Case Report
Mycotic Aneurysm of the Ascending Aorta and Aspergillus Fumigatus Mediastinitis Early after Cardiac Transplantation
Paul R. Vogt1*, Elisabeth Hasler2, Daniel Schmidlin3, Pascal Berdat1, Phillip Th. Went4, Markus
Flepp5 and Paul Mohasci,sup6
1Department of Cardiovascular Surgery, Klinik Im Park, Switzerland
2Department of Pneumology, University Hospital Zurich, Switzerland
3Department of Anaesthesia and Intensive Care, Klinik Im Park, Switzerland
4Pathology Institute Enge, Switzerland
5Department of Infectious Diseases, Klinik Im Park, Switzerland
6Department of Cardiology, Klinik Im Park, Switzerland
*Corresponding author: Paul R. Vogt, Department of Cardiovascular Surgery, Heart Centre Zurich, Klinik Im Park, Bellariastrasse 38, CH-8038 Zurich, Switzerland
Published: 16 Oct, 2017
Cite this article as: Vogt PR, Hasler E, Schmidlin D, Berdat
P, Went PTh, Flepp M, et al. Mycotic
Aneurysm of the Ascending Aorta and
Aspergillus Fumigatus Mediastinitis
Early after Cardiac Transplantation. Clin
Surg. 2017; 2: 1681.
Abstract
A 66-year old man after orthotopic heart transplantation developed severe infection with Aspergillus
fumigates covering the entire thoracic cavity with a thick fungal turf. Repeated surgical removal of
infectious and necrotic tissue together with innovative topical treatment using voriconazole and
chlorhexidine combined with systemic antifungal treatment lead to elimination of the infection.
Definite wound closure was achieved by latissimus dorsi muscle flap plasty and standard sterna
refixation. To the best of our knowledge, we report the first survival of extensive A.fumigatus
mediastinitis after heart transplantation due to repeated debridement in combination with novel
topical application of antifungal agents.
Keywords: Aspergillus fumigatus; Fungal Infection; Heart transplant; Immunosuppression;
Mediastinitis; Mycotic aneurysm; Topical treatment; Chlorhexidine; Voriconazole
Background
Despite treatment options with antifungal drugs and surgery, Aspergillus mediastinitis
is generally fatal, particularly in immunocompromised patients following orthotopic heart
transplantation (OHT), which is associated with the highest risk of postoperative mediastinitis
among all cardiothoracic procedures [1,2].
Herein, we describe a patient with early postoperative invasive A.fumigatus infection, who
was referred with complete sternal dehiscence 43 days after OHT. In this situation, usual surgical
debridement and intravenous antifungal treatment were thought to be hopeless due to the
massive degree of Aspergillus dissemination carrying a mortality rate between 90 and 100% in
immunosuppressed patients [1,3]. However, using a combination of innovative surgical and topical
antifungal treatment the patient could be cured from this usually fatal complication.
Case Presentation
A 66-year old male patient underwent OHT due to a long-standing history of ischemic heart
disease: acute anterior myocardial infarction in 1991; onset of ischemic heart failure in 1993; coronary
artery bypass grafting and left ventricular aneurysmectomy in 1994; recurrent myocardial infarctions
until 2000; heart failure with a left ventricular ejection fraction of 10% despite repeated percutaneous
coronary interventions until 2010; radiofrequency ablation, cardiac resynchronisation, MitraClip
procedure, hemofiltration and levosimendan infusions in 2011; finally, in June 2012, installation of
an intra-aortic balloon pump and extended cardiac life support with extracorporeal circulation to
successfully bridge the patient to a left ventricular assist device (HeartWare®). After full recovery,
OHT was performed in July 2013. Basiliximab was administered as inductive immunosuppression,
followed by maintenance immunosuppression consisting of tacrolimus, mycophenolat mofetil
and prednisone. Co-morbidities included chronic gastritis, amiodarone-induced subclinical
hypothyroidism, type-II diabetes mellitus, kidney cysts and chronic prostatitis, which all were well controlled. The post-transplant period was complicated by bilateral
exsudative pleuritis, thrombosis of internal jugular vein, neutropenia
and persistent sinus bradyarrhythmia with implantation of a twochamber
pacemaker using the left cephalic vein.
Thirty-seven days after OHT, progressive infection of the median
sternotomy wound with a grayish-yellow discharge was noted
starting at the epigastrium and resulting in complete skin necrosis
and wound dehiscence with direct view on the transplanted heart.
However no microorganisms were identified. CT-scan reported
pneumomediastinum and a left-sided sero-pneumothorax of 300ml
at the site of the previous LVAD, compressing the fibrotic left lower
lung lobe.
The patient was referred to our clinic 43 days after OHT pretreated
with meropenem, vancomycin, linezolid, tigecycline,
fluconazole and voriconazole. At clinical examination, the fully awake
and neurologically normal patient was breathing spontaneously but
presented in poor general condition, malnourished (serum albumin
16 g/L) and with renal insufficiency (creatinine 150 μmol/l).
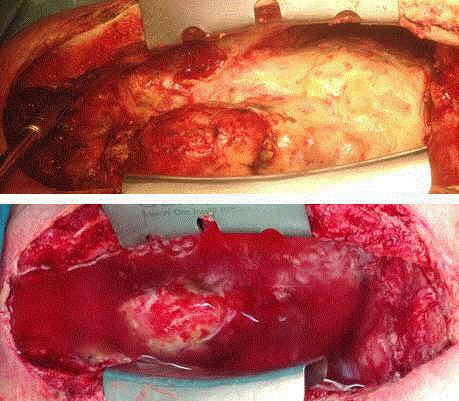
Surgical inspection revealed thick and pervasive, green-yellow
membranes covering the entire heart, great vessels, diaphragm, both
lungs as well as the entire chest wall (Figure 1). The turf also covered
the contained rupture of aorto-aortic anastomosis and the previously
constructed end-to-side anastomosis of the LVAD arterial return to
the distal native ascending aorta.
Residual parts of the vascular prosthesis were removed. There
was imminent rupture of the aorto-aortic anastomosis due to
mycotic infection of the native and transplanted ascending aorta,
both with positive cultures for Aspergillus fumigatus. The aorto-aortic
anastomosis was resected including a large proximal segment of the
native as well as a large distal segment of the transplanted ascending
aorta, which was easily possible due to the excessive length of the
entire aorta. Extensive debridement of all anatomical structures of
the thoracic cavity was performed and a vacuum-assisted closure
dressing (VAC) was applied.
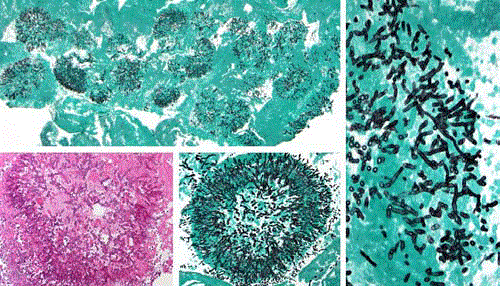
Histopathological examination (Figure 2), cultures as well
as sequencing of all specimens including pleural fluid uniformly
revealed A.fumigatus. Galactomannan test was positive (EIA index
0.1), but bacterial fungal as well as blood cultures revealed no other
microorganisms. CT-scan revealed no other organ involvement by
invasive aspergillosis.
In lack of a literature report on any similar case with such
extensive Aspergillus mediastinitis we empirically decided to pursue
the following empirical treatment: surgical wound re-opening every
third day, sharp and blunt mechanical scrubbing and cleaning of the
fungal turf, topical application of voriconazole and chlorhexidine 2%,
20-fold diluted (resulting in chlorhexidine 0.01%), dunking the entire
thoracic cavity for 40 min with each drug (Figure 3) and an open
chest treatment with VAC between the re-explorations. Pre-emptive
systemic antifungal therapy with a combination of caspofungin
and voriconazole was started. Following susceptibility testing and
determination of minimal inhibitory concentration (MIC 0.125
mg/l), voriconazole was continued at a serum concentration of 4 mg/l
- 6 mg/l. The immunosuppressive regimen was tailored and potential
organ rejection surveyed by echocardiography. Debridement and
topical therapy was performed a total of eight times. As by visible
affection of the sternal bone tissue, 5 mm of each sternal side was
removed, histopathology confirming A.fumigatus.
After the fourth surgical session, consistent with marked visible
decrease of the fungal turf overspreading the mediastinal organs,
repeated microscopic and microbiological examinations were found
to be sterile with regard to A.fumigatus. However, colonization with
multi-drug resistant Acinetobacter baumannii susceptible only to
polymyxin B (MIC 0.5 mg/l) was detected in the sixth session. Hence,
a diluted colistin-solution was applied topically to the thoracic cavity
and infected structures and colistin was administered intravenously
for one week. The postoperative course between each surgical session
was uneventful the patient remaining isolated but fully mobilized on
the regular ward.
After eight surgical revisions revealing clean intrathoracic tissues
and negative microbiological findings with respect to A.fumigatus,
definite wound closure was achieved 26 days after the first revision
with a latissimus dorsi muscle flap plasty to fill the persistent left-sided
thoracic cavity. Sternal refixation and wound closure were performed
according to our infection-prevention protocol (sternal wound
irrigation with vancomycin wax and gentamycin solution). Careful
reversal of immunosuppression allowed a stable cardiovascular course
without any signs of rejection checked by repeated endomyocardial
biopsy and echocardiography. The patient was discharged well 47
days after first surgical intervention with uneventful postoperative
recovery and wound healing. Oral voriconazole with regular
monitoring of liver function was continued for 12 months, cardiac medication and immunosuppression are continued corresponding to
state-of-the-art. One year after wound closure, no findings suspicious
for persistent or recurrent fungal infection on chest X-ray and CTscan
and negative serum galactomannan were reported. The patient
is in excellent condition with good functional capacity, normal
biventricular function, normal coronary angiography and no signs of
cardiac rejection.
Figure 1
Figure 1
Entire thoracic cavity and heart surface covered with Aspergillus
turf. Topical application of Chlorhexidin and Voriconazol solution into thoracic
cavity.
Figure 2
Figure 2
Grocott and HE stain showing fungus ball in the fibrinous tissue
taken from the sternum, heart surface, pleura and aorta with septated hypae
typical for Aspergillus fumigatus.
Discussion
Mediastinitis is a rare but severe complication of heart
transplantation, occurring in 1% to 10% of cases with Staphylococcus
spp. and Enterobacter spp. As the most common pathogens [4]. Our
patient presented with two rare pathogens of mediastinitis namely
Aspergillus fumigatus and Acinetobacter baumannii.
Aspergillus has been recognized as a major opportunistic pathogen
and accounts for the most common mycoses in cardiac transplant
recipients [1]. Diagnosis can be challenged by discriminating
colonization from invasive infection and because strains
morphologically identified as putative A.fumigatus are recognized
as different species by molecular methods with decreased antifungal
susceptibility [5]. This may lead to a delay of targeted treatment
resulting in an excessive mortality. Despite negative blood cultures
in the current patient, invasive infection could be documented by
numerous intraoperative specimens from the sternum, native and
transplanted aorta including anastomosis, all intrathoracic organs
and the thoracic wall, which uniformly showed the characteristic
hyphae consistent with Aspergillus on microscopic examination,
positive serum galactomannan test and the presence of A.fumigatus
was documented by culture and sequencing. Hence, a diagnosis of
invasive aspergillosis could be made in accordance with the definition
of the US Centres for Disease Control [6].
A.fumigatus mediastinitis in our patient occurred within the
usual time period of onset of invasive aspergillosis reported to be
between 36 and 52 days post transplantation [1]. Besides recent
cardiac transplantation, the current patient presented with previously
reported risk factors for the development of fungal infections such as
early postoperative wound problems, renal failure, administration of
multiple antibiotics, ventricular assist device before surgery, as well as
severe malnutrition and diabetes mellitus [1,3].
Given the unique extent of the disease, the basic principle
of therapy [6], i.e. intravenous antifungal treatment, tapering of
immunosuppression and surgical re-exploration alone was not
thought to be sufficient. With regards to the pathology of invasive
aspergillosis resulting in infarction and necrosis of affected organs
and poor drug delivery to tissue, our treatment regime was based on
the conviction of the necessity to clean large surfaces without blood
supply from thick layers of fungal material not only mechanically but
also with local antiseptics.
Eventually, a combination of repeated debridements with an
open chest treatment, cavity baths with aqueous voriconazole
and chlorhexidine, in addition to intravenous administration of
voriconazole and caspofungin and lastly a chest wall reconstruction
by a muscle flap plasty were necessary and lead to successful recovery
of this life-threatening disease.
A combined systemic antifungal therapy was started with
caspofungin and voriconazole, the latter guided by testing
susceptibility and serum concentration. Voriconazole, a triazole
with activity against yeasts and molds including Aspergillus spp. [7]
performed superior when compared with amphotericin B, and is
now recommended as primary treatment of invasive aspergillosis
including its uncommon manifestations such as mediastinitis and
osteomyelitis [6,8]. Voriconazole was administered systemically in
the only two reported surviving cases of Aspergillus mediastinitis
after OHT [2,4]. Monitoring of its serum levels is recommended
in transplanted patients because of impaired renal function and
significant interactions with cyclosporine, tacrolimus and sirolimus
[1,6]. In the current case, oral voriconazole was continued for 12
months to prevent relapse of infection, which has been reported to
occur several months after seemingly successful treatment. The role
of combined primary or salvage antifungal therapy is uncertain and
warrants a prospective, controlled clinical trial [6].
The use of topical voriconazole in addition to its intravenous
administration has recently been found to be effective in cases
of Aspergillus keratitis and endophtalmitis when administered
intravitreally [9-11]. Biocidal activity of chlorhexidine against
A.fumigatus was reported when applied topically at a concentration
of 0.06% for 30 min to 60 min and its stable and lasting adherence on
human tissue without being absorbed [12]. In addition, effectiveness
of topical chlorhexidine has also been shown in the treatment of
fungal keratitis without any adverse effects [13]. Based on this data,
we decided to use chlorhexidine in the same way as the voriconazole
solution as local antifungal treatment.
After the fourth surgical session, examinations revealed a
sterile condition with respect to A.fumigatus. However, a super
infection with a multi-drug resistant A.baumannii was reported.
Mediastinits due to A.baumannii is rare, with only two reported cases
in immunocompetent subjects, successfully treated with ampicillinsulbactam
and a pan-resistant type with tigecycline. [14,15] Notably,
only one case of multi-drug resistant A.baumannii in sternal wound
after OHT has been reported. Favourable outcome was achieved with
extensive debridement, systemic colistin and minocycline as well as
wound irrigation with 5% chlorhexidine and VAC dressing [16]. Safe
and effective topical use of colistin 0.19% has been reported in the
treatment of corneal infection by multi-drug resistant Pseudomonas
aeruginosa [17]. Hence, we used diluted colistin as described above
in combination with intravenous colistin. Lastly, after the completion
of topical antimicrobial therapy, the insertion of a latissimus dorsi
muscle flap into the thoracic cavity and complete closure of the
sternum resulted in a restitutio ad integrum.
Source of Funding
This work was supported by “EurAsia Heart – A Swiss Medical Foundation”, Zurich, Switzerland.
Conclusion
Our case report proves that invasive A.fumigatus infection, yet accompanied by a super infection with a multi-drug resistant A.baumannii can be eliminated with aggressive combined surgical, topical and systemic drug treatment even in an immunocompromised patient.
References
- Singh N, Paterson DL. Aspergillus infections in transplant recipients. Clin Microbiol Rev. 2005;18(1):44-69.
- Levin T, Suh B, Beltramo D, Samuel R. Aspergillus mediastinitis following orthotopic heart transplantation: case report and review of the literature. Transpl Infect Dis. 2004;6:129-31.
- Chou NK, Wang JL, Chi NH, Wu IH, Huang SC, Chen YS, et al. Surgical treatment of mediastinitis after cardiac transplantation. Transplantation proc. 2008;40:2629-30.
- Forestier E, Remy V, Lesens O, Martinot M, Hansman Y, Eisenmann B, et al. A case of Aspergillus mediastinitis after heart transplantation successfully treated with liposomal amphotericin B, caspofungin and voriconazole. Euro Soc Clini Microbio. 2005;24:347-9.
- Zbinden A, Imhof A, Wilhelm MJ, Ruschitzka F, Wild P, Bloemberg GV, et al. Fatal outcome after heart transplantation caused by Aspergillus lentulus. Transpl Infect Dis. 2012;14:E60-3.
- Walsh TJ, Anaissie EJ, Denning DW, Herbrecht R, Kontoyiannis DP, Marr KA, et al. Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America. Clin infect Dis. 2008;46:327-60.
- Johnson LB, Kauffman CA. Voriconazole: a new triazole antifungal agent. Clin infect Dis. 2003;36:630-7.
- Herbrecht R, Denning DW, Patterson TF, Bennett JE, Greene RE, Oestmann JW, et al. Voriconazole versus amphotericin B for primary therapy of invasive aspergillosis. N Engl J Med. 2002;347(6):408-15.
- Thiel MA, Zinkernagel AS, Burhenne J, Kaufmann C, Haefeli WE. Voriconazole concentration in human aqueous humor and plasma during topical or combined topical and systemic administration for fungal keratitis. Antimicrob Agents Chemother. 2007;51(1):239-44.
- Sen P, Gopal L, Sen PR. Intravitreal voriconazole for drug-resistant fungal endophthalmitis: case series. Retina. 2006;26(8):935-9.
- Jurkunas UV, Langston DP, Colby K. Use of voriconazole in the treatment of fungal keratitis. Int Ophthalmol Clin. 2007;47(2):47-59.
- Tortorano AM, Viviani MA, Biraghi E, Rigoni AL, Prigitano A, Grillot R, et al. In vitro testing of fungicidal activity of biocides against Aspergillus fumigatus. J Med Microbiol. 2005;54:955-7.
- Rahman MR, Johnson GJ, Husain R, Howlader SA, Minassian DC. Randomised trial of 0.2% chlorhexidine gluconate and 2.5% natamycin for fungal keratitis in Bangladesh. Br J Ophthalmol. 1998;82(8):919-25.
- Tekce AY, Erbay A, Cabadak H, Yagci S, Karabiber N, Sen S. Pan-resistant Acinetobacter baumannii mediastinitis treated successfully with tigecycline: a case report. Surgical infections. 2011;12:141-3.
- Lachanas E, Tomos P, Sfyras N, Miyakis S, Kostakis A. Acinetobacter baumannii mediastinitis after cardiopulmonary bypass: case report and literature review. Surgical infections. 2008;9:201-4.
- George RS, Birks EJ, Haj-Yahia S, Bowles CT, Hall A, Khaghani A, et al. Acinetobacter mediastinitis in a heart transplant patient. Ann Thorac Surg. 2006;82(2):715-6.
- Jain R MS, Motukupally SR, Jain M. Use of topical colistin in multiple drug-resistant Pseudomonas aeruginosa bacterial keratitis. Cornea. 2014;33(9):923-7.