Research Article
Yttrium-90 Radioembolisation versus Transarterial Chemoembolisation for Unresectable Hepatocellular Carcinoma: A Retrospective Comparative Analysis According to BCLC Classification
Marco Massani1*, Giovanni Pirozzolo1, Tommaso Stecca1, Davide Barbisan2, Franco Dalla
Pozza3, Giovanni Morana2 and Nicolo’ Bassi1
1Department of Surgical Oncology, IV Divisione Chirurgica Centro Regionale per la chirurgia epatobiliopancreatica, Ospedale Regionale Treviso, Italy
2Department of Radiology, IV Divisione Chirurgica Centro Regionale per la chirurgia epatobiliopancreatica,
Ospedale Regionale Treviso, Italy
3Departemnt of Nuclear Medicine, IV Divisione Chirurgica Centro Regionale per la chirurgia epatobiliopancreatica,
Ospedale Regionale Treviso, Italy
*Corresponding author: Marco Massani, Department of Surgical Oncology, IV Divisione Chirurgica Centro Regionale per la chirurgia epatobiliopancreatica, Ospedale Regionale Treviso, Ospedale Regionale Ca’ Foncello, 31100 Treviso, Italy
Published: 29 Jun, 2017
Cite this article as: Massani M, Pirozzolo G, Stecca T,
Barbisan D, Pozza FD, Morana G, et al.
Yttrium-90 Radioembolisation versus
Transarterial Chemoembolisation
for Unresectable Hepatocellular
Carcinoma: A Retrospective
Comparative Analysis According to
BCLC Classification. Clin Surg. 2017;
2: 1528.
Abstract
Introduction: Transarterial Chemoembolisation (TACE) and Transarterial Radioembolisation
(TARE) are therapeutic options for unresectable hepatocellular carcinoma (HCC). The effectiveness
of these procedures has been compared in several retrospective series, but few stratified the results
according to the Barcelona Clinic Liver Cancer (BCLC) staging system. The aim of this single
centre retrospective study was to compare the effectiveness of TARE and TACE and to evaluate the
outcomes according to BCLC stage.
Methods: A retrospective analysis of data from a single centre in Italy from 121 consecutive patients
with unresectable HCC who underwent TARE (n=39) or TACE (n=82). The primary endpoint was
Overall Survival (OS) determined by the Kaplan-Meier method. Further survival analyses by BCLC
stage, and a multivariate analysis for other factors affecting survival, were performed.
Results: There were no substantial differences in mean or median OS times between the TARE
and TACE groups (24.05 vs. 27.39 months, and 21.00 vs. 23.50 months, respectively). Increased
OS was observed with TARE versus TACE in patients with BCLC stage B HCC, but there were
no differences between treatments in patients with BCLC stage C HCC. At 24 months after the
procedure, in patients with BCLC stage B HCC, TARE resulted in significantly greater mean OS
time than TACE (21.53±1.61 vs. 17.06±1.50 months, respectively).
Conclusion: TARE and TACE were similarly effective; however, subgroup analysis showed an
enhanced survival in TARE patients with BCLC stage B disease. Prospective studies with adequate
follow-up could further clarify the observed differences, potentially producing objective data to
guide treatment in this patient group.
Keywords: Barcelona clinic liver cancer staging; Hepatocellular carcinoma; Overall survival;
Transarterial Chemoembolisation; Transarterial radioembolisation
Introduction
Primary liver cancer is the fifth most common cancer worldwide and the third most frequent
cause of cancer death [1]. Hepatocellular carcinoma (HCC) accounts for between 85% and 90%
of primary liver cancers. HCC usually develops on a background of a chronic liver disease such as
chronic infection with hepatitis B virus (HBV) or hepatitis C virus (HCV), alcoholic liver disease
or non-alcoholic fatty liver disease. The most frequent risk factor for HCC is HBV infection, which
accounts for about 50% of cases [2].
Surgery is the best treatment option for HCC patients but a significant proportion of
malignancies are diagnosed at advanced tumour stage or associated with a poor liver function.
These situations invalidate the surgical approach, and thus, require a non-surgical approach to management. Transarterial Chemoembolisation (TACE) is often
used as a therapeutic strategy in intermediate-stage disease patients
on the Barcelona Clinic Liver Cancer [BCLC] staging system [3]. The
safety and efficacy of TACE in the treatment of patients with advanced
HCC has been reported in several trials [4-6] and drug-eluting-beads
TACE (DEBTACE) for treating HCC is comparable to conventional
TACE (cTACE) in terms of effectiveness [7-12]. Transarterial
radioembolisation (TARE) is an alternative technique [13,14] that
should be considered as part of a multimodal treatment approach
[15], but its emerging role is not yet well defined. Several cohort
studies have compared TARE with TACE, and the two procedures
seem to have a similar impact on survival and response [13,16,17].
A recent paper by Kollings et al. [18] prospectively compared the
two treatment options for intermediate-stage unresectable HCC
founding that both procedures have similar median progressionfree
survival and disease-control rates. TARE may be better tolerated
than TACE, and generally only one or two TARE procedures are
needed, whereas TACE requires multiple procedures. Moreover,
because of its ability to induce hypertrophy of the untreated liver
segments and to induce complete pathologic response, TARE has
been proposed as neoadjuvant treatment and as bridge treatment to
liver transplantation [19-22].
As described by Llovet and Lencioni, a subgroup analysis based
upon BCLC classification should be performed in all HCC trials.
Survival analysis on this basis could provide results stratification
and consequently a more focused and individualised approach
to treatment in advanced HCC [23,24]; however, to date, few
comparative studies have included this subgroup analysis.
A recent meta-analysis of two randomised trials, one comparing
TARE with TACE, the other comparing TARE followed by sorafenib
with sorafenib alone, concluded that the data were insufficient to
reach conclusions on the benefits of TARE in advanced HCC [25].
Another recent meta-analysis [26], which included both prospective
and retrospective studies, reported that TARE and TACE showed
similar effects in unresectable HCC patients in terms of OS, response
rate and safety profile [26].
The aim of this single-centre, retrospective study is to compare
the effectiveness of TARE and TACE in the treatment of unresectable
HCC and to evaluate the outcomes by the BCLC stage of the HCC.
Materials and Methods
Study setting and design
The study was a single-centre retrospective analysis of registry
data from 121 consecutive patients with HCC, who had not
undergone surgical intervention, were considered unsuitable for
surgery, and received TACE (conventional TACE [cTACE] or drugeluting-
beads TACE [DEB-TACE]) or TARE with yttrium 90 (Y-90)
resin microspheres between 2009 and 2014 in the IVth Department
of General Surgery of the Treviso Regional Hospital, Italy. Patients
were assigned to TACE or TARE treatment after a multidisciplinary
discussion involving the surgeon, the hepatologist, the interventional
radiologist, and the nuclear medicine specialist, according to standard
operating procedures at the centre. Patients selected for TARE were
those who were not ideal candidates for TACE: tumour larger than 5
cm, side branch portal thrombosis, or those who could benefit more
from the compensative hypertrophy to facilitate possible surgical
interventions. Our study protocol was compliant with the Declaration
of Helsinki for clinical trials and gained all necessary ethical review
board approval.
Patient selection and enrolment
The study included patients with a diagnosis of HCC confirmed by
biopsy or radiographic findings. Initial radiographic assessment was
performed by contrast-enhanced computed tomography (CT), and in
cases where the diagnosis was unclear, contrast-enhanced magnetic
resonance imaging (MRI) or ultrasound (US) was performed,
according to the criteria of the American Association for the Study
of Liver Disease (AASLD) and the European Association for the
Study of the Liver (EASL) [27,28]. Inclusion criteria were: untreated
HCC unsuitable for surgical treatment; ECOG performance status
≤2; written informed consent; adequate contraception in women
with childbearing potential; total bilirubin level <2.0 mg/dl; serum
creatinine <2 mg/dl; albumin >2.0 g/dl; Aspartate Aminotransferase
(AST) and Alanine Aminotransferase (ALT) <270 U/l; platelet
count >50,000; absence of thrombosis in the main portal vein;
absence of extra-hepatic metastasis; exclusion from orthotopic liver
Transplantation For Clinical Conditions (for TACE-group patients).
Exclusion criteria were any of the following: patients aged <18 or >90
years; patients weighing <50 or >120 kg; patients with severe, organspecific
disorders (e.g., liver or renal failure, acute pancreatitis);
uncorrectable blood shunt to the intestine and/or lungs (TAREgroup);
pregnancy and/or lactation; history of hypersensitivity to
the investigational product or to any drug with similar chemical
structure or to any compound present in the pharmaceutical form
of the investigational product. Patients were categorised according to
their BCLC tumour stage.
Study procedures and treatments
Written informed consent for the procedures was obtained from
all patients. All procedures were performed under local anaesthesia,
antibiotic prophylaxis and anti-emetic drugs. For patients scheduled
for TARE, a technetium-99 (99mTc)-labelled macroaggregated
albumin (MAA) scan was performed before the procedure to assess
the vascular anatomy of the liver and the presence of gastrointestinal
and/or lung shunts, and to calculate the administered dose. To
be eligible for the procedure, arteriovenous blood shunting to the
lungs had to be less than 20%, and patients were not allowed to have
shunts that would allow microspheres to reach the gastrointestinal
tract. TARE was performed with biocompatible resin microspheres
containing Y-90 (SIR-Spheres®; Sirtex Medical Limited; Australia).
The resin microspheres have an average diameter of 35 μm (range,
20–60 μm) and are supplied in vials (5 ml) containing 40–80
million spheres with a specific activity of 3 GBq of Y-90 (activity per
microsphere is 50 Bq at the time of calibration). The administered dose
was determined according to the percentage of tumour involvement
in the liver (>50%, 3.0 GBq; 25%–50%, 2.5 GBq; < 25%, 2.0 GBq). The
dose was further reduced if the lung shunt was ≥10% (maximum
40% reduction for shunt values between 10% and 20%) [29,30].
For patients scheduled for TACE, angiography was performed
before embolisation to map vascular liver anatomy, search for
arteriovenous shunts, and to identify the arterial feeders of the
tumour. TACE was performed by non-selective, selective or superselective
catheterisation of the hepatic arteries, feeding the lesions
using coaxial microcatheters. In the patients treated with cTACE,
a mixture of ethiodized oil (Lipiodol® Ultra Fluid; USA) and
farmorubicin (Pfizer, Italy) was injected, followed by embolisation
with gelatin sponge particles (Gelfoam®, Pfizer, Italy). Dose selection
was based on a cTACE treatment regimen of 50 mg/m2 farmorubicin per procedure (regardless of tumour size). For patients receiving
DEB-TACE, beads (DC Bead®, Biocompatibles; UK) were loaded with
doxorubicin (Pfizer, Italy) and mixed with nonionic contrast medium.
The doxorubicin dose was 50 mg/m2 (regardless of tumour size) and
beads used were 100–300 μm in diameter. Both in the cTACE and
in the DEB-TACE groups, the entire dose was administered in each
patient. The embolisation endpoint was reached when the arterial
feeders of the HCC were no longer identifiable and a reflux of the
contrast medium was detected. The flow was revaluated after 5 min
to demonstrate that it had stopped. If flow to the tumour was still
present after the entire TACE dose had been delivered, additional
embolics (Spongostan®, ETHICON, US) were used to reach the
endpoint. Post-procedural pain and fever were managed individually
with nonsteroidal anti-inflammatory drugs or opioids.
Assessments
Baseline laboratory evaluations included; alpha-feto protein
(AFP), total bilirubin, ALT, AST, international normalised ratio
(INR), platelet count, total albumin and the presence of aetiologic
viral markers (HCV, HBV), ChildPugh score. Cross-sectional
imaging studies (e.g. CT scan and/or MRI were routinely conducted
at the time of diagnosis. Minimum follow-up time was 24 months.
Radiological response was evaluated by contrast-enhanced CT after
1 or 2 months for TACE patients and after 3 months for TARE
patients. Radiological response was evaluated by modified Response
Evaluation Criteria in Solid Tumours (mRECIST) [24]. According to
the radiological findings of local progression at follow-up, the decision
to re-treat the patients or to execute other treatments was considered
taking into account the liver function and the performance status of
each patient.
Study endpoints
The primary study endpoint was overall survival (OS). Secondary
endpoints were survival by BCLC stage.
Statistical analysis
Statistical analysis was performed with SPSS software, version
13.0. The student T–test was used in comparisons of the patients’
baseline characteristics, the chi-square test was used for the categorical data analysis, the Cox regression test was used for the
multivariate analysis, and survival curves were determined by the
Kaplan-Meier method. Survival curves were compared between
treatment groups using the Log-Rank and Wilcoxon tests. A Cox
regression analysis was performed with BCLC stage, treatment
modality, age, mean tumour size, HCC aetiology, Child-Pugh score
and previous treatments for HCC as covariates. The ‘enter’ method
(not reported) was applied for each regression and a backward
stepwise logistic regression procedure was applied to verify the model
only for significant variables (significance level of < 0.1). In view of the
relatively small number of patients, a conservative p value of < 0.1 was
assumed for statistical significance.
Table 1
Table 2
Table 3
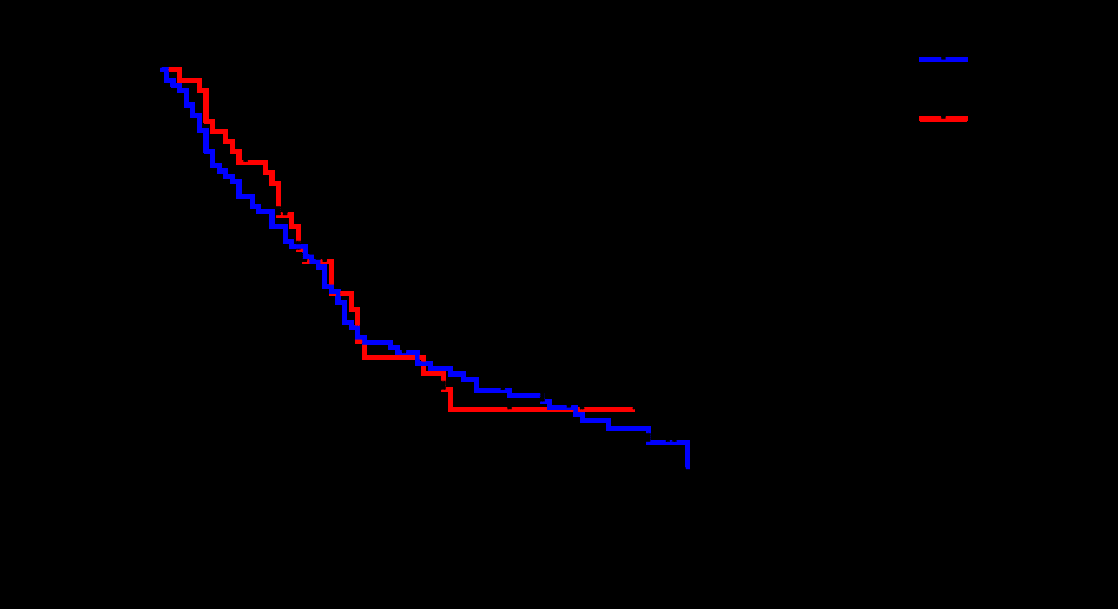
Figure 1
Figure 1
Kaplan-Meier plot of survival in patients with advanced HCC by
treatment modality (TARE or TACE).
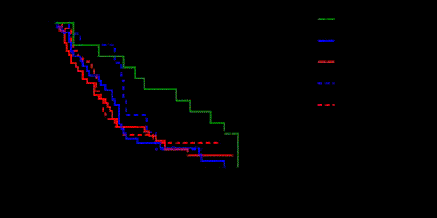
Figure 2
Figure 2
Kaplan-Meier plot of survival in patients with advanced HCC by
treatment modality (TARE or TACE) and BCLC stage.
Results
Out of the 121 patients who met the inclusion criteria, 82 received TACE and 39 received TARE treatment. No patients were enrolled more than once. The patients’ baseline characteristics are summarised in Table 1. There were no significant differences in age, sex, tumour size, AFP, INR, total bilirubin and serum albumin between the two treatment groups. However, mean tumour size was larger in the TARE group than in the TACE group (p=0.060), no patients in the TARE group had BCLC stage A disease, and a markedly higher proportion of patients receiving TARE had unilobar disease than in the TACE group (Table 1). Mean OS in the TARE group was 24.05±2.48 months (95% confidence interval [CI], 19.03– 29.08) and median survival time was 21.00 months (95% CI, 18.00- 26.00), compared with 27.39±2.47 months (95% CI, 22.47–32.31) and 23.50 months (95% CI, 17.00-28.00), respectively, in the TACE group (Figure 1). The survival analysis by Log-Rank (p=0.769) and Wilcoxon (p=0.526) tests showed no significant difference in the OS between TACE and TARE (Figure 1). Analysis of the survival curves according to treatment modality and BCLC status (excluding BCLC stage A as there were no patients with BCLC stage A disease in the TARE group) by Log-Rank (p=0.037) and Wilcoxon tests (p=0.024) showed statistically significant differences in survival rates, for each comparison (Figure 2). Mean survival times among TARE-treated patients and TACE-treated patients with BCLC stage B or stage C disease are summarised in Table 2. As the survival curves are not parallel, comparisons between the curves can be problematic; utilising the generalised, non-parametric, method of Wilcoxon can partially compensate for this. According to the Wilcoxon method, there was a statistically significant difference in OS rates (p=0.064) between the TARE and TACE groups, favouring TARE, in patients with BCLC stage B disease (Figure 2). There was no significant difference between OS rates in the TARE and TACE groups in patients with BCLC stage C disease (p=0.348; Figure 2). In a survival analysis, 24 months after the procedure, the mean survival time in TARE patients with BCLC stage B disease was 21.54±1.62 months (95% CI, 18.37– 24.71), compared with 17.07±1.51 months (95% CI, 14.11–20.02) in TACE patients with BCLC stage B disease (Log-Rank test, p=0.057; Wilcoxon test, p=0.068) (Table 3). At 24 months in patients with BCLC stage C disease the difference between the TARE and TACE groups did not reach statistical significance (Log-Rank test, p=0.718 and Wilcoxon test, p=0.353). The Cox regression analysis model was interrupted at the 9th step: the probability of death as a function of time was only significantly associated with the Child-Pugh score at baseline. The risk of death was significantly lower in patients with a Child-Pugh score than in those with a Child-Pugh score B (odds ratio [OR] 0.583; 95% CI, 0.344–0.987). Two patients in the TACE group died due to procedure-related complications: ascitic decompensation and oesophageal variceal rupture. No procedure-related deaths were reported with TARE.
Discussion
TACE is considered the standard treatment for patients with
unresectable HCC, its safety and efficacy has been investigated in
several studies [4-6] and DEBTACE is comparable to conventional
TACE in terms of effectiveness for treating HCC [7-12]
. TARE with Y-90 microspheres is an emerging treatment option for unresectable
HCC; however, few studies have compared its efficacy and safety
with that of TACE in this indication. A recent meta-analysis, which
included only randomised clinical trials, concluded that there is
insufficient evidence to assess the effectiveness and safety of TARE
in the treatment of unresectable HCC [25]. This study did not show
statistically significant differences in OS between TARE and TACE
for the study population as a whole, and results were similar to those
reported in previously published studies [13,16,17,26]. However, the
TARE and TACE cohorts were heterogeneous: TARE patients mostly
had unilobar disease, had larger tumours and none had BCLC stage
A disease (Table 1). To address this potential bias, we performed subanalyses
following BCLC classification for patients at stages B and
C. In our analysis, we showed a statistically significant difference in
OS between TARE and TACE in patients with BCLC stage B disease,
suggesting an improved survival with TARE in patients with a better
prognosis. We found only one other study which performed a survival
sub-analysis following BCLC criteria with a large sample size; Salem
et al. demonstrated a statistically significant difference in survival
in patients at BCLC stage C that favoured TARE [13]. We found
another study by Pitton et al. [31] considering almost exclusively
patients in group B, but the absence of significant showed results is
probably due to the small patient’s sample. Despite the difference
in BCLC stages between the studies, both our results and those of
Salem and colleagues suggest that including BCLC stage as a selection
criterion for TARE or TACE for unresectable advanced HCC could
be key in improving survival by a more focused and individualised
approach to treatment, as well as achieving better management of
healthcare resources. The study has a number of limitations and
potential sources of bias. The major source of bias is the retrospective
design of the study. Other sources of bias are the small sample size
and heterogeneity between patients enrolled in the TARE and TACE
arms. Patients originally selected for TARE were those with larger,
unilobar tumours, in order to exploit the compensative hypertrophy
observed after TARE procedures, thereby to facilitate possible surgical
interventions [19,20].
Conclusion
Our study suggests that overall TARE and TACE are similarly effective in the treatment of patients with unresectable advanced HCC. A subgroup analysis, however, showed enhanced survival with TARE compared with TACE in patients with BCLC stage B HCC. Future prospective studies, designed to be more with adequate follow-up, could further clarify the differences between TARE and TACE in the treatment of HCC and may produce objective data to support the use of TARE in this indication.
References
- Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55(2):74-108.
- El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterol. 2007;132(7):2557-76.
- Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012;379(9822):1245-55.
- Llovet JM, Real MI, Montana X, Planas R, Coll S, Aponte J, et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet. 2002;359(9319):1734-9.
- Lo CM, Ngan H, Tso WK, Liu CL, Lam CM, Poon RT, et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology. 2002;35(5):1164-71.
- Molinari M, Kachura JR, Dixon E, Rajan DK, Hayeems EB, Asch MR, et al. Transarterial chemoembolisation for advanced hepatocellular carcinoma: results from a North American cancer centre. Clin Oncol (R Coll Radiol). 2006;18(9):684-92.
- Massani M, Stecca T, Ruffolo C, Bassi N. Should we routinely use DEBTACE for unresectable HCC? cTACE versus DEBTACE: a single-center survival analysis. Updates Surg. 2017;69(1):67-73.
- Facciorusso A, Licinio R, Muscatiello N, Di Leo A, Barone M. Transarterial chemoembolization: evidences from the literature and applications in hepatocellular carcinoma patients. World J Hepatol. 2015;7(16):2009-19.
- Golfieri R, Giampalma E, Renzulli M, Cioni R, Bargellini I, Bartolozzi C, et al. Randomised controlled trial of doxorubicin-eluting beads vs. conventional chemoembolisation for hepatocellular carcinoma. Br J Cancer. 2014;111(2):255-64.
- Facciorusso A, Mariani L, Sposito C, Spreafico C, Bongini M, Morosi C, et al. Drug-eluting beads versus conventional chemoembolization for the treatment of unresectable hepatocellular carcinoma. J Gastroenterol Hepatol. 2016;31(3):645-53.
- Sacco R, Bargellini I, Bertini M, Bozzi E, Romano A, Petruzzi P, et al. Conventional versus doxorubicin-eluting Bead transarterial chemoembolization for hepatocellular carcinoma. J Vasc Interv Radiol. 2011;22(11):1545-52.
- Gao S, Yang Z, Zheng Z, Yao J, Deng M, Xie H, et al. Doxorubicin-eluting bead versus conventional TACE for unresectable hepatocellular carcinoma: a meta-analysis. Hepatogastroenterology. 2013;60(124):813-20.
- Salem R, Lewandowski RJ, Kulik L, Wang E, Riaz A, Ryu RK, et al. Radioembolization results in longer time-to-progression and reduced toxicity compared with chemoembolization in patients with hepatocellular carcinoma. Gastroenterology. 2011;140(2):497-507.
- Hilgard P, Hamami M, Fouly AE, Scherag A, Muller S, Ertle J, et al. Radioembolization with yttrium-90 glass microspheres in hepatocellular carcinoma: European experience on safety and long-term survival. Hepatology. 2010;52(5):1741-9.
- Coldwell D, Sangro B, Wasan H, Salem R, Kennedy A. General selection criteria of patients for radioembolization of liver tumors: an international working group report. Am J Clin Oncol. 2011;34(3):337-41.
- El Fouly A, Ertle J, El Dorry A, Shaker MK, Dechene A, Abdella H, et al. In intermediate stage hepatocellular carcinoma: radioembolization with yttrium 90 or chemoembolization? Liver Int. 2015;35(2):627-35.
- Carr BI, Kondragunta V, Buch SC, Branch RA. Therapeutic equivalence in survival for hepatic arterial chemoembolization and yttrium 90 microsphere treatments in unresectable hepatocellular carcinoma: a two-cohort study. Cancer. 2010;116(5):1305-14.
- Kolligs FT, Bilbao JI, Jakobs T, Iñarrairaegui M, Nagel JM, Rodriguez M, et al. Pilot randomized trial of selective internal radiation therapy vs. chemoembolization in unresectable hepatocellular carcinoma. Liver Int. 2015;35(6):1715-21.
- Vouche M, Lewandowski RJ, Atassi R, Memon K, Gates VL, Ryu RK, et al. Radiation lobectomy: time-dependent analysis of future liver remnant volume in unresectable liver cancer as a bridge to resection. J Hepatol. 2013;59(5):1029-36.
- Gaba RC, Lewandowski RJ, Kulik LM, Riaz A, Ibrahim SM, Mulcahy MF, et al. Radiation lobectomy: preliminary findings of hepatic volumetric response to lobar yttrium-90 radioembolization. Ann Surg Oncol. 2009;16(6):1587-96.
- Kulik LM, Atassi B, van Holsbeeck L, Souman T, Lewandowski RJ, Mulcahy MF, et al. Yttrium-90 microspheres (TheraSphere) treatment of unresectable hepatocellular carcinoma: downstaging to resection, RFA and bridge to transplantation. J Surg Oncol. 2006;94(7):572-86.
- Riaz A, Kulik L, Lewandowski RJ, Ryu RK, Giakoumis Spear G, Mulcahy MF, et al. Radiologic-pathologic correlation of hepatocellular carcinoma treated with internal radiation using yttrium-90 microspheres. Hepatology. 2009;49(4):1185-93.
- Llovet JM, Di Bisceglie AM, Bruix J, Kramer BS, Lencioni R, Zhu AX, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008;100(10):698-711.
- Lencioni R, Llovet JM. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis. 2010;30(1):52-60.
- Abdel-Rahman OM, Elsayed Z. Yttrium-90 microsphere radioembolisation for unresectable hepatocellular carcinoma. Cochrane Database Syst Rev. 2016;2:CD011313.
- Facciorusso A, Serviddio G, Muscatiello N. Transarterial radioembolization vs. chemoembolization for hepatocarcinoma patients: A systematic review and meta-analysis. World J Hepatol. 2016;8(18):770-8.
- Bruix J, Sherman M, American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53(3):1020-2.
- European Association for the Study of the Liver, European Organisation for Research and Treatment of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. Euro J Cancer.2012;48(5):599-641.
- Sangro B, Bilbao JI, Boan J, Martinez-Cuesta A, Benito A, Rodriguez J, et al. Radioembolization using 90Y-resin microspheres for patients with advanced hepatocellular, carcinoma. Int J Radiat Oncol Biol Phys. 2006;66(3):792-800.
- Ho S, Lau WY, Leung TW, Chan M, Ngar YK, Johnson PJ, et al. Partition model for estimating radiation doses from yttrium-90 microspheres in treating hepatic tumours. Eur J Nucl Med. 1996;23(8):947-52.
- Pitton MB, Kloeckner R, Ruckes C, Wirth GM, Eichhorn W, Wörns MA, et al. Randomized comparison of selective internal radiotherapy (SIRT) versus drug-eluting bead transarterial chemoembolization (DEB-TACE) for the treatment of hepatocellular carcinoma. Cardiovasc Intervent Radiol. 2015;38(2):352-60.