Case Report
A Signet Ring Cell Gastric Carcinoma Case Diagnosed with Pulmonary Lymphangitic Carcinomatosis
Özlem Şengören Dikis*, Seyhan Dulger, Tekin Yildiz and Safak Atahan
Department of Pulmonary Diseases, Health Sciences University, Bursa Yuksek Ihtisas Education and Research
Hospital, Bursa, Turkey
*Corresponding author: Pulmonary Diseases, Health Sciences University, Bursa Yuksek Ihtisas Education and Research Hospital, Bursa, Turkey
Published: 12 Oct, 2017
Cite this article as: Dikis ÖŞ, Dulger S, Yildiz T, Atahan S.
A Signet Ring Cell Gastric Carcinoma
Case Diagnosed with Pulmonary
Lymphangitic Carcinomatosis. Clin
Surg. 2017; 2: 1676.
Abstract
Background: Gastric carcinoma is one of the most common malignancies encountered in
adulthood. It may present with various clinical symptoms and signs. It commonly spreads
through haematogenous and lymphatic routes and rarely presents with pulmonary lymphangitic
carcinomatosis.
Case Presentation: A forty three years old male patient applied with the symptoms of progressive
dyspnea and cough. Although his radiological findings supported interstitial lung disease, there was
no response to antibiotics and corticosteroid treatment. Transbroncial biopsy indicated pulmonary
lymphangitic carcinomatosis originating from metastatic signet ring cell gastric adenocarcinoma.
Discussion: Pulmonary lymphangitic carcinomatosis is a rare manifestation of metastatic gastric
cancer. Cases usually present with various respiratory symptoms. The disease having a bad
prognosis is usually diagnosed using transbroncial or open lung biopsy. By keeping lymphangitic
carcinomatosis in mind, an underlying neoplasm should be suspected in patients having an
interstitial pattern in chest x-ray but no response to treatment.
Keywords: Pulmonary lymphangitic carcinomatosis; Transbronchial biopsy; Gastric
adenocarcinoma
Introduction
Pulmonary Lymphangitic Carcinomatosis (PLC) is a rare lung disease characterized with obstruction and diffuse infiltration of the pulmonary lymphatic system with tumor cells [1]. PLC, a very rare indication of gastric adenocarcinoma, is very rarely encountered in adults having metastatic gastric carcinoma [2]. PLC is recommended to be diagnosed with transbronchial or open lung biopsy [3]. Even if the diagnosis is made at an early stage, the prognosis is worse in PLC cases with signet ring cell gastric adenocarcinoma. Few cases having metastatic lymphangitic carcinomatosis have been reported in the literature [4-6]. We present a case hospitalized for interstitial lung disease or primary malignity and diagnosed with PLC originating from signet ring cell gastric adenocarcinoma using transbronchial fine needle aspiration biopsy (TBFNAB).
Case Presentation
A 43 -year-old male patient applied to the emergency clinic with the complaints of coughing
and progressive dyspnoea. His complaints started 5-6 months ago with a significant dyspnoea
during the previous 2 weeks. Our case was a smoker (20 package/year) with no significant issues in
his family history. He had no drug history except salbutamol inhaler (2 puff, 4-6 times a day).He had
no contact with pets or any material whose inhalation could be harmful to lung health. His baseline
examination findings were as follows: blood pressure 130/82 mmHg, heart rate 101 beats/min,
respiratory rate 25 breaths/min., fever 37.5 oC, and oxygen saturation (SaO2) 88% in room air and
97% in supplemental 2 lt/min. oxygen through nasal cannula. The case was significantly cashectic,
tachypneic, dyspneic, and mildly agitated and was using his auxiliary respiratory muscles. There
was no peripheral lymphadenopathy. Examination of the respiratory system revealed bibasilar fine
rales and wheezing. His chest x-ray revealed newly developed diffuse interstitial appearance which
was absent in his chest x-ray performed 5 months ago. His computed tomography showed diffuse
interstitial thickening, reticulonodular pattern and consolidation areas in the lower lobes and
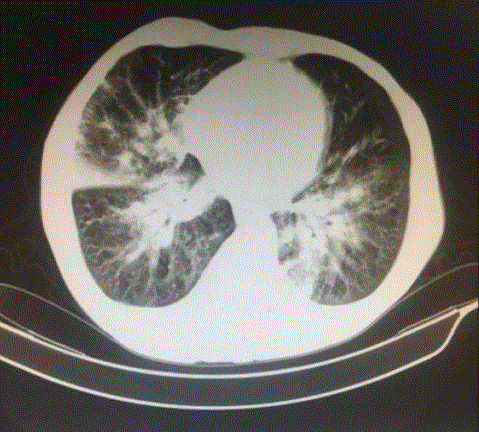
enlarged mediastinal lymph nodes (Figure 1).
The patient with acute respiratory failure was hospitalized. A wide spectrum non-specific
antibiotheraphy and 1 mg/day dose corticosteroid were initiated. There was no growth in blood, urine and sputum cultures. Upon clinical deterioration and progressive
respiratory failure, the patient was admitted to the intensive care
unit for ventilatory support. On the 2nd day of the intensive care unit,
TB-FNAB was performed using an endotracheal tube. Cytologic
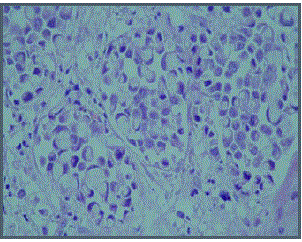
examination reported as signet ring cell stomach adenocarcinoma
with lung metastasis (Figure 2). The patient died due to progressive
respiratory failure and multiple organ failure on the 9th day of his
hospitalization.
Table 1
Figure 1
Figure 1
Computed tomography showed diffuse interstitial thickening,
reticulonodular pattern and consolidation areas in the lower lobes.
Figure 2
Figure 2
Atypical epithelial cells containing tumor-like signed ring cellcytoplasmic
mucin (H & E, x400).
Discussion
Lung metastases of malignant tumors are usually in the form of nodular lesions while PLC is observed only in 6-8% of them [2,7]. Spread of tumor cells into the pulmonary lymphatic system or adjacent interstitial tissue leads to an increase in bronchovascular bundles and thickening of septa. Desmoplastic reaction due to proliferation of neoplastic cells and lymphatic dilation by tumor secretions or edema liquids result in interstitial thickening. Spread to the lymphatic area within adjacent parenchyma and outside interstitium leads to a nodular pattern. While nearly all metastatic neoplasms such as lip cancer may lead to PLC, it is most frequently seen in breast, stomach, lung, pancreas and prostate cancers [3,8]. Usually, primary cancer symptoms are either masked with pulmonary symptoms or not present in PLC. Coughing and dyspnea are the most frequently encountered symptoms [9]. In our case, coughing and progressive dyspnea were more prominent than GIS complaints. Although 30- 50% of the PLC cases having a histologically proven disease have a normal chest x-ray, there exist various radiological changes in PLC Table 1. The baseline chest x-ray of the case at the emergency service revealed no symptoms except an increase in aeration while the radiologic evaluation on the 5th month showed a very significant diffuse interstitial pattern and mediastinal lymph adenopathies (Figure 1). Pulmonary sarcoidosis, interstitital lung disease, primary lung malignancy, hypersensitive pneumonitis, and lymphoma should be considered in the differential diagnosis of PLC. Nodular patterns can be seen in CT in these diseases. While the nodules are most often located in the upper lobes in sarcoidosis, they are mostly located in the lower lobes in PLC. Either transbronchial or open lung biopsy can be performed in the differential diagnosis of interstitial lung disease, primary lung malignancy and lymphoma. Our case was diagnosed using TBFNAB. According to the American Cancer Society, 5-year observed survival rates are 71% in stage 1A, 57% in stage 1B, 45% in stage 2A, 33% in stage 2B, 20% in stage 3A, 14% in stage 3B, 9% in stage 3C and 4.4% in stage 4 gastric adenocancer patients (10). Our case was diagnosed with PLC originating from signet ring cell adenocarcinoma died before any medical oncology consultation.
Conclusion
Pulmonary lymphangitic carcinomatosis should be considered and necessary evaluations should be done for a definite diagnosis in cases applying with the complaints of cough and progressive dyspnea and found to have bilateral pulmonary infiltrate without any known cause.
References
- Bruce DM, Heys SD, Eremin O. Lymphangitis carcinomatosa: a literature review. J R Coll Surg Edinb. 1996;41(1):7-13.
- Moubax K, Wuyts W, Vandecaveye V, Prenen H. Pulmonary lymphangitic carcinomatosis as a primary manifestation of gastric carcinoma in a young adult: a case report and review of the literature. BMC Res Notes. 2012;5:638.
- Thomas A, Lenox R. Pulmonary lymphangitic carcinomatosis as a primary manifestation of colon cancer in a young adult. CMAJ. 2008;179(4):338-40.
- Inage Y, Yamabe K, Yamamoto T, Sato Y, Ishikawa S, Onizuka M, et al. [Resection for pulmonary metastasis of gastrointestinal stromal tumor of the stomach at 10 years after gastrectomy; report of a case]. Kyobu Geka. 2002;55(10):907-11.
- Ozan E, Oztekin O, AlacacioÄŸlu A, AykaÅŸ A, Postaci H, Adibelli Z. Esophageal gastrointestinal stromal tumor with pulmonary and bone metastases. Diagn Interv Radiol. 2010;16(3):217-20.
- Kurashina K, Hosoya Y, Sakurai S, Endo S, Lefor A, Yasuda Y. Gastrointestinal stromal tumor of the stomach with extremely slow-growing hematogenous metastasis. Int J Clin Oncol. 2009;14(3):262-5.
- Richard S. Fraser, Nestor L. Muller, Neil C. Colman, P. D. Pare.). Fraser and Pare's Diagnosis of Diseases of the Chest (4 Volume set). 4th Edition.1999.
- Babu S, B S, M G, Salih S. A rare presentation of pulmonary lymphangitic carcinomatosis in cancer of lip: case report. World J Surg Oncol. 2011;9:77.
- HAUSER TE, STEER A. Lymphangitic carcinomatosis of the lungs: six case reports and a review of the literature. Ann Intern Med. 1951;34(4):881-98.
- Howlader N, Noone AM, Krapcho M, et al., editors. , eds. SEER cancer statistics review, 1975-2010. SEER Web site: SEER Cancer Statistics Review, 1975-2010. Bethesda, MD: National Cancer Institute; 2013.