Case Report
Clinical Implication of Non-Complement-Binding Donor- Specific Anti-HLA Antibodies in Heart Transplant Recipients -Risk Stratification by C1q-Binding Capacity
Takuma Sato1, Osamu Seguchi1, Yoshiaki Kanaumi2, Yuto Kumai1, Sachi Matsuda3, Haruki
Sunami1, Kensuke Kuroda1, Yorihiko Matsumoto4, Seiko Nakajima1, Koji Arai2, Kyoichi Wada3,
Hiroki Hata4, Satsuki Fukushima4, Masanobu Yanase1, Hatsue Ishibashi-Ueda5, Tomoyuki
Fujita4, Junjiro Kobayashi4 and Norihide Fukushima1*
1Department of Transplantation, National Cerebral and Cardiovascular Center, Osaka, Japan
2Department of Transfusion Medicine, National Cerebral and Cardiovascular Center, Osaka, Japan
3Department of Pharmacy, National Cerebral and Cardiovascular Center, Osaka, Japan
4Department of Cardiovascular Surgery, National Cerebral and Cardiovascular Center, Osaka, Japan
5Department of Pathology, National Cerebral and Cardiovascular Center, Osaka, Japan
*Corresponding author: Norihide Fukushima, Department of Transplant Medicine, National Cerebral and Cardiovascular Center, 5-7-1 Fujishirodai Suita Osaka, 565-8565, Japan
Published: 02 Nov, 2017
Cite this article as: Sato T, Seguchi O, Kanaumi Y, Kumai
Y, Matsuda S, Sunami H, et al. Clinical
Implication of Non-Complement-Binding
Donor-Specific Anti-HLA Antibodies
in Heart Transplant Recipients -Risk
Stratification by C1q-Binding Capacity.
Clin Surg. 2017; 2: 1703.
Abstract
Background: The development of de novo human leukocyte antigen (HLA) donor specific antibodies
(DSA), detected by either cytotoxic or solid phase assays, was considered the major risk factor for
cardiac graft failure in heart transplantation. However, it was shown that not all patients with
persistent production of DSA suffered loss of their allografts. The ability to activate complement may
be an important factor differentiating clinically relevant DSA from non- relevant DSA. Recently, a
C1q-binding assay (C1qScreen; One Lambda, Inc. Canoga Park, CA) has been developed to identify
complement-fixing HLA antibodies with high sensitivity and specificity. The aim of this study was
to investigate the association between C1q-binding ability of HLA-DSA and the clinical outcomes
post-transplant to identify clinically significant DSA after heart transplantation.
Methods: We enrolled 64 consecutive patients who received heart transplant between May 1999
and January 2015 in our institute. Sixty of 64 patients (93.7%) were screened for the presence of
circulating DSA using Luminex Single Antigen Flow Bead assays between June 2014 and August
2015, and patients with post-transplant DSA with mean fluorescence intensity (MFI) >500 were
selected to assess C1q fixation by C1q-binding assays. The clinical outcomes were compared with
the results.
Results: Of 60 patients, twelve patients were considered as DSA positive (MFI >500, range 698-5952,
class 1: 75% class 2: 17%, class1+2: 8%). All of these patients were identified as C1q negative. As the
results, we divided into two groups; group C1q negative DSA (n=12) and group non-DSA (n=48).
The rejection episodes, cardiac events, mortality, the development of cardiac graft vasculopathy and
cardiac function were not statistically different between the two groups.
Conclusion: Patients producing C1q-negative DSA had good graft survival, which was comparable
to that of DSA negative patients. Adding the assessment of the complement-binding capacity of DSA
might redefine the traditional risk stratification of DSA positive patients after heart transplantation.
Keywords: Heart transplantation; Donor specific antibodies; Antibody mediated rejection; C1q;
Complement-binding ability
Introduction
Donor-specific anti-HLA antibodies (DSA) in heart transplantation (HTx) are associated with
increased cardiac allograft injury and loss, including vascular injury and graft dysfunction [1-3].
The recent introduction of highly sensitive and specific techniques for the detection of anti-HLA
antibodies, such as the Luminex single-antigen bead (SAB) assay, has increased our ability to
identify sensitized patients and to define their immunological risk before and after transplantation.
Although this new technology is highly sensitive, the clinical impact of DSA detected only by the
solid phase assay is still controversial.
Various studies have indicated an increased risk of antibodymediated
rejection (AMR) and an inferior outcome of cardiac
transplant recipients who developed DSA that was detected only
with the SAB assay and not with complement-dependent cytotoxicity
(CDC) or flow cytometric assays [3-5]. However, it is also known
that not all patients with persistent production of DSA suffer loss of
their allografts, indicating that DSA are not equal in terms of their
detrimental effects on allograft dysfunction probably due to AMR.
Since complement activation by antibodies determines the
cytotoxic potential of these DSA, assessment of complement fixing
ability may be important for the characterization of clinically relevant
DSA.
A new solid-phase C1q-binding immunoassay (C1qScreen; One
Lambda, Inc. Canoga Park, CA) has been developed to identify the C1q
fixation capacity of anti-human leukocyte antigen (HLA) antibodies,
which is the first step in the activation of the classical complement
cascade. AC1q-positive de-novo DSA has been shown to be associated
with an increased rate of AMR and transplant glomerulopathy in
kidney transplantation [6-9]. However, the prevalence and clinical
significance of DSA characterized by C1q-binding have not been well
investigated in adult heart transplant patients.
The aim of this study was to investigate the association between
the C1q-binding ability of DSA and clinical outcomes posttransplant,
in order to elucidate clinical significance of DSA in heart
transplantation.
Methods
Study population
In this population-based study, we enrolled all 60 consecutive
patients who underwent heart transplantation at the National Cerebral
and Cardiovascular Center, Japan, between May 1999 and January
2015, and who underwent the SAB assay of antibodies against HLA.
In Japan, no patient with a positive prospective cytotoxic cross match
against the donor T lymphocytes undergoes heart transplantation,
according to the national rule for recipient selection. The stored
patient sera that were sampled between June 2014 and August 2015
were retrospectively analyzed using a C1q-binding assay. All patients
included in this study were followed for at least one year after HTx.
This study was approved by the local Ethics Committee in our
institute and was in compliance with local laws and regulations.
Immunosuppression and follow-up
After transplantation, all patients received a standard tripledrug
combination immunosuppressive therapy consisting of a
calcineurin inhibitor (cyclosporine or tacrolimus), mycophenolate
mofetil (MMF) and prednisolone (PSL). Induction therapy with
monoclonal or polyclonal antibodies, such as the murine monoclonal
anti-CD3 antibody (OKT3R) or an anti-IL-2 monoclonal antibody
(basiliximab), was also used in patients with renal dysfunction in the
perioperative period. Steroids were routinely withdrawn over time
unless there was a major rejection event or there was granulocytopenia
caused by other immunosuppressive drugs. Everolimus with reduced
calcineurin inhibitor was used since 2007 after transplantation in
patients with renal dysfunction and/or transplant cardiac allograft
vasculopathy (CAV).
After transplant, endomyocardial biopsies were performed at 1,
2, 3, 5, 7 and 11 weeks, at 4, 5, 6, 9, and 12 months, then every 6
months for the next 4 years, following which they were performed
annually. Endomyocardial biopsies were also performed whenever
acute cellular rejection (ACR) or AMR was clinically suspected.
Histopathological results were based on the International Society
for Heart and Lung Transplantation (ISHLT) standardized cardiac
biopsy grading. Additionally, AMR was monitored by performing
the flow cytometric panel reactive antibody (Flow PRA) test, or the
SAB assay, to determine the presence of DSA, and by analyzing
pathological findings that were monitored by immunohistochemical
staining of endomyocardial biopsy tissue for analysis of parameters
such as C4d staining and CD68 positive cells. Coronary angiography
and intravascular ultrasonography (IVUS) were performed within
the first to eleventh week to define a donor transmitted coronary
arterial disease, at 12 months and on an annual basis thereafter
after HTx. All the angiograms and IVUS findings were reviewed for
evidence of CAV. Diagnosis of CAV was based on a minimum of 0.5
mm progression on a maximum intimal thickness (MIT) from the
baseline on IVUS.
All outpatients were scheduled to be followed at least once a
month after HTx to check their clinical condition by performing
an electrocardiogram (ECG) and several laboratory tests including
analysis of the trough level of immunosuppressive drugs.
Echocardiography and coronary flow reverse (CFR) tests were
conducted every 6 months in every patient.
HLA typing
All recipients and donors were typed for HLA-A, HLA-B, HLA-C
and HLA-DR using the Luminex assay system and HLA typing kits
(WAKFlow HLA Typing kits, Wakunaga, Osaka, Japan). In Japan,
prospective cytotoxic cross matching is performed by the Japan
Organ Transplant Network to select recipients and any patient with
a positive T lymphocyte cross match is not selected. Non-cytotoxic
flow cytometric cross matching was also performed in our institution
in every transplant patient.
Detection and characterization of donor specific
antibodies
Monitoring of de novo DSA production had been prospectively
performed on the serum samples that were collected at regular
intervals after transplantation. We performed a Flow PRA test every
day after HTx for 2 weeks, then once a week thereafter until discharge,
unless there was clinical suspicion for rejection. For outpatients, this
test was performed at least once every 3 months in normal outpatients
and every month in highly sensitized patients.
Every patient was screened using Flow PRA class 1 and class 2
Screening beads. To determine the donor specificity of detected
antibodies, positive sera had been previously tested using SAB assays
(LAB Screen Class 1 and 2 Single Antigen Beads, One Lambda, Inc.)
according to the manufacturer’s instructions. Analysis was performed
using Fusion software (One Lambda, Inc.). Anti-HLA antibodies with
mean florescence intensity (MFI) values greater than 500 at any point
after HTx were considered positive. Transient DSA that temporarily
appeared and then disappeared without any medical intervention
were not considered to be positive DSA. Patients with positive DSA
were selected for assessment of C1q fixation using C1q-binding
assays (Figure 1). Non-HLA specific antibodies and IgG types were
not analyzed in this study.
Characterization of DSA by C1q assay
In patients with positive flow PRA, the ability of the DSA to fix
complement was determined using SAB and C1q screen kits (One Lambda Inc.) according to the manufacturer’s instructions. The same
SAB batch was used to detect pan-IgG and C1q-binding anti-HLA
antibodies in any given serum. Although the C1q assay can detect
IgG and non-IgG antibodies, all patients were primarily of the IgG
subtype because the sera tested with the C1q assay were selected on
the basis of the presence of IgG-DSA. Data were analyzed using MFI
values, and the cut-off for a positive reaction was set as an MFI value
greater than 500. Positive and negative control sera were included in
each test.
Allograft biopsies
Allograft biopsy surveillance was standardized and performed
in all patients as follows. All endomyocardial biopsy specimens were
graded based on the standard biopsy grading scheme, according to
the 1990 ISHLT classification [10,11]. ISHLT grade 3A or higher
rejection was usually treated with steroid pulse therapy. If there
was resistance to pulse therapy, cytolytic therapy consisting of
monoclonal or polyclonal antibodies was instituted. A rejection
episode ≥ISHLT grade 2 that occurred any time during the follow
up period was analyzed as a variable in this study. Diagnosis of AMR
was made based on the 2011 ISHLT consensus as follows: histological
evaluation of endothelial activation, intravascular macrophages
and capillary destruction. Additionally, immunofluorescence (for
C4d analysis) and immunoperoxidase (for CD68 analysis) staining
of endomyocardial biopsy tissue were done for evaluation of AMR.
Pathological AMR (pAMR) grading categories were: pAMR 0,
negative; pAMR 1, (I+) immunohistological AMR alone, (H+)
histological AMR alone; pAMR 2, pAMR that was both (H+) and
(I+); and pAMR 3, severe pAMR [12]. pAMR >1 that occurred at any
time during the follow-up period was analyzed as a variable in this
study. pAMR >2 was treated with corticosteroids, plasmapheresis,
intravenous immunoglobulin (IVIg), and rituximab (anti-B-cell
antibodies).
Statistical analysis
Descriptive statistics are presented as means ± standard deviation
(SD), showing the median value (range) or number (percentage)
for data with normal distribution or non-normal distribution,
respectively. Patients were divided into groups according to
DSA status. Chi-square analysis or Student’s t-test were used for
comparison of variables between groups, as appropriate. Statistical
significance was set at p< 0.05. The Kaplan-Meier method was used
to calculate cardiac event-free survival and pAMR event-free survival.
Statistical comparisons of survival curves according to DSA status
were made using Wilcoxon’s rank test. All data were analyzed using
JMP version 12.0 (SAS Institute, Cary, NC, USA).
Figure 1
Table 1
Results
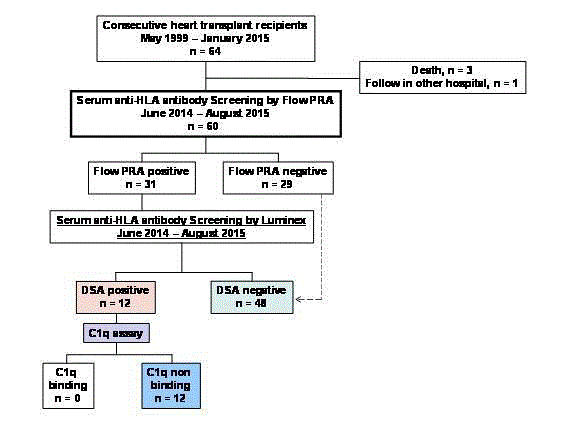
A flow chart of the study population is shown in Figure 1, and
the demographics and characteristics of the patients are summarized
in Table 1. Of 64 consecutive patients who received a HTx between
May 1999 and January 2015, 47 (78.3%) were men, with a mean age
of 36.1 ± 11.3 years (range, 17 years to 60 years) at transplant. The
mean time of follow-up of patients was 6.7 ± 4.1 years (range 1.2
years to 16.3 years). Of the 64 patients, 60 (93.7%) were screened for
HLA-antibodies using Flow PRA during the study period. Thirtyone
(51.6%) patients were Flow PRA positive against donor HLA.
Ultimately, 12 patients were considered as DSA positive. The DSA
positive patients were screened with the C1q assay, and none of them
were identified as C1q positive. No patient lost a graft during the
study period, except for 3 patients who died and were excluded from
the study (Figure 1).
Based on these findings, we divided the patients into two groups;
a DSA positive, C1q negative group (n=12; C1q negative DSA group)
and a DSA negative group (n=48; non-DSA group).Comparison
of the clinical information of the two groups is shown in Table 2.
The follow-up period, rejection episodes, cardiac events, all-cause
mortality, development of CAV and cardiac function were not
statistically different between the two groups.
The characteristics of the DSA found in the serum samples of
the 12 DSA positive patients are summarized in Table 3. Of these
patients, 75% (n=9) had only class1 DSA; 8.3% (n=1) had only class 2
DSA; and 16.6% (n=2) had both class1 and class 2 DSA. Fifty percent
(n=6) of the patients with DSA were treated with triple immune
suppressive therapy (IST) including steroids, whereas only 10.4% (n=5) of the patients in the non DSA group were treated with triple
IST (p=0.0015), which indicated a tendency to perform strict IST
in DSA positive patients. Induction therapy was performed in 75%
(n=9) of the DSA positive patients (OKT3R, n=2; Basiliximab, n=7).
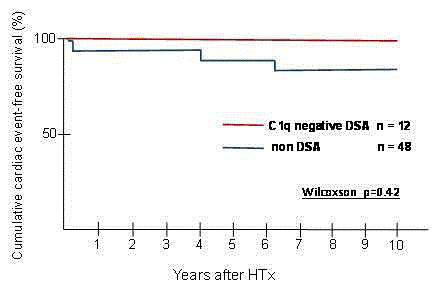
During the follow-up period, the composite endpoint of cardiac
event (percutaneous coronary intervention (PCI), n=2; coronary
artery bypass grafting (CABG), n=1) occurred in 3 patients in the
non-DSA group, but did not occur in any of the patients in the C1q
negative DSA group. There was no significant difference in cumulative
cardiac event-free survival between the 2 groups (Wilcoxon test,
p=0.42) (Figure 2A). Each patient had a history of refractory ACR or
had been treated for pAMR2. One patient underwent PCI one month
after HTx because of donor-transmitted disease.
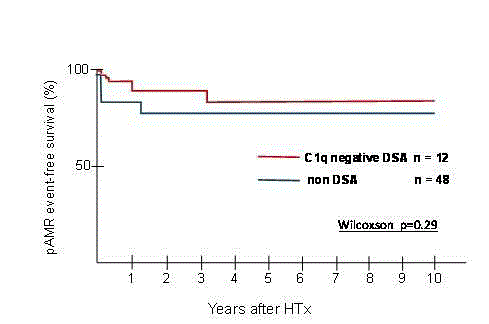
There was no significant difference in pAMR event free-survival
between the two groups (Wilcoxon test, p=0.29) (Figure 2B). Two
(16.6%) patients in the C1q negative DSA group, and 4 (8.3%) patients
in the non-DSA group experienced pAMR >grade 1.
Table 2
Table 2
Clinical information of the patients without, and with (Ciq negative), donor-specific antibobies.
Table 3
Discussion
In this study, the association of C1q binding status with anti-
HLA antibody and clinical outcomes of HTx was investigated. We
found that patients with C1q-negative DSA had good graft survival
and AMR-free survival, which were comparable to those of non-DSA
patients. In other words, patients with DSA showed a better outcome
if their anti-HLA antibodies did not show a complement-binding
capacity in the C1q assay.
An updated consensus for diagnosing AMR was published in
2011, which specifically detailed histological AMR features based
on hematoxylin eosin (H-E) staining, immunofluorescence staining,
or both. Clinical cardiac symptoms and hemodynamic status are no
longer needed for diagnosing AMR because of recent studies that demonstrated an increased development of CAV and inferior survival
in patients with asymptomatic biopsy-proven AMR [1,12-14].
However, apart from the fact that this updated method of diagnosing
AMR recognizes the importance of diagnosing pathological
asymptomatic AMR before it becomes clinically symptomatic, [15,16]
the issues of invasiveness of the biopsy procedures, timing intervals,
and the differences in immunofluorescence staining methods among
institutions have not been solved.
Recent studies have indicated a correlation between the
serological existence of DSA detected after heart transplantation and
developing AMR, CAV, graft injury and graft loss [17,18]. Due to the
limited value of histological change for the diagnosis of late AMR
and its invasiveness, and to complications accompanying repeated
biopsy procedures, DSA identified by using sensitive solid-phase
immunoassays have come to play a more important role as a noninvasive,
repeatable diagnostic method for predicting prognosis after
HTx.
Besides the clinical value of monitoring serum DSA after heart
transplantation, it is also known that not all patients with persistent
production of DSA show a worse outcome. Since the solid phase IgGSAB
assay detects both complement-fixing and non-complementfixing
DSA, we further investigated this characteristic of DSA in our
patients to clarify risk stratifications among patients with DSA.
In present study, all of the patients with DSA turned out to be
C1q-negative. Since there were no patients with positive C1q DSA, we
compared the prognosis of the patients with non-complement DSA
with that of non-DSA patients to examine the prognosis and clinical
implication of possessing non-complement DSA. We found that
there were no significant differences in patient survival between C1qnegative
DSA and non-DSA patients. These results were consistent
with the hypothesis that if these antibodies do not fix complement,
and thus do not initiate the complement cascade, these antibodies may
not cause graft injury [19,20]. To our knowledge, there have been no
reports in the adult cardiac transplant literature of systematic studies
that evaluated the prognosis of patients producing C1q-negative
DSA. However, in a previous study of kidney transplantation, the
percentage of DSA-positive patients that were C1q-negative was
reported as 60% [21] and, consistent with our study, their graft
prognosis was far better than that of the C1q-positive DSA patients
[8,22,23].
One of the possible reasons as to why there were no C1q-positive
patients in our study is that recipients with a positive CDC assay are
excluded in donor matching in Japan. As a result, highly sensitized
patients with an extremely high titer of DSA were not included from
the start of this study.
To confirm this hypothesis, we retrospectively examined the sera
of recipient and donor candidates who had been judged as positive
CDC by the Japan Organ Transplant Network and for whom HTx
was therefore not performed, in order to check C1q binding ability.
These data confirmed that positive CDC results in an extremely high
MFI level of IgG DSA with positive C1q.
Some studies have reported a correlation between C1q-binding
ability and the MFI level of IgG DSA. Thus, it has been reported
that the MFI levels of IgG DSA in the C1q-positive group were
significantly higher than those in the C1q-negative group [7,24-26].
Zeevi et al. [26] reported a significant relationship between C1qbinding
activity and antibody strength as measured by the MFI titer,
in analysis of over 800 serum samples. Thammancichanond et al. [25]
also reported that C1q binding ability correlated with the strength of
DSA, which was measured as the MFI level; 11 of the 12 patients with
C1q-positive DSA in that study had an MFI level of IgG DSA >8,000.
Schaub et al. [24] reported an IgG-DSA cut-off value of 14154 in the
prediction of C1q positivity. On the other hand, other reports have
warned that C1q binding does not always correlate with the results of
the conventional SAB assay [6,7,21,26,27]. The complement cascade,
which is initiated by the binding of C1q, requires C1q binding sites
on at least two antibodies in close steric proximity. Thus, C1q binding
depends on both the density of DSA and their IgG subclass [28,29].
Since the standard SAB assay detects all types of IgG-subclass, the
proportion of the IgG subtypes included in each serum might explain
the discrepancies between the DSA-MFI titer and C1q positivity.
In our study, the MFI value of IgG DSA ranged from 698 to 6952,
and the mean MFI value was 1880 ± 1839, which was a significantly
lower titer compared to other previous reports. The lower MFI value
of IgG DSA in our study might be due to the method of selecting the
heart donors as already described, as well as the immunosuppressive
protocol of our institution.
Regarding IST, in our study patients with DSA were more likely
to be on triple immunosuppression with tacrolimus, mycophenolate
mofetil, and prednisone than on double immunosuppression, as a precaution against the development of AMR. Although we usually
hesitate to decrease the strength of IST in DSA-positive patients, the
favorable result of this study suggested that it might be possible to
reconsider the risk and the appropriate IST for each C1q-negative
DSA patient, based on the assumption of a harmless effect of C1qnegative
DSA.A decrease in IST may avoid unwanted events such as
infectious disease, malignancy and kidney dysfunction, which can
derive from the immunosuppressive therapy itself or from an overimmunosuppressive
regimen [30]. However, from a different point of
view, it could also be said that the intense immunosuppressive therapy
of the DSA positive patients in this study might have the advantage
of leading to insufficient production of DSA for the development of
AMR-related events. For this reason, careful observation of the MFI
level of IgG DSA is still recommended when trying to wean DSA
positive/C1q negative patients off strict immunosuppressive therapy
[31].
Nevertheless, some studies have reported an unfavorable
prognosis in kidney transplanted patients that show long-term
persistence of C1q-negative DSA [24], and the clinical roles and
the effects of possessing non-complement-binding DSA are still
controversial [25,32]. An important point that needs to be kept in
mind is that a negative C1q result does not mean that the detected
DSA will remain that way forever. This is because a low titer of DSA
might significantly increase due to a strong and durable immune
memory response in sensitized situations, and a negative C1q DSA
might change to C1q-positive. Although our patients with C1q
negative DSA showed good survival in the follow up period (mean
follow up period, 6.7 ± 4.1 years), it is still necessary to closely observe
C1q negative patients with a high DSA-MFI, balancing the risk and
benefit of reducing IST, and also the cost and benefit of the C1q assay
and IgG subclass analysis.
There are several limitations of this study. First, we could
not evaluate the prognosis of positive DSA patients with positive
C1qsince there was no such study participant. Second, the impact of
non-HLA specific antibodies and IgG types were not considered in
this study. Since our study was limited to a small cohort, continuous
and additional studies are necessary to further confirm the prognosis
of patients producing C1q-negative DSA.
In conclusion, this study demonstrated that patients producing
non-complement binding DSA had good graft survival, which was
comparable to that of DSA negative patients. Therefore, addition
of the assessment of the complement-binding capacity of DSA to
the diagnosis of DSA patients might redefine the traditional risk
stratification of DSA positive patients after HTx.
Figure 2
Figure 2
Cumulative Cardiac event-free survival in patients with and without
DSA development (n=60).
Figure 3
References
- Kobashigawa J, Crespo-Leiro MG, Ensminger SM, Reichenspurner H, Angelini A, Berry G, et al. Report from a consensus conference on antibody-mediated rejection in heart transplantation. J heart lung transplant. 2011;30:252-69.
- Reed EF, Demetris AJ, Hammond E, Itescu S, Kobashigawa JA, Reinsmoen NL, et al. Acute antibody-mediated rejection of cardiac transplants. The J heart lung transplant. 2006;25:153-9.
- Smith JD, Banner NR, Hamour IM, Ozawa M, Goh A, Robinson D, et al. De novo donor hla-specific antibodies after heart transplantation are an independent predictor of poor patient survival. Am J transplant. 2011;11:312-9.
- Ho EK1, Vlad G, Vasilescu ER, de la Torre L, Colovai AI, Burke E, et al. Pre- and posttransplantation allosensitization in heart allograft recipients: major impact of de novo alloantibody production on allograft survival. Hum Immunol. 2011;72(1):5-10.
- Kaczmarek I, Deutsch MA, Kauke T, Beiras-Fernandez A, Schmoeckel M, Vicol C, et al. Donor-specific hla alloantibodies: Long-term impact on cardiac allograft vasculopathy and mortality after heart transplant. Exp Clin Transplant. 2008;6:229-35.
- Chen G, Sequeira F, Tyan DB. Novel c1q assay reveals a clinically relevant subset of human leukocyte antigen antibodies independent of immunoglobulin g strength on single antigen beads. Human immunology. 2011;72:849-58.
- Crespo M, Torio A, Mas V, Redondo D, Perez-Saez MJ, Mir M, et al. Clinical relevance of pretransplant anti-hla donor-specific antibodies: Does c1q-fixation matter? Transplant immunology. 2013;29:28-33.
- Piazza A, Poggi E, Ozzella G, Adorno D. Post-transplant development of c1q-positive hla antibodies and kidney graft survival. Clinical transplants. 2013:367-75.
- Yabu JM, Higgins JP, Chen G, Sequeira F, Busque S, Tyan DB. C1q-fixing human leukocyte antigen antibodies are specific for predicting transplant glomerulopathy and late graft failure after kidney transplantation. Transplantation. 2011;91:342-7.
- Billingham ME, Cary NR, Hammond ME, Kemnitz J, Marboe C, McCallister HA, et al. A working formulation for the standardization of nomenclature in the diagnosis of heart and lung rejection: Heart rejection study group. J Heart Transplantation. 1990;9:587-93.
- Stewart S, Winters GL, Fishbein MC, Tazelaar HD, Kobashigawa J, Abrams J, et al. Revision of the 1990 working formulation for the standardization of nomenclature in the diagnosis of heart rejection. J Heart Lung Transplant. 2005;24:1710-20.
- Berry GJ, Burke MM, Andersen C, Bruneval P, Fedrigo M, Fishbein MC, et al. The 2013 international society for heart and lung transplantation working formulation for the standardization of nomenclature in the pathologic diagnosis of antibody-mediated rejection in heart transplantation. J Heart Lung Transplant. 2013;32:1147-62.
- Berry GJ, Angelini A, Burke MM, Bruneval P, Fishbein MC, Hammond E, et al. The ISHLT working formulation for pathologic diagnosis of antibody-mediated rejection in heart transplantation: Evolution and current status (2005-2011). J Heart Lung Transplant. 2011;30:601-11.
- Loupy A, Toquet C, Rouvier P, Beuscart T, Bories MC, Varnous S, et al. Late failing heart allografts: Pathology of cardiac allograft vasculopathy and association with antibody-mediated rejection. Am J Transplant. 2016;16:111-20.
- Kfoury AG, Hammond ME, Snow GL, Drakos SG, Stehlik J, Fisher PW, et al. Cardiovascular mortality among heart transplant recipients with asymptomatic antibody-mediated or stable mixed cellular and antibody-mediated rejection. J Heart Lung Transplant. 2009;28:781-4.
- Wu GW, Kobashigawa JA, Fishbein MC, Patel JK, Kittleson MM, Reed EF, et al. Asymptomatic antibody-mediated rejection after heart transplantation predicts poor outcomes. J Heart Lung Transplant. 2009;28:417-22.
- Frank R, Molina MR, Goldberg LR, Wald JW, Kamoun M, Lal P. Circulating donor-specific anti-human leukocyte antigen antibodies and complement c4d deposition are associated with the development of cardiac allograft vasculopathy. Am J Clin Pathol. 2014;142:809-15.
- Tible M, Loupy A, Vernerey D, Suberbielle C, Beuscart T, Cazes A, et al. Pathologic classification of antibody-mediated rejection correlates with donor-specific antibodies and endothelial cell activation. J Heart Lung Transplant. 2013;32:769-76.
- Baldwin WM 3rd, Pruitt SK, Brauer RB, Daha MR, Sanfilippo F. Complement in organ transplantation. Contributions to inflammation, injury, and rejection. Transplantation. 1995;59:797-808.
- Smith JD, Hamour IM, Banner NR, Rose ML. C4d fixing, luminex binding antibodies - a new tool for prediction of graft failure after heart transplantation. Am J Transplant. 2007;7:2809-15.
- Loupy A, Lefaucheur C, Vernerey D, Prugger C, Duong van Huyen JP, Mooney N, et al. Complement-binding anti-HLA antibodies and kidney-allograft survival. N Engl J Med. 2013;369(13):1215-26.
- Sutherland SM, Chen G, Sequeira FA, Lou CD, Alexander SR, Tyan DB. Complement-fixing donor-specific antibodies identified by a novel c1q assay are associated with allograft loss. Pediatr transplant. 2012;16:12-7.
- Thammanichanond D, Mongkolsuk T, Rattanasiri S, Kantachuvesiri S, Worawichawong S, Jirasiritham S, et al. Significance of c1q-fixing donor-specific antibodies after kidney transplantation. Transplant proc. 2014;46:368-71.
- Schaub S, Hönger G, Koller MT, Liwski R, Amico P. Determinants of C1q binding in the single antigen bead assay. Transplantation. 2014;98(4):387-93.
- Thammanichanond D, Wiwattanathum P, Mongkolsuk T, Kantachuvesiri S, Worawichawong S, Vallipakorn SA, et al. Role of pretransplant complement-fixing donor-specific antibodies identified by c1q assay in kidney transplantation. Transplant proc. 2016;48:756-60.
- Zeevi A, Lunz J, Feingold B, Shullo M, Bermudez C, Teuteberg J, et al. Persistent strong anti-hla antibody at high titer is complement binding and associated with increased risk of antibody-mediated rejection in heart transplant recipients. J Heart Lung Transplant. 2013;32:98-105.
- Chin C, Chen G, Sequeria F, Berry G, Siehr S, Bernstein D, et al. Clinical usefulness of a novel c1q assay to detect immunoglobulin g antibodies capable of fixing complement in sensitized pediatric heart transplant patients. J Heart Lung Transplant. 2011;30:158-63.
- Bartel G, Wahrmann M, Exner M, Regele H, Schillinger M, Horl WH, et al. Determinants of the complement-fixing ability of recipient presensitization against hla antigens. Transplant. 2007;83:727-33.
- Kushihata F, Watanabe J, Mulder A, Claas F, Scornik JC. Human leukocyte antigen antibodies and human complement activation: role of IgG subclass, specificity, and cytotoxic potential. Transplantation. 2004;78(7):995-1001.
- Kittleson MM, Kobashigawa JA. Long-term care of the heart transplant recipient. Curr Opin Organ Transplantation. 2014;19(5):515-24.
- Hoshino J, Kaneku H, Everly MJ, Greenland S, Terasaki PI. Using donor-specific antibodies to monitor the need for immunosuppression. Transplantation. 2012;93:1173-8.
- Guidicelli G, Guerville F, Lepreux S, Wiebe C, Thaunat O, Dubois V, et al. Non-complement-binding de novo donor-specific anti-hla antibodies and kidney allograft survival. J Am Soc Nephrol. 2016;27:615-25.