Perspective
Total Pancreatectomy and Intrahepatic Islet Autotransplantation: Therapy for Chronic, Painful Pancreatitis
R. Paul Robertson*
Department of Endocrinology, Universities of Washington and Minnesota, USA
*Corresponding author: R. Paul Robertson, Department of Endocrinology, Universities of Washington and Minnesota, 720 Broadway, Seattle, WA 98122, USA
Published: 28 Jun, 2017
Cite this article as: Paul Robertson R. Total
Pancreatectomy and Intrahepatic
Islet Autotransplantation: Therapy for
Chronic, Painful Pancreatitis. Clin Surg.
2017; 2: 1521.
Perspective
From my perspective, it seems very odd that the surgical procedure of total pancreatectomy
and autoislet transplantation (TPIAT) for patients suffering from chronic pancreatitis is almost
unknown to the general medical community. TPIAT was first performed in 1980 and was
immediately successful [1,7]. Now, over 35 years later, when I speak to medical audiences about
this procedure, physicians and nurses almost to a person are amazed at what I tell them. Even
gastroenterologists acknowledge they have very little knowledge about TPIAT although many will
say they’ve heard of it. Still, the transplantation literature is replete with publications from all over
the world that document the high success rate of TPIAT. It is true that internists and nurses and
even general surgeons are not likely to put transplantation literature on the top of their reading lists,
but even gastro-intestinal surgeons, who do read transplantation literature, in my experience, seem
refractory to using TPIAT. They seem to favor repeated limited pancreatic resection of diseased
areas. It’s a puzzle to me. More importantly, it’s a real problem for patients who elect to have the
procedure. Many of them tell me it’s a tremendous problem for them when they return home from
the transplant center where their TPIAT was performed because they cannot find a physician in
the general community who is comfortable managing their post-operative course. It is common
for autoislet recipients to be met with incredulity when they seek care at an emergency room or
their local medical clinics for unrelated problems. They tell me the usual response they receive upon
giving their surgical history to local caregivers is “That’s impossible. You cannot live without your
pancreas. And you say somebody put your islets in your liver? That’s crazy!”
I write this Perspective because I am concerned about the lack of general information about the
use of TPIAT for chronic, painful pancreatitis and on the chance that an endocrine--based surgical
journal might be just the place to make this information more available to the general surgical/
medical world. My experience over the past 30 years has been University of Minnesota-based
and has involved post-transplantation pancreatic research with many TPIAT patients. I will use
conclusions from peer-reviewed, published references from my and others’ groups [2-15] to try
to make the case that TPAIT should be used much more often and much earlier for patients with
chronic, painful pancreatitis than is currently the case. Let’s consider four questions [16-21].
Who are candidates for TPIAT? The criteria for TPIAT at the University of Minnesota are:
1) Chronic pancreatitis refractory to medical treatment and endoscopic interventions.
2) Narcotic dependence and/or significantly impaired quality of life.
3) Absence of medical/ psychosocial contraindications to TPIAT.
4) The patient is either non-diabetic or has C-peptide positive diabetes.
5) The patient is willing to accept risk of diabetes to obtain pain relief.
Successful candidates spend a week at the transplant center learning about the procedure, its
risks, and its possible outcomes, including death. During this visit extensive data are gathered by
the transplant team with a major focus on the length and activity of pancreatitis; history of narcotic
use to control pain; objective quality of life measures, including the ability to work, attend school
and to provide childcare; use of tobacco or alcohol; and other concurrent illnesses. This information
is considered by a team that includes transplantation surgeons, endocrinologists specializing
in diabetes, gastroenterologists specializing in pancreas diseases and endoscopy, nurse and nutrition specialists in transplantation; and psychiatrists trained in management of chronic disease and long-term narcotic use. Then the
decision to accept or not to accept candidates is made by consensus of
the entire transplant team. The ideal candidate is one who has chronic
and recurring abdominal pain; long-term daily narcotic use to control
pain; weight loss secondary to bowel dysfunction consistent with
chronic pancreatitis and use of pancreatic enzymes; and a generally
poor quality of life.
What is surgically involved in the TPIAT procedure? Very
generally and briefly stated, the patient is admitted to the center’s
hospital, taken to the operating room for a total pancreatectomy with
a proximal duodenum sparing approach that commonly involves a
Roux-en-Y procedure, and is usually accompanied by resection of the
spleen. The resected pancreas is taken to the center’s islet isolation
laboratory where it is treated with a collagenase-type product and
then cut into small pieces that are placed into a digestive apparatus
for separation and isolation of pancreatic islets. The islets are purified
by centrifugation and taken back to the patient who usually is still in
the operating room where the islets are infused into the portal vein or
one of its tributaries. The portal circulation carries the islets into the
liver for distribution throughout the organ with careful monitoring
of hepatic portal blood pressure. At this point the transplantation
procedure is over, the abdomen is closed, and then the patient is
taken to a post-operative care unit in the hospital.
What are the clinical outcomes in terms of patient survival, pain
relief, islet survival, and prevention of diabetes? Patient survival in a
University of Minnesota series of over 400 TPIAT autoislet recipients
reviewed by Sutherland reports a patient survival rate of 1.2%. Pain
relief was experienced in 80% to 86% of patients by 6 months to 3
years post-transplant. 50% of patients withdrew from narcotics in the
first post-transplant year. Health-related quality of life (by objective
measurements, such as the SF-36) improved. The key determinant in
clinical outcomes is the amount of islets that are recovered from the
resected pancreas. If a total of 350,000 islets or 5,000 islets/kg body
weight (roughly one-third of the number of islets in a normal, healthy
pancreas) are infused into the patient’s liver, the 2-year success rate
for being free of insulin treatment and maintaining a HbA1c levels
< 7.0% is approximately 70%. The duration of chronic pancreatitis is
the key issue in recovering a sufficient number of islets because with
time the acinar inflammation involves the islets and destroys them.
What is the functional status of intrahepatic islets posttransplant?
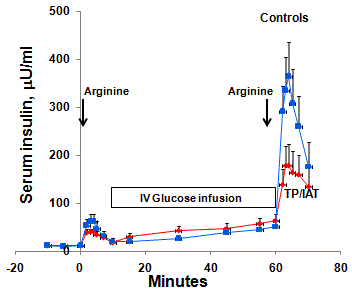
Beta cell function, as measured by arginine-stimulation
of insulin secretion, is quantitatively less than 0bserved in normal
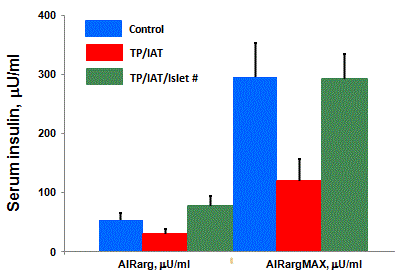
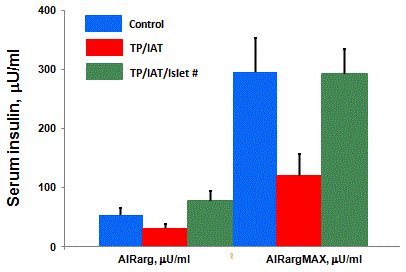
control subjects [20] (Figure 1). However, when these data are
corrected for the fact that the amount of islets transplanted in the
liver are substantially less than the number of islets contained in
the pancreases of normal controls, insulin secretion is of normal
magnitude [20] (Figure 2). Alpha cell function in response to
arginine is quantitatively equal to that observed in normal subjects
[21]. However, the glucagon secretory response to hypoglycemia is
virtually absent in TPIAT patients [21] (Figure 3). This is not the case,
however, in TPIAT patients who received a portion of their islets in
non-hepatic sites. These patients had normal glucagon responses
to hypoglycemia. We have recommended the use of non-hepatic
sites, such as the peritoneal cavity or omental sacs, because those
recipients do have a normal glucagon response to hypoglycemia. This
issue is of more than casual interest because maintaining the normal
hypoglycemia-induced glucagon response is critical for TPIAT
patients who need partial replacement therapy with exogenous
insulin.
The main features of management of chronic pancreatitis that I
hope comes through by writing this Perspective is that the TPIAT procedure needs to be more fully embraced by the surgical/medical
community and it needs to be used much earlier in the disease than
is currently the case. Many patients have had their disease for over a
decade and have had many more endoscopic procedures for stenting
than seems reasonable. Most patients only get temporary relief
from pancreatic duct stenting. Other patients many undergo partial
pancreas resection to remove the diseased portions of the gland.
However, it has become obvious that surgery does not necessarily
prevent recurrence of chronic pancreatitis and it also hastens the
development of diabetes and limits the chances of recovering viable
islets when a TPIAT is performed. For these reasons, the more
reasonable approach seems to be an early, serious consideration of
TPIAT if recurrent chronic, painful pancreatitis continues more than
2 years. It is not likely to improve by using less effective treatment
for eight years more before turning to the more definitive treatment
of TPIAT that has been shown to be successful in relieving pain,
preventing diabetes, and improving quality of life.
Grant Support: NIH NIDDK RO-1 039994-27.
Figure 1
Figure 1
Circulating insulin responses to a pulse of intravenous arginine
before and during a concurrent infusion of glucose. The first response
(AIRarg) was augmented by the intravenous glucose infusion so that the
second response (AIRargMax) was greater and reflected insulin secretory
reserve. The responses in the TPIAT patients, who had fewer islets, were
significantly less than the controls [20].
Figure 2
Figure 2
The differences in responses shown in Figure 1 disappeared after
correcting the AIRarg and AIRargMax values from the TPIAT group for the
number of autoislets infused into their livers, which were approximately half
of the number of islets assumed to be in the controls’ pancreas [20].
Figure 3
Figure 3
Circulating glucagon responses during a hypoglycemic,
hyperinsulinemic clamp. TPIAT subjects who received autoislets in their liver
failed to secrete glucagon as the circulating glucose levels were lowered from
70 to 50 mg/dl. However, intact glucagon responses in a subgroup a 5 TPIAT
subjects who had received both intrahepatic and non-hepatic islets (mostly
intraperitoneal) did have appropriate glucagon responses to insulin-induced
hypoglycemia [21].
References
- Sutherland DE, Matas AJ, Najarian JS. Pancreatic islet cell transplantation. Surg Clin North Am. 1978;58(2):365-82.
- Morgan K, Owczarski SM, Borckardt J, Madan A, Nishimura M, Adams DB. Pain control and quality of life after pancreatectomy with islet autotransplantation for chronic pancreatitis. J Gastrointest Surg. 2012;16(1):129-33.
- Takita M, Naziruddin B, Matsumoto S, Hirofumi N, Masayuki S, Daisuke C, et al. Implications of pancreatic image findings in total pancreatectomy with islet auto transplantation for chronic pancreatitis. Pancreas. 2011;40(1):103-8.
- Argo JL, Contreras JL, Wesley MM, Christein JD. Pancreatic resection with islet cell autotransplant for the treatment of severe chronic pancreatitis. Am Surg. 2008;74(6):530-6.
- Pezzelli R, Morselli Labate AM, Fantini L, Corinaldesi R. Quality of life and clinical indicators for chronic pancreatitis patients in a 2-year follow-up study. Pancreas. 2007;34(2):191-6.
- Braasch JW, Vito L, Nugent FW. Total pancreatectomy of end-stage chronic pancreatitis. Ann Surg. 1978;188(3):317-22.
- Najarian JS, Sutherland DE, Baumgartner D, Burke B, Rynasiewicz JJ, Matas AJ, et al. Total or near total pancreatectomy and islet autotransplantation for treatment of chronic pancreatitis. Ann Surg. 1980;192(4):526-42.
- Wahoff DC, Papalois BE, Najarian JS, Kendall DM, Farney AC, Leone JP, et al. Autologous islet transplantation to prevent diabetes after pancreatic resection. Ann Surg. 1995;222(4):562-75;discussion 575-9.
- Sutherland DE, Radosevich DM, Bellin MD, Hering BJ, Beilman GJ, Dunn TB, et al. Total pancreatectomy and islet autotransplantation for chronic pancreatitis. J Am Coll Surg. 2012;214(4):409-24.
- White SA, Davies JE, Pollard C, Swift SM, Clayton HA, Sutton CD, et al. Pancreas resection and islet autotransplantation for end-stage chronic pancreatitis. Ann Surg. 2001;233(3):423-31.
- Garcea G, Weaver J, Phillips J, Pollard CA, Ilouz SC, Webb MA, et al. Total pancreatectomy with and without islet cell transplantation for chronic pancreatitis: a series of 85 consecutive patients. Pancreas. 2009;38(1):1-7.
- Fontana I, Arcuri V, Tommasi GV, Viviani GL, Pellicci R, Bottino R, et al. Long-term follow-up of human islet autotransplantation. Transplant Proc. 1994;26(2):581.
- Sutton JM, Schmulewitz N, Sussman JJ, Smith M, Kurland JE, Brunner JE, et al. Total pancreatectomy and islet cell autotransplantation as a means of treating patients with genetically linked pancreatitis. Surgery. 2010;148(4):676-85.
- Berney T, Rüdisühli T, Oberholzer J, Caulfield A, Morel P. Long-term metabolic results after pancreatic resection for severe chronic pancreatitis. Arch Surg. 2000;135(9):1106-11.
- Dixon J, DeLegge M, Morgan K, Adams D. Impact of total pancreatectomy with islet cell transplant on chronic pancreatitis management at a disease-based center. Am Surg. 2008;74(8):735-8.
- Robertson RP, Lanz KJ, Sutherland DE, Kendall DM. Prevention of diabetes for up 13 years by autoislet transplantation after pancreatectomy for chronic pancreatitis. Diabetes. 2001;50(1):47-50.
- Pyzdrowski KL, Kendall DM, Halter JB, Nakhleh RE, Sutherland DE, Robertson RP. Preserved insulin secretion and insulin independence in recipients of islet autografts. N Engl J Med. 1992;327(4):220-6.
- Kendall DM, Teuscher AU, Robertson RP. Defective glucagon secretion during sustained hypoglycemia following successful islet allo- and autotransplantation in humans. Diabetes. 1997;46(1):23-7.
- Robertson RP. Islet transplantation a decade later and strategies for filling a half-full glass. Diabetes. 2010;59(6):1285-91.
- Robertson RP, Bogachus LD, Oseid E, Parazzoli S, Patti ME, Rickels MR, et al. Assessment of β-cell mass and a- and β-cell survival and function by arginine stimulation in human autologous islet recipients. Diabetes. 2015;64(2):565-72.
- Bellin MD, Parazzoli S, Oseid E, Bogachus LD, Schuetz C, Patti ME, et al. Defective glucagon secretion during hypoglycemia after intrahepatic but not nonhepatic islet autotransplantation. Am J Transplant. 2014;14(8):1880-6.