Case Report
Intralenticular Ozurdex Implant: A Case Report
Shira Sheen-Ophir1,2, Raz Gepstein2 and Ehud I Assia1,2*
1Department of Ophthalmology, Meir Medical Center, Kfar Saba, Israel
2Department of Ophthalmology, Ein Tal Eye Center, Tel Aviv, Israel
*Corresponding author: Ehud I Assia, Department of Ophthalmology, Meir Medical Center, 59 Tschernihovsky St. Kfar Saba 44281, Israel
Published: 13 Jul 2017
Cite this article as: Sheen-Ophir S, Gepstein R, Assia EI.
Intralenticular Ozurdex Implant: A Case
Report. Clin Surg. 2017; 2: 1556.
Abstract
We present the case of a 54-year-old male with deterioration of vision in the right eye. CME due to non-infectious, intermediate uveitis was diagnosed. The patient was treated with an intravitreal ozurdex implant that was inadvertently injected into the lens. The lens remained transparent for 8 months, but high, uncontrolled intra-ocular pressure and posterior sub-capsular cataract developed. The patient underwent phacoemulsification cataract surgery, posterior capsulotomy and anterior vitrectomy. An IOL was implanted in the bag.
Case Presentation
A 54-year-old male presented to our clinic for evaluation for deterioration of vision in his right
eye over the last year. He had known neurofibromatosis (NF) without systemic manifestations and
low grade myopia. Ocular history included cystoid macular edema (CME) in the right eye that was
treated unsuccessfully with intraocular anti-VEGF. On examination, visual acuity was 20/60 in the
right eye and 20/20 in the left eye. Ophthalmologic examination of the right revealed clear anterior
chamber, open angle with large amount of pigment without peripheral anterior synechia, cataract
grade NS+1, vitreous cells grade 2 without haze or snow balls. The retina was attached with inferior
pigmented atrophic retinal hole with condensed vitreous above it. Macular examination revealed
CME. Left eye exam was within normal limits.
Investigation
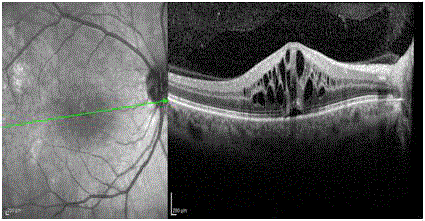
Spectral-domain optical coherence tomography revealed CME of 685 μic and sub-retinal fluid
(Figure1). Fluorescein angiography demonstrated late leakage from the inferior part of the optic
disc and CME. He had no complaints of difficulty swallowing or long standing cough; no weight
loss or diarrhea; no fever, arthritic pain or neurological signs. Systemic workup for uveitis included
complete blood count, chemistry, erythrocyte sedimentation rate, C-reactive protein, serum
angiotensin-converting enzyme (ACE), Mantoux test for tuberculosis and serology for detecting
treponema. All were within normal limits. MRI of the brain and orbit was performed to rule out
brain involvement of NF and demyelinating disease. A 6 mm meningioma near the falx cerebri on
the right side was the only finding. Chest X-ray was normal. These findings suggested a diagnosis of
noninfectious, intermediate uveitis with CME.
Management
Due to previously unsuccessful anti-VEGF treatment, we decided to treat the CME with
sustained-release 0.7 mg dexamethasone intravitreal implant (Ozurdex; Allergan, Irvine, CA).
Barrier laser photocoagulation around the atrophic hole was performed. On examination it was seen
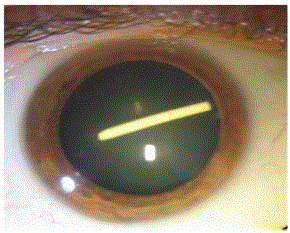
that intraocular Ozurdex implant was inadvertently injected into the lens (Figure 2). In addition,
local retinal detachment developed around the atrophic hole, which was treated with barrier laser
photocoagulation around the area of the detachment. During the next 8 months, the patient's vision
improved significantly to 20/40 pinhole, 20/25. CME resolved and remained at 244 μic to 300 μic.
The crystalline lens remained transparent for 8 months and a posterior, sub-capsular cataract
gradually developed. The Ozurdex implant was horizontally oriented involving the visual axis,
which could explain the visual acuity improvement with pinhole. The IOP gradually increased to
32 mmHg. The patient was placed on maximum medical topical and oral doses. However, he could
not tolerate the oral drugs due to side-effects. It was decided to perform cataract surgery and to
remove the ozurdex implant simultaneously. The ozurdex implant penetrated the posterior capsule
into the clear lens where the PSC developed. Routine phacoemulsification cataract surgery was
performed under topical anesthesia. After a clear corneal incision and insertion of anterior chamber
maintainer, standard capsulorhexis, hydrodelamination, hydrodissection, and phacoemulsification
were performed. At the end of nucleus removal, a tear in the posterior capsule was identified. The anterior chamber was filled with ophthalmic viscoelastic devices (OVD), a continuous, circular posterior capsulotomy was performed
in addition to anterior vitrectomy and PCIOL was implanted in
the bag. Post-operatively, maximal anti-glaucoma therapy was
initiated, along with topical antibiotics and steroids (See attached
video of the surgery). Complications involving the anterior segment
after injection of intravitreal dexamethasone implants are rare and
mainly involve migration of the implant into the anterior chamber
in pseudophakic eyes with posterior capsular tear [1-3]. A few cases
regarding inadvertent injections of dexamethasone implants into the
crystalline lens have been reported [4-9]. In all cases, cataract surgery
was performed and a 3-piece IOL was implanted in the sulcus. Except
for one case [10], fixation of the intraocular lens (IOL) into the
capsular bag was enabled due to fibrosis of the capsular defect.
Follow-up
During the first month after surgery, the anti-glaucoma therapy
was gradually tapered as the IOP decreased. UCVA was 20/40. On the
fifth week after surgery, CME reappeared to 487 μic. It was decided
with the patient to treat with periocular transeptal methylprednisolone
injection (Depo-Medrol, Pfizer, New York). One month after the
injection, CME worsened to 598 μic and subretinal fluid developed.
It was decided to start prednisone 60 mg per os and anti-glaucoma
drops. However, the CME recurred when steroids were tapered to 20
mg. We then decided to inject Ozurdex implant intravitreally, which
successfully resolved the CME. The IOP was controlled at around 18
mmHg by a combination of carbonic anhydrase inhibitors and betablockers.
Figure 1
Figure 1
Spectral-domain optical coherence tomography revealed CME of
685 μic and sub-retinal fluid.
Figure 2
Discussion
Uveitis refers to a group of intraocular inflammatory diseases that cause 10% to 15% of blindness in the developed world. Despite advances in immunosuppressive therapy, corticosteroids remain the mainstay of treatments. They can be administered systemically or locally by topical, periocular, or intravitreal routes. Persistent inflammation and cystoid macular edema) secondary to ocular inflammation are often vision-threatening and pose a significant therapeutic challenge. The Ozurdex® (Allergan, Inc., Irvine, CA, USA) dexamethasone drug delivery system is a biodegradable intravitreal implant that delivers sustained release of 700 μg of preservative-free dexamethasone to the retina and vitreous. It is approved by the United States Food and Drug Administration as a first-line therapy for the treatment of macular edema following branch or central retinal vein occlusion, as well as for noninfectious posterior uveitis [11-13]. Injecting steroids into the vitreous cavity has the advantage of minimal systemic sideeffects. The use of Ozurdex is encouraging due to its potency, dose consistency, extended duration of action, and minimal adverse effects [14-18]. However, we also need to manage local side-effects such as cataract formation, IOP elevation, subconjunctival hemorrhage, hyperemia, and conjunctival edema. These are often temporary and can be managed medically [19,20]. Dexamethasone has potent anti-inflammatory properties with a favorable side-effect profile [11,12,21]. Previous studies demonstrated that dexamethasone in a biodegradable drug delivery system (Ozurdex®; Allergan Inc, Irvine, California, USA) can improve visual acuity and macular thickness in a variety of settings [11,12,21]. In our case, the Ozurdex implant was inadvertently injected into the visual axis of the lens, but it remained clear for 8 months. PSC developed only in the area where the Ozurdex implant penetrated the lens. Our decision to perform cataract surgery due to the implant in the lens, the cataract and the high IOP that arose and needed full medical treatment. The entrance tear in the posterior capsule that was caused by the implant, was converted to posterior capsulotomy and PCIOL was successfully implanted in the bag. To the best of our knowledge, this is the only case reported where the IOL was implanted in the bag. Because the Ozurdex was implanted in the crystalline lens it degraded much more slowly than it normally would have. The very slow, sustained release of the dexamethasone in the implant prevented CME from developing for 8 months until the lens and the implant were extracted. During the long follow-up period, the patient underwent several intravitreal ozurdex injections with successful resolution of CME, with only two anti-glaucoma drugs required.
References
- Cronin KM, Govind K, Kurup SK. Latemigration of dexamethasone implant into anterior chamber. Arch Ophthalmol.2012;130(6):711.
- Malclès A, Janin-Manificat H,Yhuel Y, Russo A, Agard E, El Chehab H, et al. [Anterior chamber migration ofintravitreal dexamethasone implant (Ozurdex®) in pseudophakic eyes:report of three cases]. J Fr Ophtalmol. 2013;36(2):362-7.
- Pardo-López D, Francés-Muñoz E,Gallego-Pinazo R, Díaz-Llopis M. Anterior chamber migration of dexamethasoneintravitreal implant (Ozurdex®). Graefes Arch Clin Exp Ophthalmol.2012;250(11):1703-4.
- Coca-Robinot J, Casco-Silva B,Armadá-Maresca F, García-Martínez J. Accidental injections of dexamethasoneintravitreal implant (Ozurdex) into the crystalline lens. Eur J Ophthalmol.2014;24(4):633-6.
- Berarducci A, Sian IS, Ling R.Inadvertent dexamethasone implant injection into the lens body management. EurJ Ophthalmol. 2014;24(4):620-2.
- Martin-Moro JG, GallardoJZ, Escobar IL, Soriano RC. Dexamethasone implants into the crystalline lens.Eur J Ophthalmol. 2015;25(5):e101-e2.
- Chhabra R, Kopsidas K, Mahmood S.Accidental insertion of dexamethasone implant into the crystalline lens--12months follow-up. Eye (Lond). 2014;28(5):624-5.
- Fasce F, Battaglia Parodi M, KnutssonKA, Spinelli A, Mauceri P, Bolognesi G, et al. Accidental injection ofdexamethasone intravitreal implant in the crystalline lens. Acta Ophthalmol.2014;92(4):e330-1.
- Ram J, Agarwal AK, Gupta A.Phacoemulsification and intraocular lens implantation after inadvertentintracapsular injection of intravitreal dexamethasone implant. BMJ Case Rep.2012; pii: bcr2012007494.
- Koller S, Neuhann T, Neuhann I.[Conspicuous crystalline lens foreign body after intravitreal injection.]Ophthalmologe. 2012;109(11):1119-21.
- Kupperman BD, Blumenkranz MS,Haller JA, Williams GA, Weinberg DV, Chou C, et al. Dexamethasone DDS Phase IIStudy Group. Randomized controlled study of an intravitreous dexamethasone drugdelivery system in patients with persistent macular edema. Arch Ophthalmol.2007;125(3):309-17.
- Williams GA, Haller JA, KuppermanBD, Blumenkranz MS, Weinberg DV, Chou C, et al. Dexamethasone DDS Phase IIStudy Group. Dexamethasone Posterior-Segment Drug Delivery System in thetreatment of macular edema resulting from uveitis or Irvine-Gass syndrome. Am JOphthalmol. 2009;147(6):1048-54.
- Haller JA, Bandello F, Belfort RJr, Blumenkranz MS, Gillies M, Heier J, et al. Ozurdex® Geneva StudyGroup. Randomized, sham-controlled trial of dexamethasone intravitreal implantin patients with macular edema due to retinal vein occlusion. Ophthalmology.2010;117(6):1134-46.
- Myung JS, AakerGD, Kiss S. Treatment of noninfectious posterior uveitis with dexamethasoneintravitreal implant. Clin Ophthalmol. 2010;4:1423-6.
- Ghosn CR, Li Y, Orilla WC, Lin T,Wheeler L, Burke JA, et al. Treatment of experimental anterior and intermediateuveitis by a dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci.2011;52(6):2917-23.
- Herrero-VanrellR, Cardillo JA, Kuppermann BD. Clinical applications of the sustained-releasedexamethasone implant for treatment of macular edema. Clin Ophthalmol.2011;5:139-46.
- Saraiya NV, Goldstein DA. Dexamethasonefor ocular inflammation. Expert Opin Pharmacother. 2011;12(7):1127-31.
- London NJ, Chiang A, Haller JA. Thedexamethasone drug delivery system: indications and evidence. Adv Ther.2011;28(5):351-66.
- van Kooji B, Rothova A, de VriesP. The pros and cons of intravitreal triamcinolone injections for uveitis andinflammatory cystoid macular edema. Ocul Immunol Inflamm. 2006;14(2):73-85.
- Goldstein DA, Godfrey DG, Hall A,Callanan DG, Jaffe GJ, Pearson PA, et al. Intraocular pressure in patients withuveitis treated with fluocinolone acetonide implants. Arch Ophthalmol. 2007;125(11):1478-85.
- HallerJA, Kupperman BD, Blumenkranz MS, Williams GA, Weinberg DV, Chou C, et al.Dexamethasone DDS Phase II Study Group. Randomized controlled trial ofintravitreous dexamethasone drug delivery system in patients with diabeticmacular edema. Arch Ophthalmol. 2010;128(3):289-96.