Research Article
A Novel Technique for Composite Cultured Epithelial Autograft in a Patient with Extensive Burn Wounds: A Case Report
Wayne Kleintjes*, Grant Thomas, Brogan Salence Stephan Thaele, Nico Stevenson and Brian
Warren
Department of Surgery, Western Cape Provincial Tertiary Adult Burn Unit, Tygerberg Hospital and Stellenbosch
University, South Africa
*Corresponding author: Wayne G. Kleintjes, Department of Plastic and Reconstructive Surgery, Tygerberg Hospital, Suite 10, 1st Floor, Bellville Melomed Hospital, c/o Voortrekker & AJ, West Street, Bellville, 7530, South Africa
Published: 04 Aug, 2017
Cite this article as: Kleintjes W, Thomas G, Thaele BSS,
Stevenson N, Warren B. A Novel
Technique for Composite Cultured
Epithelial Autograft in a Patient with
Extensive Burn Wounds: A Case
Report. Clin Surg. 2017; 2: 1579.
Abstract
Objective: Our goal was to develop a simple, safe and affordable technique for cultured epithelial
autografts (CEA) without delay to save our patients life. The patient was 16 years old and suffered
45% burns, and his condition was steadily deteriorating for three months in the intensive care unit.
As a last resort we decided culture his skin with minimal resources.
Method: A skin biopsy of 7x3 cm was taken from the patient’s hip area. The next day the patient’s
wounds were covered with a xenograft, EZ Derm®. The cells cultured were nourished on a daily
basis with fresh autogenous platelet-rich plasma (PRP). The cells were grown on Cutimed Sorbact
dressing pads® in a paediatric incubator. Some keratinocytes grown in sterile specimen bottles and
PRP were explanted on day 7 onto new Cutimed Sorbact Dressing Pads® in a second incubator. At
two weeks, EZ Derm® superficial layer was removed and the CEA transplanted with the Sorbact pads
directly onto the prepared wound beds. Some cells were squirted onto the wounds and dressed with
Sorbact Dressing Pads®.
Results: Within two weeks the whole Cutimed Sorbact pad (green) was almost overgrown with
epithelial cells. An initial overall successful graft take was estimated at 75%. The patient’s final graft
take at three weeks was 78%. He was discharged from the ICU two weeks after CEA application and
took 2 years to build up strength to walk alone again.
Conclusion: The technique has proven to be effective for CEA, biologically safe, completely
autogenous, and it was life-saving for our patient. The overall graft take rate of 78% is better than
the largest series in the literature (72, 7%) and may indicate an improvement in CEA technique. The
biggest advantage is the cost-effectiveness, which implicates the possibility for saving serious burn
victims on a very large scale.
Keywords: Burn wounds; Epithelial autograft; Cutimed Sorbact
Background
A 16 year old male patient was admitted to Tygerberg Hospital on 19 July 2014, via a peripheral hospital, with 46% total body surface area (TBSA) deep partial and full thickness burns to his head, neck, chest, back, right arm and right hand, left hand and left thigh, sustained in a domestic fire. Ten% was added and included for an inhalation burn. He was received intubated and ventilated from the referring facility. After initial dressing and fluid resuscitation, he underwent procedures on days 3, 12 and 25 post-admission, culminating in partial thickness skin grafting of his burn wounds. Amputation of all fingers of his right hand was required at the level of the proximal inter-phalangeal joints due to necrosis of the distal portions of these digits. Although he could initially be extubated on day 7 post-admission, multiple nosocomial infections ensued and his nutritional status, despite optimal support, deteriorated to the extent that his skin grafts did not take and donor sites converted to full thickness skin loss, with the result that by day 51 post-admission, he effectively had 30% TBSA full thickness wounds with no prospect of further donor sites. Re-harvesting from previous donor sites was not an option with the patients Albumin levels of 11, and body weight 19 kg. From this time onwards the possibility of importing and funding the cultured epithelial autograft (CEA) Epicel® (Genzyme Inc, Boston, Massachusets) was explored. It transpired, however, that apart from prohibitive cost, there was an import restriction on the product and plans to use this option had to be abandoned. It was then decided to attempt in-house epidermal culturing with autogenous platelet-rich plasma (PRP). Expedited approval to embark on this unproven therapy as a potentially lifesaving measure was obtained from the Tygerberg Hospital Clinical Ethics Committee (at a later date approval to publish this case report was granted by the Health Research Ethics Committee of the Faculty of Medicine and Health Sciences, Stellenbosch University, reference C15/01/001 letter 30 March 2015). Consent to the proposed procedures was also given by the patient’s parents.
Methods and Materials
In preparation for skin harvesting, the seminar room (4 meters x
3.5 meters) within the Tygerberg Hospital Burn Unit was converted
into a laboratory and two borrowed paediatric incubators were
installed. These were wiped inside and out with chlorhexidine and
alcohol solution on sterile gauze. Temperature therein was set to
37°C. Two specimen bottles were sterilised with boiling water and
partially filled with a teaspoon of trypsin powder (extracted from beef
pancreas and sourced from the Division of Anatomy, Stellenbosch
University Faculty of Medicine and Health Sciences) mixed in 20
milliliters (ml) of sterile water. The small sieve from anold Recell®
kit (Avita Medical, Perth, Western Australia) was also sterilised with
boiling water.
On day 94 post-admission, the patient was taken to theatre
and a 7 cm x 3 cm full thickness skin biopsy harvested from the left
inguinal area, the defect being closed in two layers with polyglactin
4/0 subcuticular sutures. During the same procedure, a partial
thickness skin graft from the right lower leg to the right forearm and
anterior neck was performed. The remaining 26% TBSA full thickness
wounds were covered in the intensive care unit under sedation the
next day with (porcine) xenograft EZDerm® (Molnlycke Health
Care, UC LLC, Norcross, Ga). Immediately after the procedure of
day 94, the harvested skin was transferred to the “laboratory” in a
sterile container and processed on sterile surface (operating theatre
instrument pack). With the aid of optical loupes (3.5 x magnification,
45 cm focus) epidermis was separated from dermis using a scalpel
with a 15/0 blade and the epithelial fragments were cut into smaller
segments. These were then immersed in the trypsin specimen bottles
to further separate dermal elements. The trypsin was rinsed off using
the Recell sieve and remaining minute specs of epithelial cells were
transferred with forceps either to the centre of six 20 cm x 10 cm
hydrophobic dressing pads (Cutimed Sorbact®, BSN, Pinetown,
and RSA) or returned to the specimen bottles. Meanwhile six acidcitrate-
dextrose (ACD) tubes, each containing 6 ml of the patient’s
blood were centrifuged at 3500 revolutions per minute for 8 min and
supernatant plasma withdrawn into a 20 ml syringe via an 18 gauge
needle, taking care not to include any red blood cell elements. The
withdrawn plasma was sprayed onto the Cutimed Sorbact pads and
into the specimen bottles. Sorbact® ribbon gauze (BSN, Pinetown, and
RSA) was laid down between the pads in the incubators to form a
border for cell growth. Pads, as well as specimen bottles containing
epithelial cells in plasma, were thereafter incubated at 37°C. Cells
were nourished daily with newly drawn and centrifuged PRP and
the incubators were humidified by regular addition of sterile water
to the incubators’ water inlet. Since the patient was in the ICU, and
had a central venous line during the treatment period it was easy to
get blood. As a further precaution against cultured cells drying out,
modified sodium carboxymethyl cellulose, Intrasite Gel® (Smith &
Nephew Pharmaceuticals, Pinetown, KZN) was applied with a sterile
glove onto the incubated cultures every third day. Rapid growth of
cells in the specimen bottles after 7 days allowed transplantation of
the cells in suspension to more specimen jars.
By day 104 post-admission, after 14 days of culture, sufficient
epithelial cell growth had occurred and the patient was returned to
theatre. The incubator with cells was also taken to theatre and the cells
kept at 37 degrees Celsius. After anaesthesia commencement, 36 ml
of blood for PRP was taken from the arterial line. PRP was prepared
as usual. The patient was washed with Chlorhexidine soap and draped
with sterile clothes. Wound debridement was done by gentle scraping
of raw areas with a metal ruler’s blunt side/tip. This was done over
previously grafted xenograft areas with as well as a donor site on the
left medial lower leg. Haemostasis was obtained with swabs soaked
in an adrenalin solution (Ringer’s lactate 1 litre and 1 ampule of
adrenalin and 800 mg Lignocaine). Cutimed Sorbact pads with CEA’s
were then transplanted directly onto the wounds of the back, left arm,
left thigh and knee with the simultaneous application of PRP. For the patient’s right arm, thigh and leg, newly-opened Cutimed Sorbact
pads with squirted CEA’s (from the cells grown in suspension in the
specimen jars) and fresh PRP were applied. A sacral bedsore was also
treated with squirted CEA’s on Cutimed Sorbact. The back dressings
were fixed with iodine containing film dressing, Ioban® (Smith &
Nephew Pharmaceuticals, Pinetown, KZN). The rest of the dressings
were bandaged with crepe.
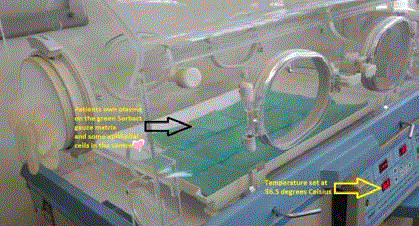
Figure 1
Figure 1
Initiation of the cell culture technique in a paediatric incubator on
day 1. A marker points at central bleb of autogenous plasma on the matrix
Cutimed Sorbact. The temperature of the incubator is set at 36.5°C, the
normal temperature of the body.
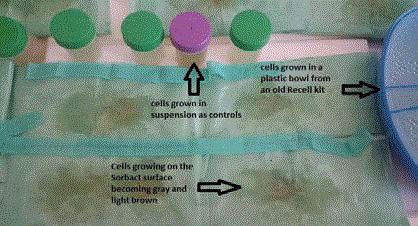
Figure 2
Figure 2
The cells on day 10 covers a larger area of the Sorbact matrix
(green), while the control cells in the sterile specimen container and old
Recell plastic bowl also grow.
Figure3
Figure 3
Above can be seen that the cells have grown to the edges
(indicated by an arrow) of the Sorbact matrix gauze on day 14 and are ready
for transplantation. Compare this image of the cell growth with the previous
images to see the progress of the cell growth on the Sorbact.
Results
The progression of cell growth is shown in Figure 1, 2 and 3
with histological confirmation of cells by simple light microscopy in
Figure 4.
The dressings were opened on day 6 post-grafting, when the
patient complained of itchiness of the wounds for the first time, and
excellent results were observed where the Sorbact Pads with the pregrown
epithelial sheets had been used. The areas where fresh Sorbact
Pads squirted with cells and plasma did not show visible confluent
epithelial layers at that time. One area on the left thigh had become
yellow and sloughy. An initial overall successful graft take was
estimated at 75%. It was possible to mobilize him out of his ICU bed,
for the first time since admission, one week after CEA. Two weeks
after CEA he was transferred to the ward. His final graft take at three
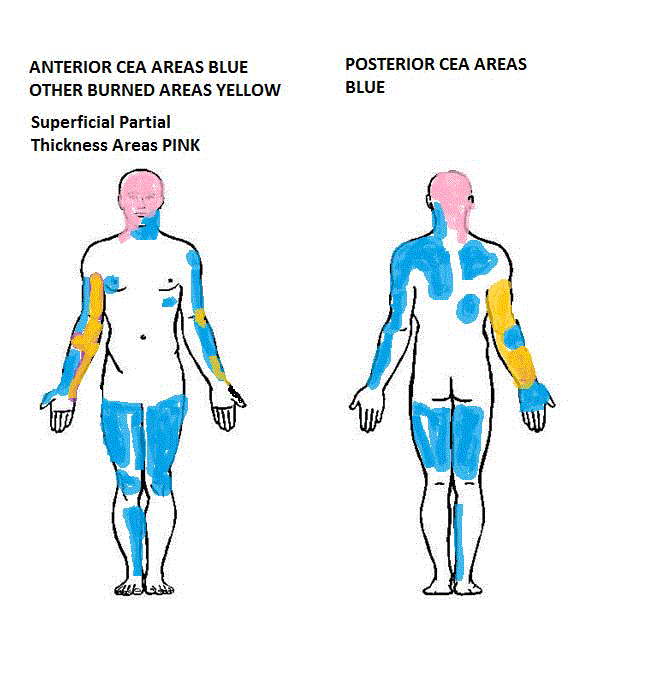
weeks post-operatively was 78%. The CEA graft areas are shown in
Figure 5.
The major problem after the patient’s wounds settled was that of
mobility and contractures of virtually all joints, but the CEA remained
stable after healing. Three months after the CEA he weighed 34 kg
and his Albumin level was 32 g/L (compared to lowest levels prior
to embarking on treatment of 19 kg and 11 g/L respectively). He was
then also mobilising better. The appearance of CEA donor site area
grafted is shown in Figure 6 and other CEA take areas in Figure 7.
The patient at discharge from hospital was in a reasonable state
of mental and physical rehabilitation. Total costs directly related to
the production of the composite CEA to 40 % of the patient’s TBSA
were calculated to be 75.26 USD (US dollars) (Table 1). Note the
cost of Sorbact Pads in the Table 1was 5.67 USD (US dollars) at that
time in 2014, but later in the text 5.97 USD for 2015. The patient only
managed to walk alone 2 years after discharge.
Discussion
Composite technique
Use of a composite CEA technique in this case was based on the
success of the largest series in the literature by the Indiana burns unit
[1]. Different techniques for CEA have been described with variable
success since the first description by Rheinwald and Green in 1975
[2]. Different substrates for growing cells have been described in the
past. Some of them are extracellular matrix from human umbilical
vein endothelial cells on gelatin coated plates, Biobrane® (Smith &
Nephew, UDL laboratories, Rockford, IL), which can be used as a
skin substitute and polyester filter substrate [3-5]. We were hopeful
that the clinical evidence that the Sorbact® dressing was effective in
stimulating wound healing via increased fibroblast activity in vitro
would support the hypothesis that it could be used as a growth
scaffold for epithelial cells with the added benefit of the dressing
having antimicrobial properties [6-8]. The transfer of cells from the
laboratory to the patient also seemed an area of progress in that if the
cells grew on the Cutimed Sorbact® it would be able to act as a transfer
dressing directly onto the patient.
It has been reported that the composite technique for CEA yields
better graft take and a more stable result compared to CEA alone [9].
Either allograft or xenograft can be used as temporary cover over
deep burn wounds. At the time of skin biopsy for CEA, the allograft
or xenograft can be applied. Two weeks later, during the application
of the CEA, the areas of allograft or xenograft can superficially be
removed to allow a raw wound bed with some dermal elements. This
is then an ideal recipient site for CEA. In the patient’s case we used a xenograft, EZ Derm®, which was generously donated to us free of
charge by the manufacturer.
We recently completed two studies evaluating Cutimed Sorbact
in burn wound care [10,11] and the hypothesis was that hydrophobic
dressings like Cutimed Sorbact could act as a growth template
for epithelial cells in a temperature controlled, relatively sterile
environment of an incubator.
The ideal dressing for cell grafting is one that is capable of
reducing the effects of shear stress, metabolic stress and tissue
infection [12]. Cutimed Sorbact has a hydrophobic coating made
from dialkylcarbamoylchloride (DACC). The DACC attracts
hydrophobic bacteria and renders them inert [6,10]. It was postulated
that the hydrophobic action of Sorbact would allow that PRP could be
applied and the cells could grow within the PRP droplets [13].
Application technique
Two grafting techniques were used. The first and easiest was the
direct application of the Cutimed Sorbact pads with the pre-grown
CEA. The second and more demanding technique was the application
of the cells that were grown in the specimen jars by a squirting
technique onto fresh Cutimed Sorbact pads. The former technique
produced superior early results in this patient and would appear to be
the method of choice.
Biological safety
Animal components should as far as possible be removed from
cell culturing [14,15]. In this case, mediums were used that came
from the patient himself. An animal feeder layer to culture the
cells, as is the case with Epicel® was avoided. Trypsin unfortunately
is bovine in origin and could not be avoided in the initial phase of
epithelial cell separation and preparation. There remains a possibility
that the cells may be infected in the culture period. Strict application
of the principles of surgical sterility to the work environment in
the laboratory assists to limit unwanted contamination of the cell
cultures. The mechanism of action of the Cutimed Sorbact® matrix
also assists to limit bacterial colonization by irreversibly binding and
deactivating bacteria.
Cost-effectiveness
Munster found that the length of stay in the hospital was longer
for patients who had CEA and therefore the cost was greater [16].
However in the group of patients that received CEA, there was a better
quality of the burn scars and a reduction in mortality compared with
the control, from 48% to 14%. The approximate cost of a 7 cm x 10 cm piece of the CEA product Epicel® was approximately 5 292.49 USD in
2014. It was estimated that approximately 136092.60 USD for Epicel®
would have been required to complete the grafting in this patient. A
piece of Cutimed Sorbact® dressing pad 10 cm x 20 cm would cost 6
USD. This is also much cheaper than Biobrane used by Frew et al. in
2013 [17]. Biobrane in South Africa would cost approximately 105.85
USD for a 13 cm x 13 cm size. Thus the CEA technique used in this
patient was cost effective and biologically safer, with an above average
graft take of 75% at one week and 78% at three weeks.
Histology results
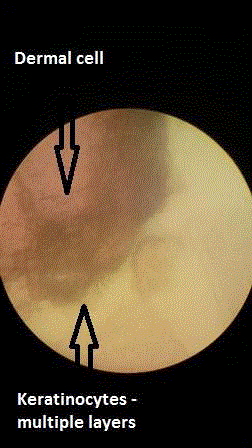
The dermal cells that were seen in Figure 4 could be explained
by the fact that there was no cell identification techniques used to
separate dermal cells from epidermal cells before the cells were
cultured. This was due to the fact that the first author did not have
the expertise to do so, and hoped that the cells that would be growing
would be the keratinocytes since this was the predominant cell type
obtained by chopping the skin up into very tiny pieces with a scalpel.
Even scraping epidermis off with a scalpel also scratches off some
dermal cells with the epidermal cells. Technically then, one could
call the technique cultured skin autograft (CSA), and not cultured
epidermal autograft.
Figure 4
Figure 4
Histological confirmation of cell growth was done by light microscopy
and Eosin staining illustrating epidermal and dermal cell differentiation.
Figure 5
Figure 5
The CEA areas are in blue, while healed superficial partial
thickness areas are in pink, and old skin grafts are in yellow.
Figure 6
Figure 6
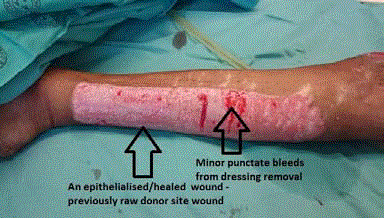
Upon removal of the Sorbact one week after CEA grafting a
completely epithelialized wound is shown with minor punctuate bleeding
points where the dressing was stuck to some small epithelial areas.
Figure 7
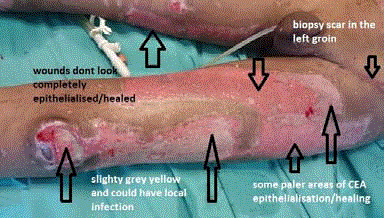
Figure 7
The left thigh initial graft take is shown with some areas that are
healing well and others that still need more epithelialisation. The original skin
biopsy site for starting the CEA process is visible in the left groin/inguinal
area. Possible early signs of wound infection are visible around the knee area
and inferior lateral thigh.
Table 1
Table 1
The estimated cost breakdown for our CEA technique. This is an
estimate created using storeroom prices obtained from the stores at Tygerberg
hospital. Many of the items used where donated for the purpose of the case
study.
Conclusion
Cutimed Sorbact® in combination with autogenous PRP has proven to be effective for CEA. There are mobile skin culture incubators being developed, as well as a fully equipped CEA treatment pack, to assist with the cost-effective use of this technique. It is planned that by the end of 2017 that these products would be available to assist clinicians. The CEA was lifesaving for our patient. Mueli and Raghunath (1997) wrote: ‘It may take one or more decades of concerted research, jointly performed by clinicians and tissue culture technology experts in order to fabricate more skin-like grafts which are robust, reliable, and less expensive. Then, “cultured skin” will conquer the world and benefit countless patients [18]. We believe that the technique described in this report is a step closer to this conquest.
Acknowledgement
• Molnlycke Health Care for donation of xenograft material. • Division of Anatomy, Stellenbosch University, for supplying Trypsin powder. • Dr Rajiv Sood for visiting South Africa and sharing his experiences with CEA. • I would also like to thank my research assistant, Ai-Ting Wong (5th Year Medical Student Stellenbosch University, MB,ChB).
References
- Sood R, Roggy D, Zieger M, Balledux J, Chaudhari S, Koumanis DJ, et al. Cultured epithelial autografts for coverage of large burn wounds in eighty-eight patients: the Indiana University experience. J Burn Care Res. 2010;31(4):559-68.
- Rheinwald JG, Green H. Serial cultivation of strains of human epidermal keratinocytes: the formation of keratinizing colonies from single cells. Cell. 1975;6:331-34.
- Solomon DE. An in vitro examination of an extracellular matrix scaffold for use in wound healing. Int J Exp Pathol. 2002;83(5):209-16.
- Frew Q, Philp B, Shelley O, Myers S, Navsaria H, Dziewulski P. The use of Biobrane® as a delivery method for cultured epithelial autograft in burn patients. Burns. 2013;39(5):876-80.
- Van Dorp AG, Verhoeven MC, Nat-Van Der Meij TH, Koerten HK, Ponec M. A modified culture system for epidermal cells for grafting purposes: an in vitro and in vivo study. Wound Repair Regen. 1999;7(4):214-25.
- Falk P, Ivarsson ML. Effect of a DACC dressing on the growth properties and proliferation rate of cultured fibroblasts. J Wound Care. 2012;21 (7):327-8, 330-2.
- Chrapusta A, Nessler MB, Drukala J, Bartoszewicz M, Mądry R. A Comparative analysis of advanced techniques for skin reconstruction with autologous keratinocyte culture in severely burned children: own experience. Postep DermAlergol. 2014;(3):164- 9.
- Gentili V, Gianesini S, Balboni PG, Menegatti E, Rotola A, Zuolo M, et al. Panbacterial real-time PCR to evaluate bacterial burden in chronic wounds tested with Cutimed Sorbact. Eur J Microbiol Infect Dis. 2011;31(7):1523-9.
- Kangesu T, Navsaria HA, Manek S, Fryer PR, Leigh IM, Green CJ. Kerato-dermal grafts: the importance of dermis for the in vivo growth of cultured keratinocytes. Br J Plast Surg. 1993;46(5):401-9.
- Kleintjes WG, Schoeman D, Collier L. A random prospective pilot study of Sorbact versus Acticoat versus Silverlon for the treatment of burn wounds in a South African adult burns unit. Wound Healing J of Southern Africa. 2015;8(1):43-52.
- Kleintjes WG, Boggenpoel A, Diango K. A random prospective non-controlled clinical study of CutimedSorbact as a skin substitute for the treatment of partial thickness burn wounds in a South African adult burns unit. Wound Healing J of Southern Africa. 2015;8(1):54-8.
- Orgill DP, Butler C, Regan JF, Barlow MS, Yannas IV, Compton CC. Vascularized collagen-glycosaminoglycan matrix provides a dermal substrate and improves take of cultured epithelial autografts. Plast Reconstr Surg. 1998;102(2):423-9.
- Du Toit DF, Kleintjes WG, Otto MJ, Erick J Mazyala, Benedict J Page, et al. Soft and hard-tissue augmentation with platelet-rich plasma: Tissue culture dynamics, regeneration and molecular biology perspective. Int J Shoulder Surg. 2007;1(2):64-73.
- De Corte P, Verween G, Verbeken G, Rose T, Jennes S, De Coninck A, et al. Feeder layer- and animal product-free culture of neonatal foreskin keratinocytes: improved performance, usability, quality and safety. Cell and Tissue Banking. 2012;13( 1):175-89
- van Dorp AG, Verhoeven MC, Nat-Van Der Meij TH, Koerten HK, Ponec M. A modified culture system for epidermal cells for grafting purposes: an in vitro and in vivo study. Wound Repair Regen. 1999;7(4):214-25.
- Munster AM. Cultured skin for massive burns. A prospective controlled trial. Ann Surg. 1996;224(3):372-5
- Frew Q, Philp B, Shelley O, Myers S, Navsaria H, Dziewulski P. The use of Biobrane® as a delivery method for cultured epithelial autograft in burn patients. Burns. 2013;39(5):876-80.
- Meuli M, Raghunath M. Burns (Part 2). Tops and flops using cultured epithelial autografts in children. Pediatr Surg Int. 1997;12(7):471-7.