Research Article
Serum Visfatin Levels and Tissue Visfatin Expression in Patients with Breast Diseases
Hande Koksal1*, Nevzat Serdar Ugras2, Sevil Kurban3, Ismail Harmankaya2, Arif Atay1, Meryem
Ilkay Eren Karanis2 and Osman Dogru1
1Department of General Surgery, Konya Education and Research Hospital, Konya, Turkey
2Department of Pathology, Selcuk University Faculty of Medicine, Konya, Turkey
3Department of Biochemistry, Necmettin Erbakan University Faculty of Medicine, Konya, Turkey
*Corresponding author: Hande Koksal, Department of General Surgery, Konya Education and Research Hospital, Konya, Turkey
Published: 23 Aug, 2017
Cite this article as: Koksal H, Ugras NS, Kurban S,
Harmankaya I, Atay A, Karanis MIE.
Serum Visfatin Levels and Tissue
Visfatin Expression in Patients with
Breast Diseases. Clin Surg. 2017; 2:
1601.
Abstract
Objective: This study aims to evaluate if serum visfatin levels and tissue visfatin expression have an
importance in patients with either benign or malign breast masses.
Patients and Methods: The patients with complaints of breast masses and have diagnosis of either
invasive ductal carcinoma or fibroadenoma were enrolled. Serum visfatin levels were measured by
ELISA and tissue visfatin expressions were evaluated by immunhistochemistry.
Results: There were 29 patients with invasive ductal carcinoma and 14 patients with fibroadenoma.
Immunhistochemical visfatin staining was positive in all patients with fibroadenoma and invasive
carcinoma. When the patients with malignancy were evaluated separately, the patients with positive
ER and PR were seen to have higher levels of serum visfatin than the patients with negative hormone
receptors (p=0.023 and p=0.034 respectively). Serum visfatin levels were also high in patients with
lymph node involvement and T2 tumors in diameter (p=0.04 and p=0.037 respectively).
Discussion: Serum visfatin levels were high in patients with positive hormone receptors, nodal
involvement and T2 tumors. Visfatin staining was positive in both benign and malign diseases of
the breast; it is not possible to declare visfatin as a prognostic tool in breast cancer.
Keywords: Breast; Breast cancer; Visfatin
Introduction
Visfatin, an adipocyte hormone- also known as nicotinamide phophoribosyltransferase is a pre-B cell colony-enhancing factor. Hypoxia, inflammation and hyperglycemia upgrade; but insulin, somatostatin and statins down regulate the visfatin levels. Although it has been identified in many different tissue and organs, visceral adipose tissue preferentially expresses visfatin. It has many important well known functions in cell proliferation, biosynthesis of nicotinamide monoand dinucleotide and hypoglycemia [1]. It has many other functions such as promoting vascular smooth muscle cell maturation and inhibiting neutrophil apoptosis. The associations between visfatin and different cancer types also have been reported [2-7]. Worldwide, breast cancer is one of the most common cancers in women. The well-known risk factors of the breast cancer are high body mass index, sedentary lifestyle, increased alcohol consumption, hormone replacement therapy for menopause, exposure to radiation, early age at menarche, and giving birth late or not at all. Few studies showing the relationship between visfatin and breast cancer have been reported up to now [8-17]. The aim of this study is to determine whether serum visfatin levels and tissue visfatin expression have an importance or not, in patients with either benign or malign breast masses.
Material and Methods
Newly diagnosed 29 patients with invasive ductal carcinoma and 14 patients with fibroadenoma were enrolled in this study. The clinicopathologic features of the patients were obtained from their oncologic charts. The staging of breast cancer was determined according to the tumor-nodemetastasis (TNM) system. Visfatin (400-450) (Human) - antibody for immunohistochemistry (Phoenix Pharmaceuticals, Inc) was used for immunohistochemical staining. The detailed protocol was obtained from product description (Catalog No.: H-003-84). Finally, the slides were counterstained with hematoxylin and then examined under a light microscope by two independent pathologists. Only the cytolasmic staining in tumor cells (approximately 1,000 cells in 3–4 hpf) was calculated. The results for visfatin staining were scored according to the percentage of positively stained cells in 4 quantitative categories: score 1, 25% or less positive cells; score 2, 26% to 50% positive cells; score 3, 51% to 75% positive cells; and score 4, 76% or more positive cells [9]. Blood samples for visfatin from the patients with invasive ductal carcinoma and fibroadenoma were obtained in preoperative period. Serum visfatin levels were measured using a commercial enzyme immunoassay kit (Phoenix Pharmaceuticals, Belmont, CA) according to instructions of Manufacturer Company. The study was approved by the ethics committee of Selcuk University, and informed consent was obtained from each patient. For statistical assessment; The Statistical Package for Social Sciences (SPSS for Windows version 13.0, Chicago, IL, USA) program was used. Median values were used to analyze demographic characteristics. The parametric data is given as arithmetic means ± standard deviation (SD) and non-parametric data is given as median (minimum–maximum). For comparing categorical variables Pearson chi-square test was used and comparison between groups was assessed by Student's t test or Mann– Whitney U test (parametric data, non-parametric data respectively). In the statistical evaluations, a p value was regarded as significant if <0.05.
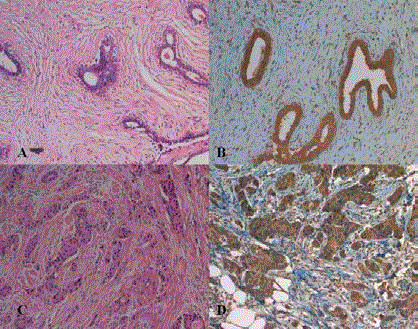
Figure 1
Figure 1
Immunhistochemical visfatin staining: A: Fibroadenom HE x 200 B:
Fibroadenom Visfatin x 200, C: İnvaziv duktal karsinom HE x 200, D: İnvaziv
duktal karsinom Visfatin x 200.
Results
There were 14 patients with fibroadenoma and 29 patients with invasive ductal carcinoma. The age of the patients with fibroadenoma were ranging from 15 to 53 years with a median age of 37 years while the age of the patients with invasive ductal carcinoma ranged from 27 to 87 years with a median age of 51 years. The diameters of the tumors were between 6 mm and 135 mm (median 25 mm) in patients with fibroadenoma and 15 mm to 60 mm (median 23 mm) in patients with invasive ductal carcinoma. In patients with malignancy, 24 of 29 patients had nodal involvement and 3 had Stage IV diseases at the time of diagnosis. Estrogen receptor was positive in 24 patients, progesterone receptor was positive in 24 patients and HER2 was positive in 12 patients. Immunhistochemical visfatin staining was positive in all patients with fibroadenoma and invasive carcinoma (Figure 1). The serum visfatin levels of the patients with fibroadenoma and invasive ductal carcinoma were 24.56 ± 3.13 and 25.03 ± 3.06 ng/ml, respectively. There were no differences between the patients with malign and benign breast masses in the means of serum visfatin levels (p=0.64). In patients with invasive ductal carcinoma, when the patients with malignancy were evaluated separately, serum visfatin levels were also high in patients with T2 tumors in diameter and lymph node involvement (p=0.038 and p=0.039 respectively). The patients with positive ER and PR were seen to have higher levels of serum visfatin than the patients with negative hormon receptors (p=0.033 and p=0.033 respectively), (Table 1). Also, the serum visfatin levels of 3 patients with T3 tumor were 22.06, 25.52 and 27.72 ng/ml. Both ER and PR negative patients’ serum visfatin levels (n: 3) were 24.31, 20.44 and 23.76 ng/ml. The serum visfatin level of the patient with triple negative disease was 24.59 ng/ml.
Table 1
Table 1
The serum visfatin levels according to the subgroups of the patients with
invasive ductal carcinoma.
Discussion
The most common malignancy in women especially in developed
countries is breast cancer, unfortunately its prevalence is increasing
rapidly, and it is the leading cause of cancer related death among
women. So, it becomes more and more critical to discover new
prognostic parameters; therefore new therapeutic targets; to overcome
this worldwide problematic disease. Epidemiologic studies have
demonstrated many well-established risk factors as age, hormoneassociated
reproductive factors like earlier age at menarche, later
age at menopause, older age at first birth, decreased parity and use
of hormone replacement therapy, increased body mass index, ratio
of the waist-to-hip circumference, family history of breast cancer,
lesions with atypical cells in mammary gland, and high breast density
on mammographic screening. The established prognostic factors
for breast cancer exist including tumor size, nodal involvement,
histologic grade, histologic type, and hormone receptor status
besides with obesity, insulin resistance and serum adipocytokine
levels. Recent studies indicate that obesity is both significant risk and
prognostic factor for breast cancer with poor prognosis. Adipocytes
produce adipocytokines; growth factors and cytokines; including
adiponectin, leptin, resistin and visfatin. The influences of obesity and
increased adiposity on the risk of breast cancer are partially explained
by the changes in adipocytokines secreted from adipose tissue and
from the epithelial tissue of breast tumors [18]. Visfatin regulates cell
cycle, apoptosis and angiogenesis in mammalian cells [9]. There have
been many studies showing the correlation between high expression
of visfatin and various cancers including central nervous system,
gastrointestinal and genitourinary systems [2-4]. The over expression
of visfatin in human vascular smooth muscle culture and endothelial
cells is positively correlated with acute oxidative stress, delayed senescence, and increased replication in life cycle [19,20]. Visfatin
is highly expressed in human breast cancer cells both in vitro and
in vivo and it increases the proliferation rate and DNA synthesis of
human breast cancer cells, suggesting that it may contribute to breast
cancer growth and moreover it is present in mammary glands during
lactation and milk [21]. Accumulating evidences suggest that visfatin
has some roles in mammary epithelial cells and gland, unfortunately
its probable role in breast cancer has not been understood clearly yet.
Obesity is a risk and also a prognostic factor for breast cancer and the
role of the visfatin on growth, apoptosis and angiogenesis were well
described. In this study, we want to evaluate if serum visfatin levels
and tissue visfatin expression have an importance in patients with
either benign or malign breast masses. In a study with colorectal cell
line HCT-116, Ghaemmaghami searched out that visfatin can affect
colorectal cancer cells in an autocrine or paracrine and probably
slightly in an endocrine manner. In a review by Shackelford et al.
[6], the relation between increased serum visfatin levels and several
types of human malignant tumors including colorectal, ovarian,
endometrial, breast, gastric, prostate, thyroid, melanoma and
astrocytomas were well established. Increased visfatin expression also
had been noticed in malignant lymphomas. It also seems to have a
role in hepatocellular carcinoma. Most studies documented increased
visfatin levels between benign and malign tissue while several of them
correlated visfatin expression with the changes in tumor behavior,
like Long et al. [22] who had founded visfatin expression 13 folds high
in gastric cancer tissue than in benign gastric tissue and concluded
that higher visfatin expression is correlated with deeper tumor
invasion, lymph node metastases, higher TNM stages and decreased
survival rates. Interestingly in some researches they have found that
visfatin expression also deals with chemotherapy resistance including
doxorubicin, paclitaxel and fluorouracil [23,24]. High visfatin levels in
breast cancer tissue were determined in patients with more malignant
cancer behaviour as ER and PR negativity, which is indicators of poor
prognosis and doubtless poor survival [11] and it, was shown to be an
independent predictor of disease free and overall survival. Despite the
absence of clear understanding of molecular mechanisms, the genetic
studies validate visfatin as a novel oncogene with an important role
in carcinogenesis [16]. These results may give chance to researchers
to gain speed in improving new therapies for triple negative breast
cancer by inhibiting the visfatin induced pathways and validating
visfatin as a new therapeutic target.
Zhang et al. [2] indicated that their results suggest that serum
visfatin level may serve as a biomarker of bladder cancer and an
independent prognostic marker of non muscle invasive bladder
cancer by their study in 2014. The similar correlation is also valid
for gastric cancers. A study by Guo-Wen Lu [3] again in 2014,
also showed that preoperative serum visfatin levels were higher
in patients with gastric carcinoma than otherwise healthy persons.
They suggested that visfatin levels were associated with invasion
depth, lymph node metastasis, distant metastasis, peritoneal
dissemination, tumor size and ultimately of course: stage. They
revealed serum visfatin level as an independent predictor for overall
survival and concluded that preoperative plasma visfatin level may
play a role as prognostic biomarker in survival of the patients with
gastric cancer. Contrarily; Skoczen et al. [5] showed that plasma
visfatin concentrations were higher after stem cell transplantation in
children with leukemia than before. The lower levels during complete
remission before the transplantation may due to myelosuppression
and immunsuppression so that the normalization of visfatin levels
after recovery of stem cell transplantation is related with process
of immune restoration. In our study, immunhistochemical visfatin
staining was positive in all patients either with fibroadenoma or
invasive carcinoma. In the patients with malignancy, the patients
with positive ER and PR, assumed to have better prognosis, were
determined to have higher levels of serum visfatin than the patients
with negative hormone receptors. Vice versa serum visfatin levels
were also high in patients with lymph node involvement and T2
tumors in diameter that indicates poor prognosis.
Conclusion
Visfatin is over expressed in several human malignancies where it is often associated with poor prognosis. Additionally some studies indicate visfatin as a novel oncogene and therapeutic target. Possibly it seems to play an important role both in carcinogenesis and treatment. However there are many poorly understood molecular mechanisms waiting to be solved. Further studies are needed to clear metabolism related carcinogenesis from the aspect of visfatin.
References
- Adeghate E. Visfatin: structure, function and relation to diabetes mellitus and other dysfunctions. Curr Med Chem. 2008;15(18):1851-62.
- Zhang K, Zhou B, Zhang P, Zhang Z, Chen P, Pu Y, et al. Prognostic value of serum nicotinamide phosphoribosyltransferase in patients with bladder cancer. Croat Med J. 2014;55(5):507-13.
- Lu GW, Wang QJ, Xia MM, Qian J. Elevated plasma visfatin levels correlate with poor prognosis of gastric cancer patients. Peptides. 2014;58:60-4.
- Ghaemmaghami S, Mohaddes SM, Hedayati M, Gorgian Mohammadi M, Dehbashi G. Resistin and Visfatin Expression in HCT-116 Colorectal Cancer Cell Line. Int J Mol Cell Med. 2013;2(3):143-50.
- Skoczen S, Tomasik PJ, Gozdzik J, Fijorek K, Krasowska-Kwiecien A, Wiecha O, et al. Visfatin concentrations in children with leukemia before and after stem cell transplantation. Exp Hematol. 2014;42(4):252-60.
- Shackelford RE, Mayhall K, Maxwell NM, Kandil E, Coppola D. Nicotinamide phosphoribosyltransferase in malignancy: a review. Genes Cancer. 2013;4(11-12):447-56.
- Chen M, Wang Y, Li Y, Zhao L, Ye S, Wang S, et al. Association of plasma visfatin with risk of colorectal cancer: An observational study of Chinese patients. Asia Pac J Clin Oncol. 2016;12(1):e65-74.
- Kim JG, Kim EO, Jeong BR, Min YJ, Park JW, Kim ES, et al. Visfatin stimulates proliferation of MCF-7 human breast cancer cells. Mol Cells. 2010;30(4):341-5.
- Lee YC, Yang YH, Su JH, Chang HL, Hou MF, Yuan SS. High visfatin expression in breast cancer tissue is associated with poor survival. Cancer Epidemiol Biomarkers Prev. 2011;20(9):1892-901.
- Kim SR, Park HJ, Bae YH, Ahn SC, Wee HJ, Yun I, et al. Curcumin down-regulates visfatin expression and inhibits breast cancer cell invasion. Endocrinology. 2012;153(2):554-63.
- Dalamaga M, Archondakis S, Sotiropoulos G, Karmaniolas K, Pelekanos N, Papadavid E, et al. Could serum visfatin be a potential biomarker for postmenopausal breast cancer? Maturitas. 2012;71(3):301-8.
- Bajrami I, Kigozi A, Van Weverwijk A, Brough R, Frankum J, Lord CJ, et al. Synthetic lethality of PARP and NAMPT inhibition in triple-negative breast cancer cells. EMBO Mol Med. 2012;4(10):1087-96.
- Santidrian AF, Matsuno-Yagi A, Ritland M, Seo BB, LeBoeuf SE, Gay LJ, et al. Mitochondrial complex I activity and NAD+/NADH balance regulate breast cancer progression. J Clin Invest. 2013;123(3):1068-81.
- Li XY, Tang SH, Zhou XC, Ye YH, Xu XQ, Li RZ. Preoperative serum visfatin levels and prognosis of breast cancer among Chinese women. Peptides. 2014;51:86-90.
- Park HJ, Kim SR, Kim SS, Wee HJ, Bae MK, Ryu MH, et al. Visfatin promotes cell and tumor growth by upregulating Notch1 in breast cancer. Oncotarget. 2014;5(13):5087-99.
- Zhou T, Wang T, Garcia JG. Expression of nicotinamide phosphoribosyltransferase-influenced genes predicts recurrence-free survival in lung and breast cancers. Sci Rep. 2014;4:6107.
- Santidrian AF, LeBoeuf SE, Wold ED, Ritland M, Forsyth JS, Felding BH. Nicotinamide phosphoribosyltransferase can affect metastatic activity and cell adhesive functions by regulating integrins in breast cancer. DNA Repair (Amst). 2014;23:79-87.
- Vona-Davis L, Rose DP. Adipokines as endocrine, paracrine, and autocrine factors in breast cancer risk and progression. Endocr Relat Cancer. 2007;14(2):189-206.
- van der Veer E, Ho C, O'Neil C, Barbosa N, Scott R, Cregan SP, et al. Extension of human cell lifespan by nicotinamide phosphoribosyltransferase. J Biol Chem. 2007;282(15):10841-5.
- Borradaile NM, Pickering JG. Nicotinamide phosphoribosyltransferase imparts human endothelial cells with extended replicative lifespan and enhanced angiogenic capacity in a high glucose environment. Aging Cell. 2009;8(2):100-12.
- Yonezawa T, Haga S, Kobayashi Y, Takahashi T, Obara Y. Visfatin is present in bovine mammary epithelial cells, lactating mammary gland and milk, and its expression is regulated by cAMP pathway. FEBS Lett. 2006;580(28-29):6635-43.
- Long HL, Che XM, Bi TQ, Li HJ, Liu JS, Li DW. The expression of nicotinamide phosphoribosyl transferase and vascular endothelial growth factor-A in gastric carcinoma and their clinical significance. Zhonghua Wai Ke Za Zhi. 2012;50(9):839-42.
- Folgueira MA, Carraro DM, Brentani H, Patrão DF, Barbosa EM, Netto MM, et al. Gene expression profile associated with response to doxorubicin-based therapy in breast cancer. Clin Cancer Res. 2005;11(20):7434-43.
- Bi TQ, Che XM, Liao XH, Zhang DJ, Long HL, Li HJ, et al. Overexpression of Nampt in gastric cancer and chemopotentiating effects of the Nampt inhibitor FK866 in combination with fluorouracil. Oncol Rep. 2011;26(5):1251-7.