Case Report
Surgical Repair of a Pseudoaneurysm of the Ascending Aorta to Pulmonary Artery Fistula after Replacement of the Ascendan Aorta and Aortic Valve
Ilhan Golbasi*, Cemal Kemaloglu, Ozan Erbasan and Ömer Bayezid
Department of Cardiac and Vascular Surgery, Akdeniz University, Turkey
*Corresponding author: Ilhan Golbasi, Department of Cardiac and Vascular Surgery, Akdeniz University, 07070, Turkey
Published: 05 Jul 2017
Cite this article as: Golbasi I, Kemaloglu C, Erbasan
O, Bayezid Ö. Surgical Repair of a
Pseudoaneurysm of the Ascending
Aorta to Pulmonary Artery Fistula after
Replacement of the Ascendan Aorta
and Aortic Valve. Clin Surg. 2017; 2:
1534.
Abstract
We report the case of a patient with a pseudoaneurysm of the ascending aorta with right pulmonary
artery fistula diagnosed after aortic valve and supra-coronary ascending aorta replacement for aortic
dissection 4 years previously. He was referred to our hospital because of dyspnea and chest pain. A
preoperative diagnostic evaluation revealed a pseudoaneurysm of the ascending aorta (10 cm × 9 cm
× 7 cm) close to the proximal anastomotic site of the graft. During surgery, it was determined that
the pseudoaneurysm originated from an intimal defect in the aortic wall on the right side of the left
coronary artery at the proximal suture line. Pseudoaneurysm of the ascending aorta with pulmonary
artery fistula was closed by a 2.5 cm × 4 cm PTFE patch, and aortic defect was repaired with a 1, 5
cm × 2 cm PTFE patch using a femorofemoral bypass. The patient was discharged 9 days later and
remained well at a follow-up 6 months thereafter.
Pseudoaneurysm after aortic valve and ascending aorta replacement develops usually due to aortic
graft anastomotic dehiscence, infectious, periannular abscess, and perigraft hematoma. Erosion
into the pulmonary artery with resultant fistula formation may develop probably due to continuous
pulsatile friction between the pseudo-aneurysmatic wall and pulmonary artery. Pseudoaneurysm
with aorta-pulmonary fistula is an extremely rare and significant complication following aortic
surgery. We report the case of a patient with pseudoaneurysm of the ascending aorta with right
pulmonary artery fistula that was successfully repaired surgically.
Keywords: Aortic pseudoaneurysm; Aortic root replacement; Ascending aorta aneurysm; Aorta
to pulmonary artery fistula
Case Presentation
A 62-year-old male was admitted due to progressive exertional dyspnea and peripheral edema,
cough, and chest pain for 4 month. Medical history included surgical replacement of the aorta and
mechanical aortic valve due to aortic dissection 4 years ago. Physical examination was remarkable
for elevated jugular venous pressure and a prominent systolo-diastolic murmur heard in the right
second and third intercostal spaces. Chest radiography showed cardiomegaly, enhancement of
the right pulmonary vasculature and pulmonary congestion. Computed tomography (CT) also
showed the aortic pseudoaneurysm (10 cm × 9 cm × 7 cm) and the communication between the
pseudoaneurysm and the right pulmonary artery. The neck of the pseudoaneurysm was small (7
mm), located 3 cm above the level of the prosthetic valve (Figure 1).
Echocardiography showed an aortic prosthesis functioning normally and a huge mass in the
anterior portion of the ascending aorta measuring 9 cm × 11.5 cm in its larger axis and sign of
aortopulmonary shunt. Catheterization of the right heart catheter revealed a central venous pressure
of 14 mmHg and a pulmonary pressure of 53/22 mmHg. The patient was taking metoprolol and
warfarin (Coumadine).
After initial preparation, the common femoral artery and vein were cannulated, with the use of
femoro-femoral bypass, extracorporeal circulation was installed and the patient was cooled to 22°C
before sternotomy. Later, the venting catheter was inserted directly into the left atrium through
the superior right pulmonary vein. The ascenden aorta was cross clamped and retrograde blood
cardioplegia was delivered after sternotomy. The retrograde cardioplegia was repeated every 20 min.
Resternotomy was performed and the ascending aorta pseudoaneurysm was directly approached with cardiopulmonary bypass. The pseudoaneurysm originated from
an intimal defect in the aortic wall in the proximal aorto-dacron suture
line. The pseudoaneurysm appeared to be chronic. It was resected
from the surrounding outflow tract and aspirated. Fistula between the
pseudoaneurysm and pulmonary artery was closed by a 2.5 cm × 4 cm
PTFE patch after right pulmonary artery was clamped from proximal
and distally of the defect. Aortic defect was repaired with a 1.5 cm ×
2 cm PTFE patch. Mattress stitches of 4/0 polypropylene reinforced
with Teflon pledgets are used to approximate the patch.
Rewarming and weaning of bypass followed. The total
cardiopulmonary bypass time was 180 min and cross clamp time was
66 min. The postoperative period was uneventful, and the patient
was discharged 9 days later and remained well at a follow-up 12
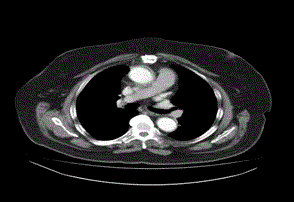
month thereafter. The radiologic assessment by torax CT was normal
12month after the surgery (Figure 2).
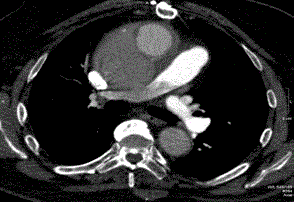
Figure 1
Figure 1
Axial plane of helical tomography depicting a pseudoaneurysm (P)
of the aorta (A), the communication between the pseudoaneurysm and the
right pulmonary artery.
Figure 2
Discussion
Pseudoaneurysm is a pulsating hematoma that develops secondary
to anastomotic dehiscence. The differential diagnosis of a perigraft
cavity or collection observed after aortic root replacement surgery
includes pseudoaneurysm from an aortic graft or coronary artery
anastomotic dehiscence, infectious pseudoaneurysm, periannular
abscess, and perigraft hematoma. The presence of friable periannular
tissue in these patients contributes to anastomotic failure. Surgical
management for pseudoaneurysm is always mandatory, because of
potentially lethal complications [1-4].
Ascending aortic pseudoaneurysms are usually asymptomatic,
unless they compress the adjacent structures. Large pseudoaneurysms
may compress an adjacent native vascular structure, cardiac chamber,
or aortic graft and may obstruct blood flow. Especially, the superior
vena cava is compressed. Thromboembolism or ruptures are other
potential complications [5]. Erosion into the pulmonary artery and
fistula formation is probably caused by continuous pulsatile friction
between pseudo aneurysmatic wall and the right pulmonary artery
[6]. Although symptoms are usually dominated by rather atypical
findings of heart failure, the auscultatory presence of a continuous
systolo-diastolic murmur should raise suspicion for fistulization.
Fistulizing pseudoaneurysm should be differentiated from
ruptured sinus of Valsalva aneurysm. Echocardiography, computed
tomography, aortography, right heart catheterization and magnetic
resonance angiography can differentiate and confirm the diagnosis
of pseudoaneurysm with aortopulmonary fistula. During pulmonary
artery catheter placement, high oxygen saturation in the pulmonary
artery and step-up between the right atrium and the pulmonary artery
are useful clues for the diagnosis of aortopulmonary fistulization.
The risk of the rupture of a pseudoaneurysm should be taken into
consideration as an indication for emergency surgery, especially in
the case of large masses. Pseudoaneurysm formation can generate in
a variable period of time, ranging from the early postoperative period
to the late postoperative period [6-8].
Rupture during the redo sternotomy or mediastinal dissection
due to the reduction in pressure around the formation when opening
and sliding the sternum can cause surgical catastrophes so the
treatment of ascending aorta pseudoaneurysms remains a challenge.
As the pseudoaneurysm is adherent to sternum, sternotomy cannot
be performed until profound hypothermia is established using
femoral or axillary-femoral bypass [7,9].
In summary, postoperatively pseudoaneursym should be
considered in patients with extensive atherosclerotic disease
or dissection in the ascending aorta. Pseudoaneursym must be
investigated in patients with mediastinal enlargement on chest X-ray
or systolo-diastolic murmur.
References
- Sullivan KL, Steiner RM, Smullens SN, Griska L, Meister SG. Pseudoaneurysm of the ascending aorta following cardiac surgery. Chest. 1988;93(1):138-43.
- Bachet J, Pirotte M, Laborde F, Guilmet D. Reoperation for giant false aneurysm of the thoracic aorta: how to reenter the chest? Ann Thorac Surg. 2007;83(5):1610-4.
- Mohammadi S, Bonnet N, Leprince P, Kolsi M, Rama A, Pavie A, et al. Reoperation for false aneurysm of the ascending aorta after its prosthetic replacement: surgical strategy. Ann Thorac Surg 2005;79(1):147-52.
- Dhadwal AK, Abrol S, Zisbrod Z, Cunningham JN Jr. Pseudoaneurysms of the ascending aorta following coronary artery bypass surgery. J Card Surg. 2006;21(3):221-4.
- Katsumata T, Moorjani N, Vaccari G, Westaby S. Mediastinal false aneurysm after thoracic aortic surgery. Ann Thorac Surg. 2000;70(2):547-52.
- Kitamura T, Motomura N, Ohtsuka T, Shibata K, Takayama H, Kotsuka Y, et al. Aortopulmonary fistula in pseudoaneurysm after ascending aortic surgery. J Thorac Cardiovasc Surg. 2003;126(3):904-5.
- Hussain J, Strumpf R, Wheatley G, Diethrich E. Percutaneous closure of aortic pseudoaneurysm by Amplatzer occluder device-case series of six patients. Catheter Cardiovasc Interv. 2009;73(4):521-9.
- Malvindi PG, van Putte BP, Heijmen RH, Schepens MA, Morshuis WJ. Reoperations for aortic false aneurysms after cardiac surgery. Ann Thorac Surg. 2010;90(5):1437-43.
- Tae-Eun Jung, Dong-Hyup Lee. Surgery for pseudoaneurysm of the ascending aorta under moderate hypothermia. J Cardiothorac Surgery. 2011;6:125.