Research Article
Detection of Anal Dysplasia by Chromoendoscopy with Narrow Band Imaging and Acetic Acid (NBIA) in 182 Patients
Michelle D Inkster and James S Wu*
Digestive Diseases and Surgery Institute, Cleveland Clinic, Cleveland, Ohio, USA
*Corresponding author: James S Wu, Digestive Diseases and Surgery Institute, Cleveland Clinic, 6770 Mayfield Road, Mayfield Heights, OH 44124, USA
Published: 07 Aug, 2017
Cite this article as: Inkster MD, Wu JS. Detection of Anal
Dysplasia by Chromoendoscopy with
Narrow Band Imaging and Acetic Acid
(NBIA) in 182 Patients. Clin Surg. 2017;
2: 1583.
Abstract
Anal squamous intraepithelial lesion (SIL) precedes the development of anal squamous cell
carcinoma (SCC). Detection of SIL is facilitated by chromoendoscopy with narrow band imaging
and acetic acid (NBIA). Between 2011 and 2017, 182 patients with abnormal anal cytology
underwent NBIA to identify and ablate SIL. The majority of patients were HIV positive men. Other
associated diagnoses included urogynecologic dysplasia, inflammatory bowel disease and organ
transplantation. The histopathologic diagnosis of SIL/SCC was found in 65% of cases overall (52%
for cytology showing atypical cells, 81% for cytology showing low-grade SIL and 73% for cytology
showing high-grade SIL). Illustrative examples showing the endoscopic appearance of LSIL, HSIL,
and SCC are shown. The procedure can be done in any facility where endoscopy is practiced.
Keywords: Squamous intraepithelial neoplasia; Chromoendoscopy; Narrow band imaging;
Human papillomavirus; Human immunodeficiency virus; Organ transplantation; Inflammatory
bowel disease
Introduction
Anal squamous cell carcinoma (SCC) and its precursor lesion, anal squamous intraepithelial lesion (SIL) are human papillomavirus-related neoplasms [1]. Risk factors include immunosuppression, human immunodeficiency virus (HIV) infection, urogynecologic dysplasia, inflammatory bowel disease and solid organ transplantation [2,3]. Detection of premalignant anal lesions using an endoscope and acetic acid was described in 1989 by Scholefield et al. [4] More recently, the application of fiberoptic flexible endoscopy with narrow band imaging (NBI) using retroflexion/rectal insufflation and en face views with a transparent hood has been reported [5-9]. We have previously described the application of fiberoptic chromoendoscopy with NBI and acetic acid (NBIA) in 60 consecutive patients who had abnormal anal cytology [10]. We now describe the findings of NBIA done in 182 consecutive patients with abnormal anal cytology.
Methods
In 2011, we obtained approval from the Cleveland Clinic Institutional Review Board to identify and evaluate all patients within our Health System who had received the diagnosis of anal intraepithelial neoplasia. This was a non-randomized prospective study. Patients were interviewed. A history and physical examination were obtained with attention focused on risk factors for anal dysplasia, anal examination and white light anoscopy. Cytology was reported according to the Bethesda System [11]. Those with abnormal anal cytology [12,13] were offered NBIA. Informed consent for endoscopic examination of the anorectum was obtained. The procedure was performed in an endoscopy unit using a high-definition gastroscope (Olympus GHF180). Pre-procedural bowel preparation was administered. Blood pressure, pulse oximetry and EKG/heart rate were monitored during and after the procedure. Intravenous moderate sedation with Fentanyl or Meperidine plus Midazolam was administered. Topical intra-anal 2% Lidocaine gel was applied for local anesthesia. The perianal skin was examined. The gastroscope was introduced through the anus and passed up to the sigmoid colon. The anal transitional zone (ATZ) and anal canal were inspected using both en face and retroflexed views (Figure 1 and 2) with white light and NBI before and after treatment of the anal transitional zone and anoderm with 3% acetic acid. Evaluation of the anal canal was done by passing the gastroscope through a self-lighted beveled Anospec™ anoscope. Lesions showing acetowhite changes and/or abnormal vascular features including punctuation or mosaicism [14] were biopsied and ablated. Hot biopsy forceps were preferred for small lesions. A Gold ProbeTM Electrohemostasis catheter (Boston Scientific) set at 30 W soft cut was used to ablate large and irregular lesions. Photo documentation was preserved as part of the permanent electronic medical record (Provation™). Patients were recovered and discharged home. When surgery was required, NBIA was done in the operating room. Histopathology was reported according to the lower anogenital squamous terminology standardization (LAST) project for HPV-associated lesions [15]. Anal cytology readings of atypical cells of uncertain significance (ASCUS), low-grade squamous intraepithelial lesion (LSIL) or high-grade intraepithelial lesion (HSIL) were considered abnormal. Biopsy readings of LSIL, HSIL and SCC were recorded.
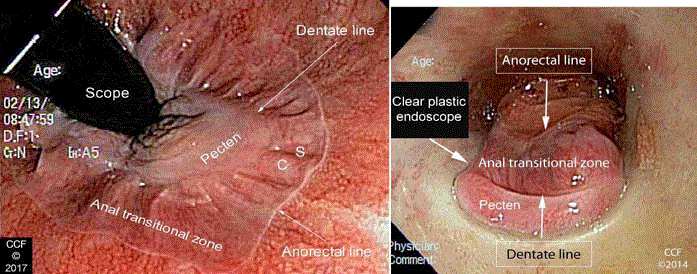
Figure 1
Figure 1a
Figure 1b
Retroflexion with rectal insufflation shows the anorectal line, the ATZ, the dentate line and the pecten.
En face view of the anal canal through a clear plastic anoscope shows the anorectal line, the ATZ, the dentate line and the pecten. The anoscope
increases the diameter of the canal facilitating magnified endoscopic visualization of the ATZ and its borders.
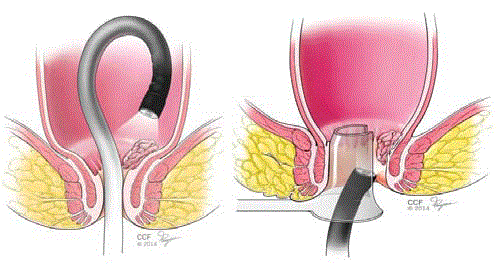
Figure 2a
Figure2b
Figure 2a
Figure 2b
Illustration of a lesion located in the ATZ seen with retroflexion
and rectal insufflation.
Illustration of en face view of an anal canal lesion seen through
a lighted anoscope.
Figure 3
Figure 3
Anal LSIL (arrows) seen through a beveled lighted anoscope by
chromoendoscopy with NBI and acetic acid (NBIA). The lesion is raised and
shows diffuse punctuation.
Figure 4a
Figure 4b
Figure 4a
Figure 4b
White light inspection of the ATZ with retroflexion shows a raised
lesion (arrow) close to the flexible endoscope.
Multiple lesions (arrows) not visible with white light are seen on
retroflexed view of the ATZ after treatment with acetic acid and illumination
with NBI. Biopsies showed HSIL.
Figure 5
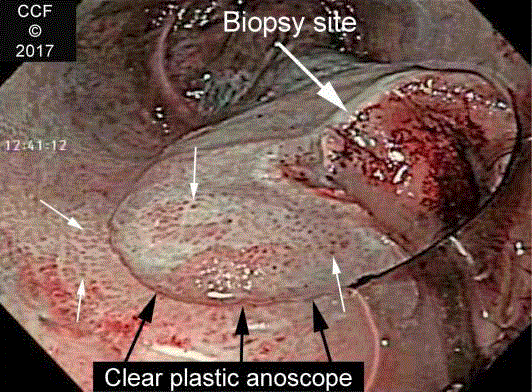
Figure 5
NBIA in a renal transplant patient. En face view of the anal canal
with NBI after treatment with acetic acid shows diffuse punctuation (small
arrows). The changes are seen directly and also through the clear plastic of
the anoscope (edge marked with black arrows). The biopsy site is shown.
Results
Between 2011 and 2017, 182 individuals were referred for
management of abnormal anal cytology. Patient demographics are
shown in Table 1. The majority were HIV positive men who have
sex with men (MSM). Others had risk factors for anal dysplasia that
included urogynecologic dysplasia, inflammatory bowel disease or
organ transplantation. Detection of anal SIL or SCC by NBIA in these 182 patients is summarized in Table 2. The results are for the
first NBIA examination performed on each patient. Biopsies showed
SIL/SCC in 65% of patients overall. The pre-procedural diagnosis of
ASCUS yielded a pathologic finding of SIL in 52% (35% LSIL, 17%
HSIL). The pre-procedural diagnosis of LSIL yielded a pathologic
diagnosis of SIL/SCC in 81% (57% SIL, 23% HSIL, SCC 1%). The
pre-procedural diagnosis of HSIL showed SIL in 73% (33% LSIL,
40% HSIL). There were no post procedural admissions to hospital
or emergency room visits for these patients. Illustrative examples of
NBIA follow.
Example 1: En face view of the anal canal after treatment with
acetic acid showed diffuse punctuation when illuminated with NBI
(Figure 3). Biopsy of this area showed LSIL.
Example 2: Retroflexed view of the anorectum with white light
shows a single discrete raised lesion (Figure 4a). Re-examination
with NBI after treatment with acetic acid (NBIA) revealed a complex
collection of slightly raised lesions with enhanced vascularity (Figure
4b). Biopsies of this area showed HSIL. All lesions were ablated with
hot biopsy forceps.
Example 3: Chromoendoscopy (NBI/acetic acid) in a renal
transplant patient, Figure 5, showed diffuse punctuation. Biopsy
showed HSIL.
Example 4: An HIV positive HPV positive male with abnormal
anal cytology underwent NBIA. Three discrete lesions were identified.
Targeted biopsies showed both LSIL and HSIL (Figure 6).
Example 5: An HIV positive male was found to have an anterior
raised acetowhite lesion with punctuation in the ATZ (Figure 7a). The
lesion was biopsied and ablated using hot biopsy forceps (Figure 7b).
Example 6: NBIA with en face view of the anal canal shows a flat
anal lesion through the wall of the clear plastic anoscope (Figure 8).
Biopsy showed HSIL. The entire lesion was excised. Final pathology
showed SCC. Treatment by combined chemoradiation was initiated.
Example 7: A patient presented for assessment of anorectal bleeding.16 Flexible sigmoidoscopy with retroflexed and en face views
of the ATZ illuminated with NBI showed an irregular, friable mass
(Figures 9a and 9b). Biopsies showed SCC. Chemoradiation was
initiated.
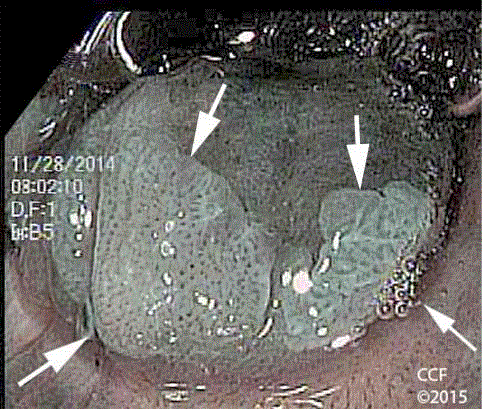
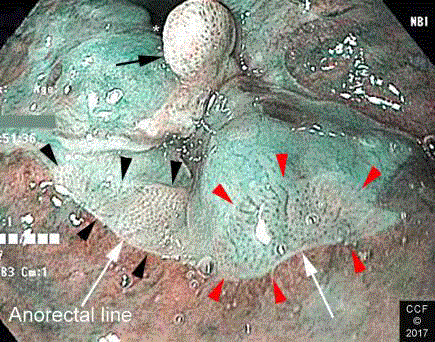
Figure 6
Figure 6
NBIA with rectal retroflexion shows three discrete lesions in the
anterior ATZ. Biopsies of the lesions labelled with the black arrow and arrow
heads showed LSIL. Biopsy of the lesion labelled with red arrowheads
showed HSIL. The endoscope is identified with an asterisk. The two lesions
marked with arrowheads are located in the ATZ at the anorectal line (white
arrows).
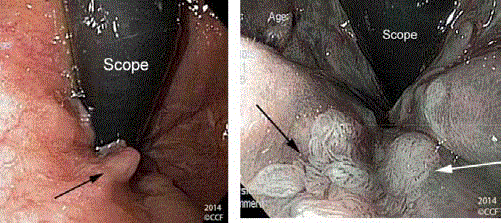
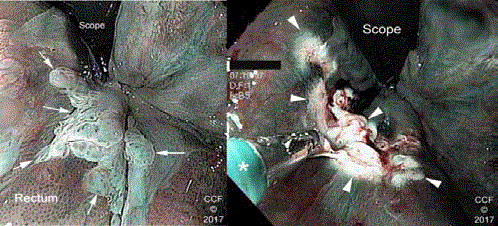
Figure 7a
Figure 7b
Figure 7a
Figure 7b
The serpiginous borders of a raised acetowhite anterior ATZ
lesion are identified with white arrows.
The lesion (arrow heads) has been ablated with hot biopsy
forceps (asterisk). Pathology showed LSIL.
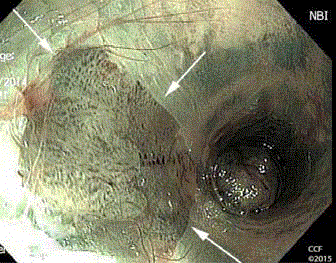
Figure 8
Figure 8
NBIA with en face view through the wall of a clear plastic anoscope
shows a discrete anal canal lesion with punctuation. The lesion proved to
be SCC.
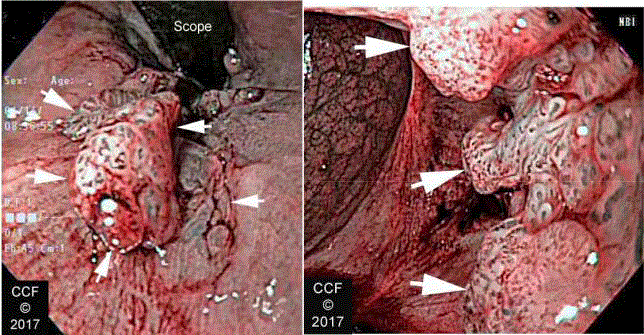
Figure 9
Figure 9
ATZ SCC seen with NBI with (a) retroflexed and (b) en face views.
Arrows delineate the lesion. The hypervascular character of the cancer is
enhanced by NBI.
Table 1
Table 2
Table 2
Chromoendoscopy (NBIA) in search of anal dysplasia in 182 individuals with abnormal anal cytology Histopathology.
Discussion
The majority of anal cancer cases are squamous cell carcinoma
[17]. Nigro credits Gabriel for suggesting that cancer of the anal
region be divided into two groups according to the site of origin
[18,19]. Anal margin cancer, developing in the perianal skin, is
identified by direct inspection. Anal canal cancer originates in the
cloacogenic zone, the tissue immediately proximal to the area of the
dentate line [18]. Fenger defines this area as the anal transitional
zone, “the zone interspersed between uninterrupted colorectal
mucosa above and uninterrupted squamous epithelium below [20]”.
Between the dentate line and the skin is the pecten, described by
Stroud as a stratified epithelium that “has few or no openings or
sweat glands” [21]. Visualization of the ATZ, anal canal pecten and
neighboring anatomic landmarks are necessary for the identification
of lesions found within their boundaries. This paper describes our
experience with chromoendosopic (NBIA) detection of anal dysplasia
in 182 consecutive patients. The following features of this technique
facilitate the detection of anal dysplasia.
• Retroflexion with rectal air insufflation provides a
circumferential view of the effaced ATZ and pecten.
• Examination through a beveled self-lighted anoscope
provides clear en face views of the ATZ and pecten.
• Chromoendoscopy with NBI and acetic acid (NBIA)
facilitates identification of SIL.
• Lesions can be biopsied and ablated endoscopically using
standard equipment.
• Photodocumention is preserved as part of the permanent
electronic medical record.
The clinical correlation between anal cytology, chromoendoscopic
findings and histopathology is not perfect. Long-term follow-up
is needed to determine limitations of NBIA with regard to lesion
detection and elimination.
Summary
We have described chromoendoscopy using NBI and acetic acid (NBIA) for the detection, biopsy and ablation of anal dysplasia in the ATZ and anal canal in 182 consecutive patients with abnormal anal cytology who each underwent a single examination. Overall, SIL /SCC were found in 65% of patients. Of the 97 patients with the pre-procedure cytology of ASCUS, 52% had SIL on biopsy. For the 70 patients with LSIL on cytology, SIL/SCC was found in 81%; carcinoma was detected by biopsy in 1.4%. When cytology showed HSIL, dysplasia was found on biopsy in 73%. Although the majority of patients in this report were HIV positive males, the referral of patients with urogynecologic dysplasia, inflammatory bowel disease and organ transplantation reflects an increasing awareness of the SIL in other high-risk groups. Providers who perform esophagogastroduodenoscopy and/or colonoscopy routinely should be able to perform anal chromoendoscopy to detect anal SIL since the techniques described here are already part of routine gastrointestinal endoscopic practice.
References
- Muñoz N, Castellsagué X, de González AB, Gissmann L. Chapter 1: HPV in the etiology of human cancer. Vaccine. 2006;24(3):S3/1-10.
- Suneson KG, Nørgaard M, Thorlacius-Ussing O, Laurberg S. Immunosuppressive disorders and risk of anal squamous cell carcinoma: a nationwide cohort study in Denmark, 1978-2005. Int J Cancer. 2010;127(3):675-84.
- Inkster MD, Szmulowicz UM, Wiland HO, Wu JS. Anal condyloma and anal dysplasia. In Anorectal Disease, Zutshi M (ed), Springer. 2015;8:189-209.
- Scholefield JH, Sonnex C, Talbot IC, Palmer JG, Whatrup C, Mindel A, et al. Anal and cervical intraepithelial neoplasia: possible parallel. Lancet.1989;334(8666):765-9.
- Chou YP, Saito Y, Matsuda T, Nakajima T, Mashimo Y, Moriya Y, et al. Novel diagnostic methods for early-stage squamous cell carcinoma of the anal canal successfully resected by endoscopic submucosal dissection. Endoscopy. 2009;41:E283-5.
- Oono Y, Fu K, Nakamura H, Iriguchi Y, Yamamura A, Kishi D, et al. Narrowband imaging colonoscopy with a transparent hood for diagnosis of a squamous cell carcinoma in situ in the anal canal. Endoscopy. 2010;42(2):E183-4.
- Tanaka E, Noguchi T, Nagai K, Akashi Y, Kawahara K, Shimada T. Morphology of the epithelium of the lower rectum and the anal canal in the adult human. Med Mol Morphol. 2012;45(2):72-9.
- Morisaki T, Isomoto H, Akazawa Y, Yamaguchi N, Ohnita K, Takeshima F, et al. Beneficial use of magnifying endoscopy with narrow-band imaging for diagnosing a patient with squamous cell carcinoma of the anal canal. Dig Endosc. 2012;24(1):42-5.
- Horimatsu T, Miyamoto S, Ezoe Y, Muto M, Yoshizawa A, Sakai Y. Gastrointestinal: Case of early-stage squamous cell carcinoma of the anal canal diagnosed using narrow-band imaging system with magnification. J Gastro Hepatol. 2012; 27(8):1406.
- Inkster MD, Wiland HO, Wu JS. Detection of anal dysplasia is enhanced by narrow band imaging and acetic acid. Colorectal Dis. 2016;18(1):17-21.
- Solomon D, Nayar R, editors. The Bethesda System for Reporting Cervical Cytology. 2nd Ed. New York: Springer-Verlag. 2004.
- Sherman ME, Friedman HB, Busseniers AE, Kelly WF, Carner TC, Saah AJ. Cytologic diagnosis of anal intraepithelial neoplasia using smears and cytyc thin-preps. Mod Pathol. 1995;8(3):270-4.
- Friedlander MA, Stier E, Lin O. Anorectal cytology as a screening tool for anal squamous lesions: cytologic, anoscopic, and histologic correlation. Cancer. 2004;102(1):19-26.
- Darragh TM, Berry JM, Jay N, Palefsky JM. The anal canal and perianus; HPV-related disease. In: Modern Colposcopy, Textbook and Atlas, 3rd Ed. Mayeaux EJ Jr, Cox JT, editors. ASCCP. Williams and Wilkins, Philadelphia. 2012;484-538.
- Darragh TM, Colgan TJ, Cox JT, Heller DS, Henry MR, Luff RD, et al. The lower anogenital squamous terminology standardization project for HPV-associated lesions: Background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. Arch Pathol Lab Med. 2012;136(10):1266-97.
- This patient underwent chromoendoscopy without anal cytology and is not included among the 182 patients described above. Acetic acid was not needed to see the cancer.
- National Comprehensive Cancer Network (NCCN). NCCN clinical practice guidelines in oncology. Anal carcinoma Version 2.2017, MS-2. NCCN.org.
- Nigro ND. Multidisciplinary management of cancer of the anus. World J Surg. 1987;11(4):446-51.
- Gabriel WB. Squamous-cell Carcinoma of the Anus and Anal Canal: An Analysis of 55 Cases: (Section of Proctology). Proc R Soc Med. 1941;34(3):139-60.
- Fenger C. The anal transitional zone. Acta Pathol Microbiol Immunol Scand Suppl. 1987;289:1-42.
- Stroud BB. I. On the Anatomy of the Anus. Ann Surg. 1896;24(1):1-15.