Case Report
A Rare Case of Tricuspid Regurgitation in the Setting of Orthotopic Heart Transplantation
Maria Rosal Martins1*, Alain Poncelet2, Michel Van Dyck1, Olivier Van Caenegem3, Christine
Watremez1, Thierry Sluysmans4 and Mona Momeni1
1Department of Anesthesiology, Université Catholique de Louvain, Cliniques universitaires Saint-Luc, Belgium
2Department of Cardiothoracic and vascular surgery, Université Catholique de Louvain, Cliniques universitaires
Saint-Luc, Belgium
3Department of Cardiovascular Intensive Care, Université Catholique de Louvain Cliniques universitaires Saint-
Luc, Belgium
4Department of Cardiac Pediatrics, Université Catholique de Louvain, Cliniques universitaires Saint-Luc, Belgium
*Corresponding author: Maria Rosal Martins, Department of Anesthesiology, Université Catholique de Louvain, Cliniques universitaires Saint-Luc, Belgium
Published: 07 Jul 2017
Cite this article as: Martins MR, Poncelet A, Van Dyck
M, Van Caenegem O, Watremez C,
Sluysmans T, et al. A Rare Case of
Tricuspid Regurgitation in the Setting of
Orthotopic Heart Transplantation. Clin
Surg. 2017; 2: 1547.
Abstract
A 13-year-old female was admitted to our hospital with terminal heart failure. After 50 days of extracorporeal life support, she underwent orthotopic heart transplantation (OHT). The donor had no cardiac history. During the following days after OHT, the patient presented right ventricular failure with severe tricuspid regurgitation (TR). The decision to perform a tricuspid valve repair was taken after right heart catheterization and multiple echocardiographies. The diagnosis of Ebstein’slike anomaly was made based on intraoperative echocardiographic features and anatomical observance during surgery. This case illustrates that congenital cardiac defects must be considered when facing an important TR after OHT.
Introduction
Tricuspid regurgitation (TR) is the most common valve abnormality after an orthotopic heart transplant (OHT) [1]. In the first few hours or days and in the majority of the situations, its cause is functional. However, an anatomic cause must be considered. While rare, an Ebstein’s anomaly is among the congenital heart lesions that can present into adulthood without any or only few symptoms [2]. We present a case of Ebstein’s-like anomaly diagnosed on the recipient after OHT from a donor with unknown history of cardiac disease.
Case Presentation
A 13-year-old female, with a respective weight and height of 38.1 kg and 159.5 cm, was followedup
in our institution after a previous mitral valve repair due to a congenital mitral valvulopathy that
was associated with left ventricular dysfunction. At the last follow-up visit, very poor biventricular
function was observed with severe mitral and tricuspid regurgitation and elevation of the right heart
pressures. In fact, the patient had become more symptomatic during the last 3 previous months but
did not consult. In face of extremely severe heart failure symptoms, it was decided to introduce an
extracorporeal life support (ECLS) as bridging therapy until OHT. After 53 days of ECLS therapy,
the transplantation team accepted a heart despite the advanced age of the donor. This decision was
taken because of the prolonged duration of the ECLS therapy and the difficulty to find a matching
organ.
The donor was a 65 year-old-female, with a respective weight and height of 60 kg and 165
cm, with known hypercholesterolemia, high arterial blood pressure, treated rheumatoid arthritis
but no history of cardiac disease. The cause of death was cerebral hemorrhage. The transthoracic
echocardiography (TTE) performed in order to propose for heart donation did not reveal any
cardiac abnormalities either functional or anatomic. A bicaval OHT was accomplished with no
difficulties although the patient needed two hours of assistance and high doses of inotropic support
before weaning from the cardiopulmonary bypass (CPB). The ischemic time was 249 min. The
intraoperative transesophageal echocardiography (TEE) showed a mild TR after improvement of
the right ventricular (RV) function with the inotropic agents. The electrocardiogram performed
postoperatively showed a right bundle branch block. The evolution during the following days
was slow. Weaning from the ventilator was achieved after 6 days but renal failure and signs of RV dysfunction persisted. A TTE was performed regularly during the following days after OHT. From day 12 after OHT and further on, TR
progressively aggravated and became severe with a leaflet coaptation
defect of 1 cm. The cause of this TR was unclear. An evaluation of
the right-sided heart pressures was performed under light sedation.
The central venous pressure was 23 mmHg - 24 mmHg, RV systolic
and diastolic pressures were respectively 34 mmHg and 6 mmHg
and the pulmonary artery systolic, diastolic and occlusion pressures
were respectively 33 mmHg, 12 mmHg and 20 mmHg. The decision
to proceed to a new surgical intervention was thoughtfully discussed
between all parties as the patient already had undergone multiple
heart surgeries and the OHT was still recent. Based on the TR
aggravation and cardiac catheterization results, the decision to repair
the tricuspid valve was taken.
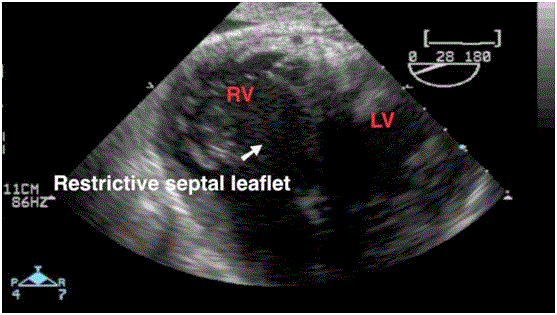
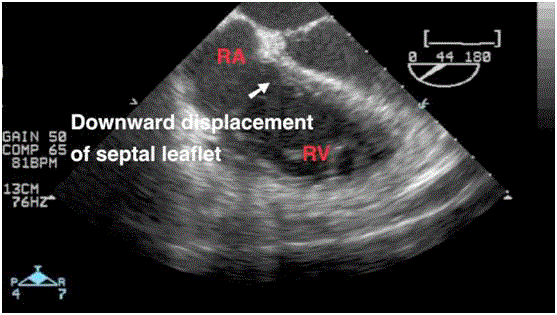
The intraoperative TEE that we show here revealed an Ebstein’slike
anomaly on the transplanted heart characterized by a restrictive
motion (Video 1) and a downward displacement of the septal leaflet
(Video 2).
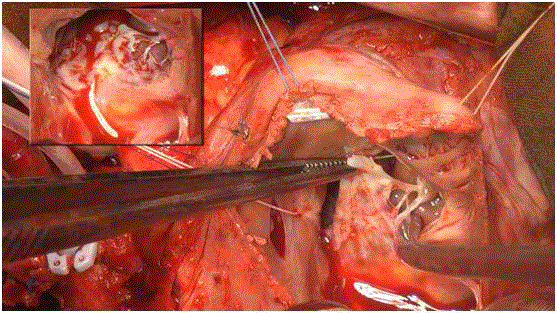
The anatomical observations during the tricuspid valve repair
confirmed the diagnosis (Figure 1). One year after OHT, the patient
is clinically doing very well. The most recent TTE showed a good
ventricular function associated to a mild to moderate TR.
video 1
video 2
Discussion
Comment
TR after OHT is not uncommon, with a reported incidence of up
to 84%. Its incidence varies strongly based on the definition used, on
the surgical techniques performed and on the timing of the diagnosis.
Here we will exclusively focus on the immediate TR after OHT. The
TR etiology can be divided into two main mechanisms: functional
and anatomic [1]. Functional TR is generally accepted as the most
frequent cause of TR after OHT.
Functional TR
The common causes of functional TR after OHT include:
• RV dysfunction/failure: As RV dysfunction may lead to
TR it must be considered as one of the main causes of TR. There are
different reasons responsible for RV dysfunction such as prolonged
ischemic time of the donor heart, donor-recipient size mismatch, and
acute rejection, mechanical obstruction at the pulmonary anastomosis,
pre-existing pulmonary arterial hypertension and pulmonary arterial
hypertension induced by the protamine administration [2,3].
• Surgical techniques: biatrial versus bicaval. Many studies
have concluded that the bicaval technique, also used by our surgical
team, reduces the risk of TR after OHT among other benefits [4].
Anatomic TR
The donor’s heart is usually considered to be free of defects
because an initial functional and structural evaluation is systematically
performed on a heart considered for donation. Nevertheless,
anatomical defects should be considered while facing a TR after OHT.
There are two forms of anatomic TR:
• Acquired: chest trauma is probably the most likely to be
encountered in the context of a heart transplant. Other causes of
acquired valvulopathy can be considered but are unlikely to be
observed in this context: endocarditis, rhumatic valve disease, etc. [5].
• Congenital: In Belgium, congenital cardiac defects account
for 6.6 per 1000 births including life births, late fetal deaths and
termination of pregnancy for fetal anomaly [6].
• Ebstein’s anomaly is one of the congenital diseases of the
tricuspid valve (TV). Its incidence is one to five per 200.000 births
accounting for less than 1% of all congenital heart diseases. The main
feature of this anomaly is an incomplete delamination of the septal
and sometimes posterior leaflets of the TV during embryological
development with an apical displacement of the functional annulus
of the TV. This can lead to different abnormalities:
1. Atrialization of the RV resulting in a hypoplastic functional
ventricle.
2. Variable TR with a central regurgitant orifice. A stenotic
TV can be exceptionally found.
3. Enlargement of the right atrium.
4. Variable RV and LV dysfunction.
5. Variable RV inflow and/or outflow tract obstruction.
The echocardiographic features of an Ebstein’s anomaly are
traditionally defined as a variable degree of tethering of the RV
leaflets (septal and posterior) towards the right ventricular free wall
and the ventricular septum resulting in a "bubble-like" appearance.
These abnormalities of the tricuspid leaflets result in a reduction in
the size of the functioning RV and the so called "atrialization" of the
RV. It has been suggested that the displacement of the annular hinge
of the septal leaflet as compared to the annular hinge of the anterior
mitral leaflet as seen in the apical 4-chamber view can be diagnostic.
Other echocardiographic features of the Ebstein's anomaly are dilated
tricuspid valve annulus, redundant and/or fenestrated anterior leaflet,
shortened cordal support, absence of the septal or mural leaflets and
dilated RV. An atrial septal defect or a patent foramen ovale is often
observed [7].
The conduction system is often abnormal because it is prolonged
in the enlarged right atrium. Right bundle branch block is extremely
common and this was also the case with our patient. The accessory
atrioventricular pathways in Ebstein's anomaly commonly result in
supraventricular and ventricular tachyarrhythmias. The timing of the
occurrence and the presentation of the symptoms will be dictated by
the severity of the disease. The most severe forms will present with
heart failure in the neonatal period. Patients with a moderate form
may present the first symptoms in adulthood as dyspnea, fatigue
and arrythmias. In the immediate postoperative period of an OHT,
a functional TR is clearly the most common mechanism of tricuspid
pathology. In this case, the association of an anatomical defect as an
Ebstein's-like anomaly to a functional stress caused intraoperatively,
such as prolonged ischemic time and donor-recipient size-mismatch,
triggered a TR which most likely was not present at the time of
donor proposal. As shown in this case, an Ebstein’s-like anomaly can
possibly be asymptomatic in adulthood and as such undiagnosed in
suitable donor hearts. While most of the cardiac abnormalities will
be detected during the initial evaluation for donor organ proposal,
it is acceptable to consider that an experienced adult cardiologist
would miss a rare and minor form of a congenital heart disease. This
case illustrates that, while rare, congenital cardiac defects must be
considered when facing an important TR after OHT. Therefore, a
thorough intraoperative TEE examination of the TV at the moment
of OHT should exclude any acquired and congenital tricuspid
malformations. Moreover, a reliable preoperative echocardiography
examination should be part of the work-up leading to a transplant
free of anatomical malformations.
Figure 1
References
- WongRC-C, Abrahams Z, Hanna M, Pangrace J, Gonzalez-Stawinski G, Starling R, et al.Tricuspid regurgitation after cardiac transplantation: an old problemrevisited. J Heart Lung Transplant. 2008;27(3):247-52.
- SilversidesCK, Salehian O, Oechslin E, Schwerzmann M, Vonder Muhll I, Khairy P, et al.Canadian Cardiovascular Society 2009 Consensus Conference on the management ofadults with congenital heart disease: complex congenital cardiac lesions. Can JCardiol. 2010;26(3):e98-117.
- Hensley FA, Martin DE, Gravlee GP. Anestheticmanagement of cardiac transplant, Pract. Approach Card. Anesth. 5thedition. Philadelphia, Wolters Kluwer Health/Lippincott Williams & Wilkins.2013;481-505.
- Jacob S,Sellke F. Is bicaval orthotopic heart transplantation superior to the biatrialtechnique? Interact Cardiovasc Thorac Surg. 2009;9(2):333-42.
- Hensley FA, Martin DE, Gravlee GP. AnestheticManagement for the surgical treatment of valvular heart disease, Pract.Approach Card. Anesth. 5th edition. Philadelphia, Wolters KluwerHealth/Lippincott Williams & Wilkins. 2013;319-58.
- Dolk H,Loane M, Garne E; European Surveillance of Congenital Anomalies (EUROCAT)Working Group. Congenital heart defects in Europe: prevalence and perinatalmortality, 2000 to 2005. Circulation. 2011;123(8):841-9.
- Negoi RI,Ispas AT, Ghiorghiu I, Filipoiu F, Negoi I, Hostiuc M, et al. Complex Ebstein’sMalformation: Defining Preoperative Cardiac Anatomy and function. J Card Surg.2013;28(1):70-8.