Case Report
Caveats and Technique for the Reconstruction of Burn Contractures Using an Artificial Dermis in a Tropical Burns Centre
Wan-SzePek*, Janna-Vale Joethy and Si-Jack Chong
Department of Plastic, Reconstructive and Aesthetic Surgery, Singapore General Hospital, Singapore
*Corresponding author: Wan-SzePek, Department of Plastic, Reconstructive and Aesthetic Surgery Singapore General Hospital, Outram Road, 169608, Singapore
Published: 11 Oct, 2017
Cite this article as: Wan-SzePek, Joethy J-V, Chong
S-J. Caveats and Technique for the
Reconstruction of Burn Contractures
Using an Artificial Dermis in a Tropical
Burns Centre. Clin Surg. 2017; 2: 1674.
Abstract
There is a paucity of data on the use of dermal substitutes in a tropical burns centre, as its use was
previously complicated by a high infection rate in the Southeast Asian region, further hindered
by high costs. We hence describe our successful experience with a bi-layered artificial dermis
(PelnacTM) and thin split-thickness skin graft (STSG) in treating post-burns hypertrophic scars
and contractures when a lack of donor sites precluded reconstruction with traditional locoregional
flaps. A two-staged procedure comprising artificial dermis and STSG was used to reconstruct full
thickness wounds after excision of burns contractures over 11 sites (5 patients) - three wrists, three
necks, two ankles, one elbow, one chin, and one ear auricle. Time to skin grafting was 18.4 days,
with a 100% STSG take. One wound (9%) developed hypertrophic scarring around its border but
the rest healed with a pliable consistency and satisfactory cosmetic outcome. Full range of motion
was restored in treated scars located over joints. No infective complications were encountered. We
demonstrate that a bi-layered artificial dermis with STSG may be used reliably in a tropical burns
centre with no infective complications, whereby a consistent technique contributes greatly to its
success.
Keywords: Burn contracture; Artificial dermis, Scar, Reconstruction
Introduction
After initial wound healing is achieved, patients with extensive burns injuries often suffer
a second blow in the form of hypertrophic scarring, keloid formation and consequent scar
contractures. These scars frequently cause functional impairment necessitating surgical release
and reconstruction. Further more, scarring may cause physical discomfort in the form of pain or
itch, and negatively affect a patient’s emotional wellbeing and social functioning [1]. These late
complications pose a significant challenge in extensively burned patients who lack healthy donor
sites for traditional reconstructive methods such as local or regional flaps after scar release.
Dermal substitutes have yielded good results in acute and reconstructive burns wounds, of which
the use of Integra® is among the most widely reported [2-5]. Integra® however has been associated
with infective complications [4,6,7]. This, combined with a high product cost, has resulted in the
near absence of Integra and other dermal substitutes in the armamentarium of burns treatment in
Southeast Asia for the last decade.
PelnacTM (Gunze Corp., Osaka, Japan) is a dermal regeneration template first described in 1990
[8] as a modification of the “artificial skin” developed by Yannas et al. in the 1980s [9]. There is a
paucity of evidence for the use of PelnacTM in the existing literature, particularly in the setting of a
tropical burns centre.
We thus present our experience with the treatment of hypertrophic burns scars and scar
contractures using PelnacTM in patients where donor sites were limited. We describe a consistent
surgical technique which contributed to a successful outcome, with no infective complications in
our tropical burns center.
Materials and Methods
Patients
5 patients were enrolled in the study after informed consent was obtained. Medical records were
reviewed for patient demographics and details of the initial burn injury. Pre- and post- operative photographs were obtained. All patients underwent excision of
hypertrophic burns scars and reconstruction with a bi-layered
artificial dermal template (PelnacTM), followed by thin split thickness
skin grafting (STSG) from December 2015 to August 2016. Treatment
details that were considered were as follows: location of burns scar,
time to STSG, complications, post-operative scar consistency and
functional outcomes. Patients were followed up prospectively after
surgery.
Surgical technique
Areas of hypertrophic scarring were excised sharply down to
healthy underlying subcutaneous tissue to release all scar contractures.
Wounds were washed and meticulous hemostasis achieved. PelnacTM
was prepared according to the standard manufacturer’s guidelines.
Prior to application, PelnacTM was first immersed in sterile saline
solution, trimmed to the shape of the wound and then placed over
the wounds with the atelocollagen sponge layer in contact with the
wound surface. The peripheries of the PelnacTM material were secured
to the wound edges using vicrylrapideTM 5-0 absorbable sutures.
Quilting sutures were placed. Negative pressure woundtherapy
(NPWT) dressing was then applied over PelnacTM. This was replaced
with foam dressingsafter 7 days for better patient comfort and cost
management. An STSG (9:1000th of an inch) wasthen applied and
secured with either a NPWT dressing or a proflavin tie-over bolster
dressing. Wounds were inspected five days after STSG.
Figure 1
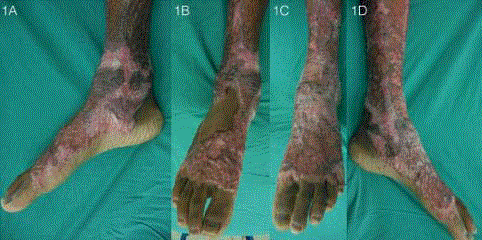
Figure 1
Pre-operative appearance of bilateral ankle scars (Patient 2).
Thick scars over the medial (A, D) and dorsal (B, C) aspects of bilateral ankles
resulted in contractures and limitation in ankle dorsiflexion, plantarflexion and
eversion. Walking was limited by pain and the restricted range of motion of
the ankle joint.
Table 1
Results
A total of 11 hypertrophic burns scars and contractures in 5
patients were treated at our tropicalburns centre using the consistent
method described above. The mean age of our patients was 38.6
years (range 26 to 55 years). The aetiology of their burn injuries
were flame burns in four patients and chemical burns in one patient.
Hypertrophic scars and scar contractures located over three wrists,
three necks, two ankles, one elbow, one chin, and one ear auricle
were treated. Patient and wound characteristics are detailed in Table
1. All scars had developed over previous areas of deep partial to
full thickness burns. Scars that extended over joints had previously
resulted in a reduction in a loss of range of motion and functional
impairment. Patients were followed up for a mean of 9.8 months
(range 4 to 25 months).
There were no instances of wound infection after PelnacTM
application. All wounds were ready for grafting with a single
application of PelnacTM. The presence of a well-vascularised neodermis
with a reddish hue was used as the indicator of a suitable bed for skin
grafting. The mean time to application of the split thickness skin graft
was 18.4 days (range 14-23) days, with a 100% rate of STSG take.
None of the wounds required further debridement or secondary
procedures.
At two months’ follow up, one (9.1%) of the wounds located
over a wrist had developed hypertrophic scarring around the border
of the skin graft. This however did not significantly affect the range
of motion of the underlying joint or the pliability of the graft itself.
The rest healed well and the resultant scars remained flat, soft and
pliable. The colour of the scars was also found to remain similar to
the site from which the STSG was harvested. At 6 months’ followup,
keloid scarring had partially recurred in the wound of a second
patient (9.1%). Secondary contraction had occurred in 2 wounds
(18.2%) although the consistency of the skin remained soft. The rest
of the wounds remained pliable. Where treated scars were located
over joints, the patients experienced no restriction of movement.
Case Presentation
Patient 2 was a 35-year-old man of Indian ethnicity involved
in an industrial accident, sustaining flame burns to 70% of his total
body surface area (TBSA) that were deep dermal to full thickness.
He required repeated burn wound excisions and skin cover with
autografts, allografts, culturedepithelial autografts and micrografts.
His prolonged recovery was complicated by pneumonia, acute kidney
injury and septic shock. Hypertrophic scarring developed over most
wounds but surgical release of those over the volar aspect of his right
wrist, left elbow and bilateral ankles figure 1 was planned as they were
causing functional restrictions. All affected areas were excised down
to healthy subcutaneous tissue before PelnacTM was applied (Figure 2).
When healthy neodermis had formed on the wound beds (Figure 3) a
thin split-thickness skin graft was applied. At four months’ follow-up,
all treated areas maintained a soft, pliable texture with a good range of
motion that allowed the patient to stand and walk without restriction
(Figure 4).
Patient 4 was a 55-year-old man who sustained chemical burns to
his face, trunk and upper limbs requiring excision and split thickness skin grafting. He developed hypertrophic scarring and meatal stenosis
of his right auricle that was causing hearing loss and was difficult to
clean. Thick scarringover the radial aspect of his left wrist, thumb
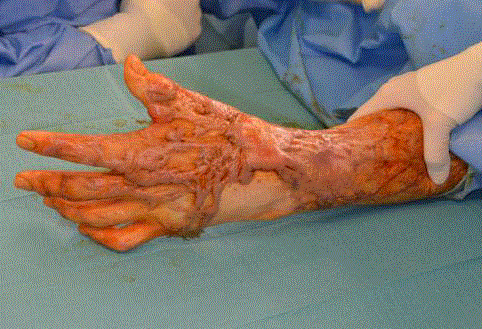
and radial snuffbox restricted wrist flexion and pincergrip (Figure 5).
The wrist scar was excised till adequate range of motion was achieved
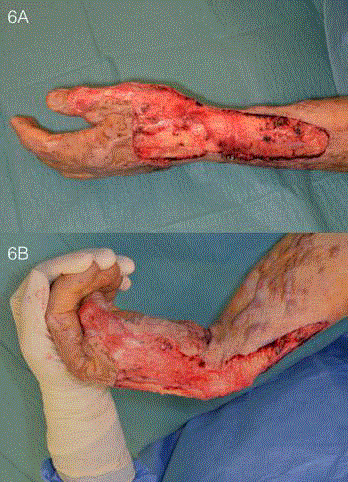
(Figure 6) and PelnacTM applied. Scar release and meatoplasty of his
right auricle were performed, after which the pre-auricular wound
was resurfaced with PelnacTM. He regained a good range of motionin
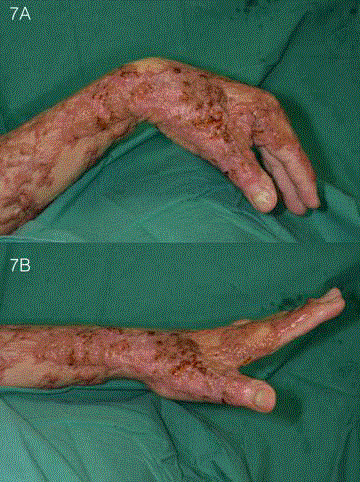
his left hand after the skin grafts had taken (Figure 7).
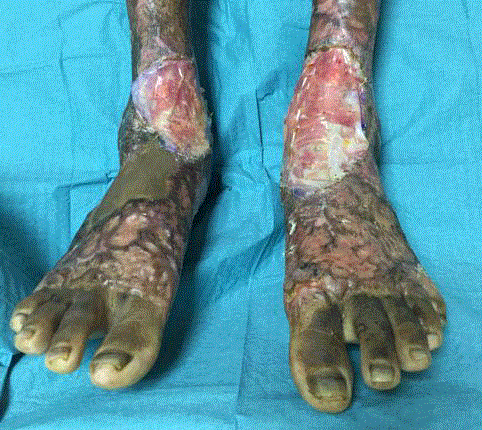
Figure 2
Figure 2
Wound appearance after excision of scars and application of
PelnacTM. PelnacTM was cut to fit the size of the defect following excision of
the offending scars. The edges were secured using vicrylrapideTM 5-0 sutures
and quilting sutures were applied.
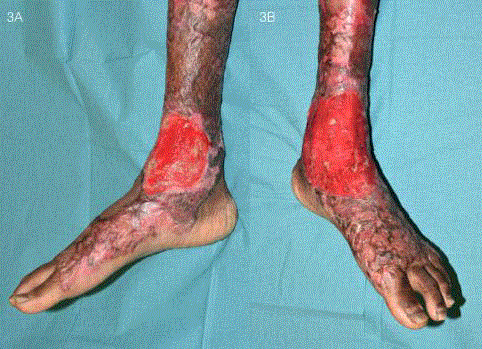
Figure 3
Figure 3
Wound appearance before application of STSG. A reddish hue
indicated the presence of a healthy, well-vascularised neodermis that would
allow for the STSG to take successfully.
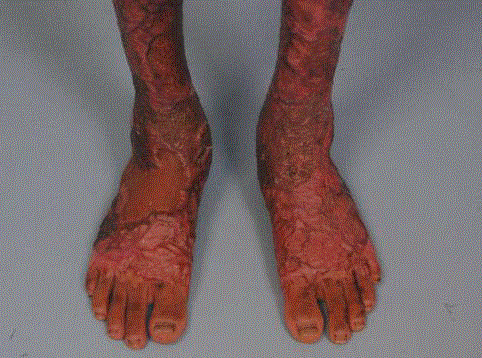
Figure 4
Figure 4
Post-operative wound appearance. The patient regained
a full range of motion in both his ankles, allowing him to stand and walk
comfortably. The treated areas remained soft and pliable, with the STSG
retaining the colour of the donor site.
Discussion
Hypertrophic scarring has been found to develop in up to 70% of
burns wounds [10] and poses a significant challenge to reconstruction,
especially in patients with extensive burns, as was the casein our
series with a mean TBSA involvement of 58.8% (range 46%-70%).
Our experience adds to the existing evidence that PelnacTM is an
efficacious alternative to conventional treatment methods of burns
scars and contractures. Recently, Widjaja et al. described their results
with PelnacTM in the treatment of complex wound defects [11]. To
our knowledge, ours is the first series to focus on the use of PelnacTM
in the treatment of hypertrophic burns scars and scar contractors in
a tropical burns centre.
PelnacTM consists of an inner atelocollagen layer derived from
porcine tendon, reinforced by anouter silicone film for mechanical
strength. After placement over the wound bed, the soft collagen
sponge conforms well, even on wounds to irregular surfaces. The were resurfaced with this dermis-like tissue has been shown to have
significantly less post-operative contracture compared to controls.
The use of atelocollagen instead of in soluble bovine collagen used
in Integra® minimises the antigenicity of the material, and as a
consequence the inflammatory response that can result in rejection
and failure [8,12].
We found that these beneficial properties of PelnacTM translated
to successful outcomes in our series. Asians, especially those with
darker pigmentation have a greater tendency for hypertrophic scar
and keloid formation compared to Caucasians [13], compounding
the challenge of managing patients with extensive burns in our
population. We were able to achieve fairly good results with PelnacTM
even in these patients, with most treated areas remaining soft and
pliable up to 25 months after skin graft placement. In our series, no
infective complications were encountered. This gives it a compelling
advantage over Integra®, which has been shown to have microbial
colonisation or infection as a common post-operative complication
[4]. A part from the intrinsic properties of PelnacTM, we believe that
the consistent use of negative pressure wound therapy (NPWT)
acted as an effective barrier to infection and reduced seroma and
hematoma accumulation which could contribute to a higher infection
rate. In addition, PelnacTM offers benefits comparable to that of other
dermal substitutes such as Integra® at more than half the cost at our
institution, making it an attractive alternative in our practise.
Apart from a reduction in infective complications, NPWT has
been associated with improved outcomes when used in combination
with dermal substitutes, including a higher elasticity of scars
compared to wounds treated with dermal substitutes alone [14].
Studies have also demonstrated an accelerated incorporation of
artificial dermis with NPWT [15-17]. The mean time to grafting in
our series was 18.4 days (14-23 days). We believe the consistent use
of NWPT in all our patients contributed to the absence of infection
and a shorter duration to STSG application. The use of NWPT also
allowed for patients to be managed in the outpatient setting without
the concerns of a breach in wound sterility or inadvertent shearing of
the artificial dermis from the wound bed.
Despite the many advantages of using PelnacTM in the treatment of
hypertrophic scars, we did notice a fair amount of wound contraction
in 18% (n=2) of the wounds, although the scarsremained flat and
soft with retention of the skin colour of the STSG donor site (patient
3–chinwound, patient 4-pre-auricular wound). This is consistent
with the results of Hur et al. [17] who demonstrated that wounds
resurfaced with artificial dermis (Matriderm) contracted to 85% of
the original graft size 6 months following grafting. Partial recurrence
of keloid scarring occurred in 9% (n=1) of the wounds in a patient
with a known tendency to keloid formation. The efficacy of PelnacTM
in these patients remains questionable from our study.
This study represents our initial experience with PelnacTM which
has shown to be a promising alternative in the management of burns
scar contractures in patients with a shortage of donor sites. It is limited
by small patient numbers and lack of randomisation, resulting in an
inability to draw definitive conclusions from this study alone. Larger,
prospective, long-term studies with formal outcome evaluation
using validated scar assessment tools are required to establish the
use of PelnacTM in the armamentarium of dermal substitutes for the
treatment of burns scars. How the results of PelnacTM compare with
other established dermal substitutes such as Integra® also remains to
be answered.
Figure 5
Figure 5
Pre-operative appearance of left wrist and thumb scars
(Patient 4). The thick, hypertrophic scars over the radial and dorsal aspect of
this patient’s wrist and thumb resulted in a limitation in wrist extension, flexion
and thumb pincer grip.
Figure 6
Figure 6
Wound appearance after contracture release. Scars that were
causing functional restriction were excised sharply down to healthy underlying
fat (A) and the adequacy of release was assessed intra-operatively before
the application of PelnacTM (B).
Figure 7
Figure 7
Post-operative appearance after healing of STSG. The patient
regained a good range flexion and extension in the wrist, as well as pincer
grip after the STSG had healed.
Conclusion
In conclusion, PelnacTM application followed by a thin STSG in a two-stage procedure is an efficacious and technically simple method of burns scar reconstruction when healthy donor sites are lacking. Infective complications may be avoided when it is used in combination with NPWT.
References
- Brown BC, McKenna SP, Siddhi K, McGrouther DA, Bayat A. The hidden cost of skin scars: quality of life after skin scarring. J Plast Reconstr Aesthet Surg. 2008;61:1049-58.
- Branski L K, Herndon DN, Pereira C, Mlcak RP, Celis MM, Lee JO, et al. Longitudinal assessment of Integra in primary burn management: A randomized pediatric clinical trial. Crit Care Med. 2007;35:2615-23.
- Bloemen MC, van Leeuwen MC, van Vucht NE, van Zuijlen PP, Middelkoop E. Dermal substitution in acute burns and reconstructive surgery: a 12-year follow-up. Plast Reconstr Surg. 2010;125(5):1450-9.
- Frame JD, Still J, Lakhel-LeCoadou A, Carstens MH, Lorenz C, Orlet H, et al. Use of Dermal Regeneration Template in Contracture Release Procedures: A Multicenter Evaluation. Plast Reconstr Surg. 2004;113(5):1330-8.
- Nguyen DQ, Potokar TS, Price P. An objective long-term evaluation of Integra (a dermal skin substitute) and split thickness skin grafts, in acute burns and reconstructive surgery. Burns. 2010;36(1):23-8.
- Peck MD, Kessler M, Meyer AA, Bonham Morris PA. A trial of the effectiveness of artificial dermis in the treatment of patients with burns greater than 45% total body surface area. J Trauma. 2002;52(5):971-8.
- Dantzer E, Braye FM. Reconstructive surgery using an artificial dermis (Integra): results with 39 grafts. Br J Plast Surg. 2001;54(8):659-64.
- Suzuki S, Matsuda K, Isshiki N,Tamada Y, Yoshioka K, Ikada Y. Clinical evaluation of a new bilayer “artificial skin”composed of collagen sponge and silicone layer. Br J Plast Surg. 1990;43:47-54.
- Yannas IV, Burke JF. Design of an artificial skin. I. Basic design principles. J BiomedMater Res. 1980;14(1):65-81.
- Bombaro KM, Engrav LH, Carrougher GJ, Wiechman SA, Faucher L, Costa BA, et al. What is the prevalence of hypertrophic scarring following burns? Burns. 2003;29:299-302.
- Widjaja W, Maitz P. Changes in the plasma cytokine and growth factor profile are associated with impaired healing in pediatric patients treated with INTEGRA® for reconstructive procedures. Eur J Plast Surg. 2016;39:125-13.
- Suzuki S, Matsuda K, Isshiki N, Tamada Y, Ikada Y. Experimental study of a newly developed bilayer artificial skin. Biomaterials.1990;11:356-60.
- Niessen FB, Spauwen PH, Schalkwijk J, Kon M. On the nature of hypertrophic scars and keloids: a review. Plast Reconstr Surg. 1999;104:1435-58.
- Bloemen MC, van der Wal MB, Verhaegen PD, Nieuwenhuis MK, van Baar ME, van Zuijlen PP, et al. Clinical effectiveness of dermal substitution in burns by topical negative pressure: a multicenter randomized controlled trial. Wound Repair Regen. 2012;20(6):797-805.
- Kim EK, Hong JP.Efficacy of negative pressure therapy to enhance take of 1-stage allodermis and a split-thickness graft. Ann Plast Surg. 2007;58:536-40.
- Molnar JA, DeFranzo AJ, Hadaegh A, Moryk- was MJ, Shen P, Argenta LC. Acceleration of Integra incorporation in complex tissue defects with subatmospheric pressure. Plast Reconstr Surg. 2004;113:1339-46.
- Eo S, Kim Y, Cho S. Vacuum-assisted closure improves the incorporation of artificial dermis in soft tissue defects: Terudermis(®) and Pelnac(®). Int Wound J. 2011;8(3):261-7.
- Hur GY, Seo DK, Lee JW. Contracture of skin graft in human burns: effect of artificial dermis. Burns. 2014;40(8):1497-503.