nephrectomy (PN) for renal tumors has been in practice for some period. Renal tumors with size less than 4 cm, clinical stage T1a (cT1a), had been the focus for PN. Other considerations for a safe PN that ensures complete
tumor removal included a peripherally located tumor, a unilaterally
functioning kidney and presence of bilateral renal tumors [13]. This
procedure is increasingly done using minimally invasive methods
such as laparoscopic [14] and robotic [15]. The oncology outcomes
have been similar comparing these minimally invasive procedures
and the open partial nephrectomy but with shorter hospital stay and
less transfusion in the former [14,15].
Open partial nephrectomy however continues to be performed
for larger tumors in situations where there is decreased performance
status and for tumors in solitary kidney seven in centers with
minimally invasive setups [14]. Partial nephrectomy has been
expanded to include clinical stage T1b (cT1b) and currently clinical
stage T2 (cT2) renal tumors in the existence of good contralateral
kidneys. cT2 renal tumors are renal tumors with size greater than 7
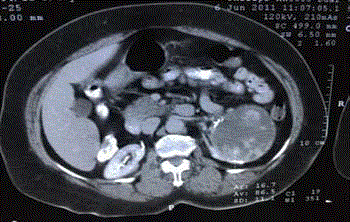
cm but confined to the kidney (Figure 1).
This expanding use has been attributed to advances in tumor
biology, radiological imaging and surgical technology [13].
Though initial results were conflicting as to the superiority
of PN over RN with regards to overall survival, Kopp et al. [16]
in a study found no significant difference in 5 year cancer specific
survival, progression free survival and overall survival between PN
and RN for cT2 renal tumors using the R.E.N.A.L nephrometric
score to achieve satisfactory matching so as to eliminate inherent
biases in tumor characteristics that influence the choice of either PN
or RN [16]. In a meta-analysis by Amir et al. [17], they identified
four studies on cT2 renal tumors that compared PN (n=212) to RN
(n=1792). In these patients, they found that the estimated blood loss
was higher for PN (p< 0.001), as was the likelihood of complications
(RR 2.0; p< 0.001). However, the recurrence rate (RR 0.61; p=0.004)
and cancer-specific mortality (RR 0.65; p=0.03) were lower for PN
with the better likelihood of renal function preservation [17]. They
concluded that despite these advantages, there was the need for good
patient selection to achieve the intended benefits for choosing PN
for cT2 renal tumors [17]. Similar conclusion was arrived at after the
review of oncological and functional outcomes for PN in the setting
ofcT2 tumors by Nahal et al. [18]. They also affirmed the advantages
of PN over RN for cT2 tumors indicating that PN has emerged as an
acceptable alternative for surgical management of cT2 renal tumors
over the last decade [18].
Points of concern
The need for cross sectional imaging with 3D reconstruction as
part of the pre-operative planning is important in these large tumors
especially in assessing the nearness to/ involvement of the renal hilum
and in the determination of renal parenchyma that is likely to be left
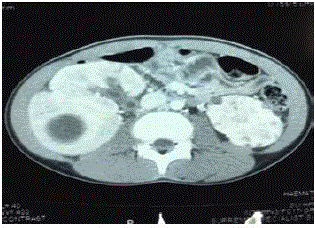
behind after the procedure (Figure 2).
Warm ischaemia have a negative impact on renal function.
The median warm ischaemic times have been noted to be longer in
larger renal tumors undergoing PN compared to smaller tumors
[15]. Therefore, efforts at reducing the effect of warm ischaemia
need special consideration in these larger tumors. A study of 360
solitary kidneys that had PN showed that each additional minute of
warm ischemia correlated with a 6% increased incidence of de novo
severe CKD and that suggested a cause/effect relationship [19]. In
current practice, a warm ischaemic time of 30 min is considered safe.
However, for procedures anticipated to go beyond this time, various
methods have been employed to reduce the ischaemic time, with
hypothermia being one of the foremost modalities used to preserve
renal function. The clinical effectiveness of pharmacological agents
such as intravenous mannitol infusion, dopamine, fenodolpam and
use of antioxidants such as allupurinol, vitamin C and vitamin E in
preventing ischaemic renal damage during PN have been challenged
[20]. Thus nephron sparing surgeries for cT2 tumors requires one to
be technically experienced in order to perform the procedure in an
acceptable time period to avoid a prolonged warm ischaemic time
or if need be institute some of the preventive methods early to save
renal function.
Tumor recurrence either local or metastatic is of great concern
when PN is performed for cT2 renal tumors. Percutaneous renal
biopsies on these large renal tumors prior to intervention could
identify benign lesions or less aggressive lesions that will assist
in the clinical decision as regards the extent of resection at PN in
cT2 renal tumors. There is however the need for a lifelong follow
up or surveillance to diagnosis or detect local recurrence early for
intervention.
There is no consensus on the follow up plan of patients after PN
for small renal masses and less so forcT2 renal tumors. Kopp et al. [16]
in their study used physical examination, chest X-ray and abdominal
CT/MRI every 6 months for 3 years, then yearly afterwards after PN
for cT2 renal tumors with long term follow-up continuing indefinitely
[16]. This may be modified if the histology report comes as a benign
lesion.
Further studies on PN for cT2 renal tumors will lead to the
procedure being performed more safely supported by robust and
standardized predictor of success and recurrence. Increasing
experience, will also allow more of these large tumors to be managed
by minimally invasive techniques.
Figure 1
Figure 2
Figure 2
CT scan showing a cT2 right renal tumor, initial plan of performing
open partial nephrectomy was abandoned as the hilum was found intraoperatively
to be involved by tumor.
Conclusion
Partial nephrectomy for Renal tumors have been demonstrated to be safe in well selected patients with cT2 renal tumors offering long term tumor control with the advantage of preserving renal function. These tumors have heterogeneous characteristics and so require careful consideration of the patient and tumor characteristics as well as the surgeons experience in its pursuit. The call for prospective randomized studies is in order as it will firmly place this approach as a viable and probably the preferred option in the management of patients with cT2 renal tumors.
References
- Motzer RJ, Bander NH, Nanus DM.Renal-cell carcinoma. N Engl J Med. 1996;335:865–75.
- Chow WH, Devesa SS, WarrenJL,Fraumeni JF Jnr. Rising incidence of renal cell carcinoma in the UnitedStates. JAMA. 1999;281:1628–31.
- Bosniak MA. The small (less than orequal to 3.0 cm) renal parenchymal tumor: detection, diagnosis andcontroversies. Radiology. 1991;179:307–17.
- Maturen KE, Nghiem HV, Caoili EM,Higgins EG, Wolf JS Jr, Wood DP Jr. Renal mass core biopsy: accuracy and impacton clinical management. AJR Am J Roentgenol. 2007;188:563–70.
- Wood BJ, Khan MA, McGovern F,Harisinghani M, Hahn PF, Mueller PR. Imaging guided biopsy of renal masses:indications, accuracy and impact on clinical management. J Urol. 1999;161:1470–4.
- Sahni VA, Silverman SG. Biopsy of renal masses: when and why. CancerImaging. 2009; 9(1):44–55.
- Frank I, Blute ML, Cheville JC,Lohse CM, Weaver AL, Zincke H. Solid renal tumors: an analysis of pathologicalfeatures related to tumor size. J Urol. 2003;170:2217–20.
- McKiernan J, Yossepowitch O,Kattan MW, Simmons R, Motzer RJ, Reuter VE, et al. Partial nephrectomy forrenal cortical tumors: pathologic findings and impact on outcome. Urology.2002;60:1003–9.
- Kyei MY, Klufio GO, Mensah JE,Gyasi R, Gepi-Attee S, Ampadu K. Nephrectomy in Adults: Experience at the KorleBu Teaching Hospital Accra, Ghana. Saudi J Kidney Dis Transpl. 2015;26(3):638-42.
- Huang WC, Elkin EB, Levey As,Jang TL, Russo P. Partial Nephrectomy Versus Radical Nephrectomy in PatientsWith Small Renal Tumors—Is There a Difference in Mortality and CardiovascularOutcomes? J Urol. 2009;181:55-62.
- Coppin C, Porzsolt F, Awa A,Kumpf J, Coldman A, Wilt T. Immuno-therapy for advanced renal cell cancer.Cochrane Database Syst Rev. 2005:CD001425.
- Mazza C,Escudier B, Albiges L. Nivolumab in renal cell carcinoma: latest evidence andclinical potential. Ther Adv Med Oncol. 2017; 9(3):171-81.
- Herr Harry. History of partialnephrectomy. J Urol. 2005;173(3):705-8.
- Gill IS, Kavoussi LR, Lane BR,Blute ML, Babineau D, Colombo JR Jr, et al. Comparison of 1,800 Laparoscopicand Open Partial Nephrectomies for Single Renal Tumors. J Urol. 2007;178(1):41-6.
- Patel MN, KraneLS, Bhandari A, Laungani RG, Shrivastava A, Siddiqui SA, et al. Robotic PartialNephrectomy for Renal Tumors Larger Than 4 cm. Eur Urol. 2010;57:310-6.
- Kopp RP, Mehrazin R, Palazzi KL,Liss MA, Jabaji R, Mirheydar HS, et al. Survival outcomes after radical andpartial nephrectomy for clinical T2 renal tumours categorised by R.E.N.A.L. nephrometryscore. BJU Int. 2014;114:708–18.
- Mir MC, Derweesh I, Porpiglia F,Zargar H, Mottrie A, Autorino R. Partial Nephrectomy Versus Radical Nephrectomyfor Clinical T1b and T2 Renal Tumors: A Systematic Review and Meta-analysis ofComparative Studies. Eur Urol. 2017;71:606- 17.
- Nahar B, Gonzalgo ML. What is thecurrent role of partial nephrectomy for T2 tumors? Can J Urol. 2017;24(2):8698-704.
- Thompson RH, Lane BR, Lohse CM,Leibovich BC, Fergany A, Frank I, et al. Every minute counts when the renalhilum is clamped during partial nephrectomy. Eur Urol 2010;58(3):340-5.
- Mir MC, ErcoleC, Takagi T, Zhang Z, Velet L, Remer EM, et al. Decline in Renal Function afterPartial Nephrectomy: Etiology and Prevention. J Urol. 2015;193:1889-98.