Review Article
Recent Advances in the Diagnosis and Treatment of Presacral Tumours
Konstantinos Paschos*, Evelina Tsiomita, Maria Sachanidou and Anestis Chatzigeorgiadis
Department of General Surgery, General Hospital of Drama, Greece
*Corresponding author: Konstantinos Paschos, Department of General Surgery, General Hospital of Drama, Terma Ippokratous st, GR- 66100, Drama, Greece
Published: 07 Jun, 2017
Cite this article as: Paschos K, Tsiomita E, Sachanidou M,
Chatzigeorgiadis A. Recent Advances
in the Diagnosis and Treatment of
Presacral Tumours. Clin Surg. 2017; 2:
1496.
Abstract
Presacral or retrorectal tumours (PST) s is rare lesions, which range from cysts (usually benign) to
malignant masses, invading surrounding tissues in the pelvis. According to the tumour’s origin,
characteristics and behavior, PSTs are classified as follows: congenital, neurogenic, osseous,
miscellaneous, and inflammatory. They present variable signs and symptoms, a fact that may delay
their diagnosis and/or cause an inappropriate treatment with bad prognosis. Nonetheless, modern
imaging modalities, such as CT and MRI may discover the true nature of a lesion, as well as whether it
infiltrates neighboring viscera. In that way, they substantially contribute to a definitive diagnosis and
a correct preoperative planning. While neoadjuvant chemotherapy and postoperative radiotherapy
may offer benefits in certain PSTs, surgical resection is the primary therapeutic management. The
anterior, posterior and combined abdominosacral approach are applied, according to tumour
location and its relation to adjacent structures. In general, clinicians should maintain high clinical
suspicion of the disease to avoid delayed or false diagnosis. Multidisciplinary approach is crucial for
a prompt and accurate treatment.
Keywords: Presacral; Retrorectal; Retroperitoneum; Rectum; Sacrum
Introduction
Presacral tumours (PST) s or retrorectal as they are also termed, represent heterogeneous and
rare lesions, which may range from benign cysts to malignant masses that can infiltrate surrounding
pelvic tissues and organs. Their incidence in the general population worldwide is unknown, because
the majority of reports concerning these neoplasms come from tertiary medical centers [1]. Notably,
the only case series on the disease not from a referral center was published by Uhlig and Johnson
in 1975, demonstrating an incidence of two PSTs per year in the metropolitan population of
Portland, USA [2]. Furthermore, Jao et al. [3] from Mayo Clinic concluded in 1985 that the disease is diagnosed in 1 patient for every 40000 hospital admissions.
PSTs are usually deficient of signs and symptoms, until they reach a considerable size, leading
to a delayed diagnosis and thus the involvement of other sensitive structures and bad prognosis.
However, modern imaging modalities, new surgical approaches and the progress in adjuvant therapy
have contributed to a better management of PSTs, and decreased morbidity [4]. The following text
attempts to summarize recent medical knowledge on these lesions through an in depth analysis of
up-to-date medical literature.
Anatomy-Physiology
The presacral or retrorectal space represents the continuation of the retroperitoneum into
the pelvis. This potential space is located between two anatomical structures, the presacral fascia
of the sacrum (Waldeyer’s fascia) and the parietal peritoneum of the posterior abdominal wall.
Its boundaries are formed anteriorly by the mesorectum, posteriorly by the anterior aspect of
the sacrum, superiorly by the peritoneal reflection and inferiorly by the retrosacral fascia [1,5].
Laterally, the presacral area extends to the ureters, the internal iliac vessels, the lateral sacral
artery, the sympathetic trunk, the hypogastric nerves, and the inferior hypogastric plexus at the
lower levels. Embryologically, the presacral space is the area where fusion of the hindgut and the
neuroectoderm of the spinal cord occur. In adults, this site contains retrorectal fat, loose connective
tissue, lymph nodes, the median sacral vessels, the superior rectal vessels, as well as sympathetic and
parasympathetic branches [5,6].
In case these neural and vascular structures are harmed or injured, the rectoanal physiology
is seriously affected and may cause substantial musculoskeletal and/or neurologic morbidity.Notably, if the S3 nerve is injured bilaterally, the external sphincter malfunctions, not contracting when the rectum is dilated, leading
to various degrees of incontinence. Interestingly, anorectal function
is maintained if all unilateral nerve roots are sacrificed. Moreover,
during sacrectomy, pelvic stability is maintained if greater than half of
the body of S1 vertebra remains intact. However, if the site is radiated
preoperatively, spinopelvic stability may be seriously compromised
[1,7,8].
Classification
Various tumours, either congenital or acquired, may arise
from and within the structures of the presacral space. A variety of
classification systems attempting to categorize PSTs have been
proposed by different authors, although none has been unanimously
accepted [9]. The classification system first described by Uhlig and
Johnson is the most frequently used one. According to the tumour’s
origin this system includes the following categories: congenital,
neurogenic, osseous, inflammatory and miscellaneous [2]. Another
important aspect for tumour classification is its benign or malignant
behavior. The latter is usually encountered in solid lesions. Lev-
Chelouche et al. [10] have classified PSTs into congenital versus
acquired and benign versus malignant. Metastatic and locally
advanced colorectal and genitourinary neoplasms are usually not
included in the category of presacral tumours (Table I).
Figure 1
Table 1
Clinical Manifestation
Congenital
Congenital tumours develop from embryonic tissue remnants and
are usually benign. They may appear either as cystic or solid lesions.
The former include developmental cysts and anterior meningoceles,
while the latter include teratomas, sacrococcygeal chordomas, and
adrenal rest tumours. Congenital tumours are the most common
retrorectal lesions, 55% to 70% of all lesions in the presacral area, and
the patients are usually females [11,12].
1. Cystic
2. Developmental Cysts
Epidermoid and dermoid cysts, enterogenous cysts (rectal
duplication cysts) and cystic hamartomas (tailgut cysts) fall under
this category. They represent 60% of all congenital presacral lesions.
Embryologically, they may develop from any of the three germ layers
[11]. Epithelium always lines the developmental cysts (squamous for
dermoid and epidermoid cysts, cuboidal, transitional or columnar
epithelium for enterogenous cysts and cystic hamartomas). These
lesions are usually multilocular and their walls are surrounded by
fibres of disorganized smooth muscle [12]. Developmental cysts
may be complicated with hemorrhage, infection or malignant
degeneration. The treatment of choice is complete surgical excision
of the cystic epithelial lining [13].
Dermoid and epidermoid cysts: Usually, benign they mainly
affect middle-aged females. They arise when the ectodermal tube fails
to close normally. Both types are constituted of stratified squamous
epithelium. Moreover, dermoid cycts have skin appendages (sweat
glands, sebaceous cysts, hair follicles), which is a useful feature for
their differential diagnosis from epidermoid cycts. Dermoid, as well
as epidermoid cysts, may communicate with the skin; if this is the
case, a postanal dimple or sinus may also exist, representing a residual
connection to the embryonic ectoderm [4,6]. Up to 30% of these
cysts may be infected, impeding their differentiation from a complex
perirectal abscess [11].
Enterogenous cysts (Duplication): They develop from
sequestration of the embryonic hindgut, which classifies them
as endodermal. Columnar, transitional, cuboidal or squamous
epithelium may line them. They often appear as multilobular, with a
single dominant cyst and multiple satellite ones. Diagnostic criteria
are continuity with the rectum, a well-defined muscular wall with a
myenteric plexus and mucosal lining. Similarly with epidermoid and
dermoid cysts, enterogenous cysts are more common in females and
may become infected. Commonly, they are benign in nature, but
malignant transformation should not be ruled out [1,6].
Cystic hamartomas (Tailgut cysts): They arise from the
hindgut, due to regression failure of a portion of the embryonic tail.
Although multicystic and well-circumscribed in appearance, they
do not have a capsule. Usually, a cystic hamartoma appears like a
soft mass with thick walls filled with mucous. Histologically, they
are similar to the intestinal tract, including squamous, columnar or
transitional epithelium. These lesions mainly affect females and are
benign in nature; however, malignant degeneration (most commonly
adenocarcinoma) has been reported [14-16].
Anterior sacral meningocele: These rare lesions (5%) develop
through a defect in the anterior sacrum that allows herniation of the
dural sac [17]. This defect may appear in combination with presacral
lipomas and/or cysts and may be associated with other congenital
abnormalities, such as spina bifida, tethered spinal cord, urinary tract
or anal malformations, and uterine or vaginal duplication [6]. In adults,
the female-to-male ratio is 6:1, while it is 2:1for children [18]. When
the sac contains neural elements, it is then called myelomeningocele
[19]. Anterior sacral meningocele is usually of sporadic occurrence,
but it also may be part of the hereditary Currarino syndrome. This
rare syndrome is caused by congenital caudal anomalies and its three
main characteristics (Currarino triad), are a deformity of the sacral
bone, malformations of the anus and rectum, and a presacral mass
[20]. It may also be encountered in conditions with duralectasia
such as Marfan’s syndrome and neurofibromatosis type 1, but this
is a very rare case [21]. The “scimitar sign” is pathognomonic of the
disease, and it describes a sacrum with a rounded concave border,
although without destruction of the bone on plain radiograph.
The continuity between the dural sac and subdural space means
that the sac also contains cerebrospinal fluid. An anterior sacral
meningocele may increase the pressure of the cerebrospinal fluid,
giving rise to typical presenting symptoms, such as headache and
defecation, associated with nausea and vomiting triggered by changes
in body position. Secondary symptoms arise from compression of
neighboring structures, such as constipation, urinary abnormalities
and lower back pain. Aspiration or biopsy of the lesion should not be
attempted, due to the risk of life-threatening meningitis, intracranial
hypotension syndrome or even death.
Teratomas: Presacral teratomas are rare in adult life, although
they represent the most common teratomas in the newborns. They
arise from totipotential cells, and as a result these tumors contain tissue
elements from all three germ layers (epithelium of the gastrointestinal
tract, respiratory tract, and nervous system). They may be cystic,
solid, or both, but most are cystic and benign [1,22]. Teratomas are
categorized as mature, immature, or malignant (teratocarcinoma).
The first contain recognizable epithelial or mesenchymal cells, while
the second are comprised of endodermal, mesodermal or ectodermal
elements. Germ cells are found in malignant teratomas, unlike the teratomas with malignant transformation, which include malignant
degenerated somatic cells [23]. Teratomas are more common in
female patients, with the female to male ratio being 10:1. In pediatric
patients, only 4% of the teratomas diagnosed at birth are malignant;
those diagnosed later in childhood tend to be aggressive and are linked
to poor prognosis. Generally, the risk of malignancy increases with
age. Teratomas in children are believed to harbor some association
with anomalies of the anorectum, urinary tract, and vertebrae. In
the adult population, around 30% of teratomas have elements of
malignancy at the time of resection, and there is a risk of 40% to
50% malignant transformation, which increases with incomplete
resection. Teratomas often adhere to the coccyx; in that case, en bloc
coccygectomy is the required surgical approach. Recurrence rate is
7.5% to 22%, and, when the coccyx is not respected, this rate may
increase up to 37%.
Solid
Sacrococcygeal chordomas: They arise from the embryonic
notochord and are the most common malignant PSTs (30 to 50% of all
chordomas occur in this area). They are usually found on the midline
cerebrospinal axis and are most commonly located at the sphenoclival
region and the sacrum [11,24]. Chordomas are more common
in males and are rarely diagnosed before adulthood. They tend to
grow slowly, but are locally invasive and give metastases (liver, bones
and lungs) in about 20% of cases. Their macroscopic appearance is
a lobulated, gelatinous mass, brownish-grey in color, commonly
with a pseudocapsule. Sometimes they may appear cystic, due to
intratumoural hemorrhage. Their characteristic histopathologic
finding is the presence of vacuolated and rich in mucin and glycogen
physaliphorous cells. Chordomas may cause either specific or
nonspecific symptoms, when the tumour has reached a considerable
size. By that time, resection on negative margins is much more
difficult. Local recurrence is high and 10-year survival rates from 9%
to 35% [11,25].
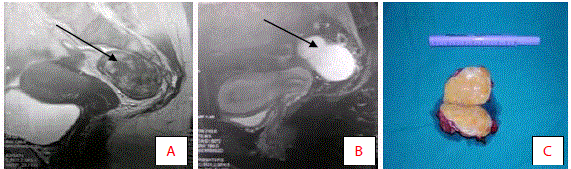
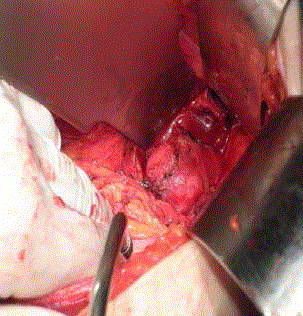
Figure 2
Figure 2
Anterior approach in a 52-year old female patient with a desmoid
presacral tumour (arrow) (authors’ archive).
Neurogenic Tumors
These arise from pelvic nerve roots or peripheral nerves and are the second most common PSTs, accounting for about 10% of all [12]. The majority (around 85%) is benign including neurofibromas, neurilemomas (schwannomas), and ganglioneuromas. Inversely, neuroblastomas, ganglioneuroblastomas, ependymomas, and malignant peripheral nerve sheath tumours (malignant schwannomas, neurofibrosarcomas and neurogenic sarcomas) fall under the category of malignant neurogenic lesions. Generally, they grow slowly and present non-specific symptoms. Therefore, they may be of large size at the time of diagnosis, leading to considerable blood loss intraoperatively and high complication rates. In case of symptoms, the route of the affected nerve determines the pain distribution and neurologic dysfunction [26-28].
Osseous
They may develop from bone, fibrous tissue, cartilage and marrow. They account for 10% of all PSTs and include benign and malignant lesions. Giant-cell tumour, osteoblastoma, and aneurismal bone cyst are benign, while osteogenic sarcoma, Ewing sarcoma, myeloma, and chondrosarcoma are malignant. They usually grow rapidly and are of significant size at the time of diagnosis; consequently, bone destruction or soft tissue calcification may usually be identified on plain radiograph. The lungs are their primary metastatic target and their overall prognosis is poor [6,12].
Inflammatory and Miscellaneous
Miscellaneous tumors represent 10% to 25% of all presacral ones. They include lipoma, fibroma, leiomyoma, hemangioma, endothelioma, desmoid (locally aggressive) (Figure 1), liposarcoma, fibrosarcoma, malignant histiocytoma, leiomyosarcomas, hemangiopericytoma, metastatic adenocarcinoma, and inflammatory tumours. Although Uhlig and Johnson originally classified inflammatory tumors as a separate category of presacral ones, they should be considered secondary reactions to foreign substances (e.g., foreign body granulomas from barium leaks or suture material), or extensions of infectious processes from either the abdomen or perirectal space (e.g., pelvic sepsis, Crohn disease, and perforated diverticulitis).
Clinical Manifestations
Symptoms
PSTs, especially benign ones, are either asymptomatic for a
long time, or they produce minimal or non-specific symptoms,
such as constipation, paradoxal diarrhea, rectal tenesmus and
sexual dysfunction. Most of the symptoms occur due to tumour
compression on the rectum or other neighboring structures [11].
Pain occurs primarily in malignant tumours [29], in male patients
older than 60 years [11] or in benign infected tumours [12]. The latter
may also cause perianal discharge or drainage. Kye BH and Macafee
DA report lower abdominal pain or discomfort as the most common
symptom at first visit [30,31], whereas Menteş BB et al. [23] describe
rectal pain and perirectal mass sensation as the main symptoms in
their respective case series. When nerve roots or the sacral plexus are
invaded, urinary incontinence or retention and bowel incontinence
may occur [12]. Obstructive labor by the presence of a PST has
been reported in women of reproductive age and this is an absolute
indication for surgical resection, even if the tumour is benign or
asymptomatic. Interestingly, some tumours produce more specific
symptoms. A constant aching of mild intensity in the lower lumbar,
pelvic and/or gluteal region is associated with chordomas, while an
anterior sacral meningocele is linked to headache during defecation
or intercourse, due to increased intracranial pressure.
Physical examination
PSTs are usually discovered randomly during routine physical
examination (pelvic or rectal). The latter may determine the cranial
extent of the lesion, as well as whether it is fixated or freely mobile,
and its relationship to other pelvic structures. On palpation, PSTs
usually feel soft and easy to compress. When tender, the tumor might
be an infected developmental cyst or a primary perirectal abscess that
extends supralevatorly. A thorough neurologic and muscoloskeletal
examination must be performed in order to reveal any neurologic
deficiency and document preoperative functional status [1,4,12].
Bimanual pelvic examination is very important in female patients
to exclude conditions like ovarian or uterine adenocarcinomas,
which are much more common conditions. In addition, complete
colonoscopy should be performed in all patients, aiming to discover a
synchronous colorectal adenocarcinoma.
Imaging
Plain radiographs
The role of plain radiographs has been limited by the use of
Computed Tomography (CT) scans and Magnetic Resonance
Imaging (MRI), as they are the imaging modalities of choice in
diagnosing PSTs nowadays. Plain radiographs are usually normal and
have little additional information to offer. However, they may reveal
osseous destruction of the sacrum, calcifications, and small bone
fragments or teeth in teratomas. The “scimitar sign”, as described
above, is pathognomonic for anterior sacral meningocele [4,6].
CT and MRI
The use of CT is to reveal any destruction of the bone cortex,
the solid or cystic nature of the lesion and whether tumour is
infiltrating any neighboring viscera. MRI, being more specific that
CT may provide a detailed view of the anatomical correlations and
the histology of the tumour [29], as well as neural tissue involvement
[32]. Although both CT and MRI can demonstrate the cystic or solid
nature of a lesion, it is difficult to determine its benign or malignant
characteristics [30]. However, smooth-walled cystic lesions on MRI
are normally benign, while solid or heterogeneous ones tend to be
malignant [33]. Although CT and MRI are useful for preoperative
planning, neither of them can provide a definitive diagnosis [34].
In general, anatomy and topography may be determined
preoperatively, using the findings yielded by MRI and CT. The aim
is to achieve optimal surgical planning and accurate-successful
surgical resection [35,36]. While MRI appears to play a superior role
in the diagnosis and preoperative planning of PSTs, both CT and
MRI should be used in a complementary and not mutually exclusive
manner [4,37].
Transrectal ultrasound
Another useful imaging tool in the diagnostic process is
transrectal ultrasonography (TRUS). Most importantly, this
modality presents a sensitivity of 100%, when combined with rigid
proctoscopy. TRUS may aid to determine whether a lesion is cystic or
solid, as well as whether rectal involvement is present. Normal TRUS
findings eliminate the possibility of a PST, with the precondition that
an experienced examiner is involved [6,29].
Other imaging techniques
Angiograms, venograms and fistulograms have occasionally
been reported to play a supplementary role in the diagnosis and
management of PSTs. In case there is tissue distortion due to mass
effect by the neoplasm, an angiogram and/or venogram may be
added to MRI (MR angiogram and venogram), in order to determine
vascular anatomy and involvement. In patients that present with a
chronically draining sinus, fistulograms may be useful to investigate
the possibility of a developmental cyst [38]. In any case, imaging
modalities aim to guide towards the correct surgical approach
(anterior, posterior, or combined), as well as to determine the
intraoperative extent of excision (local or en bloc resection).
Preoperative Biopsy
Preoperative biopsy in the management of PSTs appears to be
rather “controversial”, due to its potential complications and the
accuracy of modern imaging techniques [39]. Highlighting the risk
of infection of cystic lesions and needle-tract seeding with malignant
cells (in case of malignant tumors), surgeons and radiologists have
traditionally been discouraged from performing preoperative biopsy
on lesions that are surgically excisable, as biopsy rarely affects the
necessity of surgical intervention [10,11]. However, the scenery
seems to change recently, with authors suggesting that biopsy may
have a role for patients with malignant tumours that would benefit
from adjuvant therapy. The suggested route of biopsy seems to
be transperineal or presacral. The argument for this is that, in case
of malignant lesions, the biopsy tract must be respected en bloc
with the specimen to reduce the risk of recurrence in that tract
[39,40]. Therefore, transanal, transvaginal, transperitoneal and
transretroperitoneal routes should be excluded [1]. Yang BL et al.
[33], in their exploration of the benefits of preoperative use of MRI,
claim that the risk of a routine biopsy should be avoided and its
indications limited to patients with metastatic disease or lymphoma.
Rising to the defense of this argument, Macafee et al. [31] suggest that
careful clinical evaluation and preoperative planning utilizing MRI
combined with avoidance of routine preoperative biopsy, result in a
good postoperative outcome.
A large retrospective study from Mayo Clinic Rochester, USA,
reviewed all patients who underwent biopsy of PSTs, with the primary
outcomes measured being the complications that were related to the
biopsy, as well as the precision of preoperative imaging and biopsy,
compared to the final pathology. It was concluded that preoperative
biopsy is safe and concurrent with postoperative pathology in
comparison with imaging. Consequently, percutaneous preoperative
biopsy should be performed, so as to guide decisions, in view of the
substantial differences in therapeutic approach for benign versus
malignant solid PSTs and the current limitations of imaging [39].
Preoperative Therapy
Neoadjuvant chemotherapy may offer substantial benefits in
certain PSTs, such as Ewing sarcomas and osteogenic sarcomas.
Furthermore, the tyrosin kinase inhibitor Imatinib has been
demonstrated to increase disease-free survival time, in cases of
advanced chordomas. Similarly, the epidermal growth factor
inhibitors Gefitinib and Cetuximab, have been shown to cause
favorable results in patients with recurrent and metastatic chordomas
[41,42].
Encouraging results have also been reported for chordomas, via
transcatheter arterial embolization. This technique may be used in the
immediate preoperative time, leading to decreased blood loss during
the operation and facilitating total resection of the tumour [43,44].,br/>
Radiotherapy has an unclear role in the management of PSTs. Generally, chordomas and other PSTs are considered to be
radioresistant, although research on preoperative radiation on PSTs
has been actively continued. Local control of these tumours may be
achieved through recent modalities, including fractionated irradiation
with charged particle carbon ion radiotherapy. Radiotherapy aims to
aid surgical resection decreasing tumour size, treat tumour recurrence
and clean infiltrated surgical margins [6,45].
Surgical Approach
Undoubtedly, surgical resection is the treatment of choice for
all PSTs, even if they are asymptomatic. The reasons for that may be
summarized as follows [1,4]:
1. Spontaneous infection is possible for cystic lesions, a
condition that complicates resection. The possibility of recurrence is
also increased and a second operation might be deemed necessary.
2. PSTs may hinder natural vaginal delivery in women of
reproductive age.
3. Malignancy may lurk even in cystic lesions that appear
benign.
4. There is a chance of malignant degeneration for teratomas.
5. PSTs may cause discomfort of varying degree and their
excision substantially contributes to the improvement of the patient’s
quality of life.
The surgical excision may be accomplice through three approaches:
anterior, posterior and combined abdominosacral. The selection of
a certain approach depends on tumour characteristics (nature, size
and location). The potential infiltration of the sacrum, pelvic sidewall
and adjacent structures should also be taken into consideration.
Lesions that extend above S4 level are usually respected through the
anterior approach, while lower lesions through the posterior one. In
case the upper extent of the lesion is palpable on rectal examination,
it is possible for the lesion to be resected transsacrally [46,47]. The
combined approach is reserved for lesions that are larger or located
in an intermediate position [48].
In any case, a multidisciplinary team including colorectal
surgeons, neurosurgeons, orthopaedic and plastic surgeons should
be activated, assess the various operative approaches and make the
optimal decision [9]. Multidisciplinary collaboration raises the
resection rate of the tumours, preventing unnecessary injuries and
improves the prognosis and patient’s quality of life [48].
Anterior approach (Transabdominal)
This is selected when the tumour is large, located above the level
of S3 or S4 and no nerves are involved (Figure 2). The patient is placed
in lithotomy position. A median abdominal incision is performed,
granting the surgeon a good view of the pelvic structures, iliac vessels
and ureters. The retrorectal space is then dissected, the sigmoid colon
is mobilized, the rectum is pulled to the front and excision of the
tumour follows. It is highly important to protect and preserve the
mesorectum as well as the rectal vessels. Since the middle sacral blood
vessels and the presacral venous plexus are located in this region,
they should be treated with care to avoid presacral hemorrhaege. The
ureter and main nervous branches in the retrorectal region should be
kept safe from injury [9,47,48]. Usually, laparotomy is performed, but
several recent papers have proposed laparoscopic surgery, which may
yield equally satisfactory results, minimizing tissue trauma, especially
when malignancy has been ruled out. Moreover, it is associated with
low rates of postoperative complications, short hospital stay, no
neurological dysfunction and satisfactory long-term results [49,50].
Generally, the anterior approach offers good visualization of
the tumour’s location and extent, as well as its relationships with
the surrounding structures, which is essential when performing
maneuvers of total excision [51].
Posterior approach
This includes the transacral, transsacrococcygeal, transsphincteric,
transrectal and transanorectal techniques. Each of them has its own
advantages and disadvantages and their use depends on the nature
and peculiarities of the tumour, as well as the surgeon’s experience.
The posterior approach is preferred for benign tumors that do not
exceed 8 cm in diameter or for perineal fistulae located below the
level of S4. Notably, it offers good access of the caudal section of the
tumour [47]. The patient takes the Kraske position. An “S”-shaped
or longitudinal incision is made at the level of the S3 vertebra, with
precaution to avoid damaging the anal sphincter complex. Resection
of the coccyx and distal sacrectomy may be performed, so as to
attain better exposure of the presacral area. If sacrectomy is deemed
necessary, at least one side of S2 must be maintained in order to
prevent bowel and urinary disturbance [9,52].
When complete exposition of the tumor is achieved, digital rectal
examination should be performed to attest the extension of the tumour
[48]. The lesion is dissected from adjacent structures, including the
rectal wall, which in most of the cases is not involved. When dealing
with benign lesions, a fat plane is usually encountered between the
lesion and the mesorectum, which makes the dissection easier [1].
In the case of very small, cystic lesions, the following maneuver may
be helpful: the surgeon double-gloving their non dominant hand,
inserts the index finger in the anal canal and the lower one-third of
the rectum, and then applies pressure on the lesion, thus impelling
it out towards the incision. In that way dissection of the lesion off
the rectal wall is achieved and iatrogenic injury to the rectal wall is
prevented. However, if the lesion has been infected, this dissection
may be difficult, especially if the plane between the lesion and rectum
is eradicated. In case secure separation of the lesion cannot be
performed due to its adherence to the rectum, a portion of the rectal
wall should be removed along with the lesion and the defect repaired
in two layers. While routine coccygectomy used to be recommended,
particularly in the case of teratomas, this seems to be no longer the case, unless there is direct invasion of the coccyx by a malignant
lesion or a lesion of uncertain malignant potential [12].
Importantly, the posterior approach is associated with certain
disadvantages, including the deficient control over the pelvic vessels
and the risk of injury to the lateral pelvic nerves. Careful selection of
the cases may minimize these downsides [9].
Combined abdominosacral approach (Abdominoperineal)
A combination of the two aforementioned approaches is
preferred, when the distal margin of the tumour is lower than S3
and the cephalic margin higher [33]. Furthermore, this approach is
applied to cases where the neoplasms invade the rectum or adjacent
structures. It is often the case that the presacral vessels are involved
and should be ligated. It is also the choice in the case of infected
cysts that involve the rectum or the presacral fascia. Then, normal
planes are often unclear and many adhesions to the adjacent tissues
complicate the operation [47].
This approach includes initially a lower midline laparotomy with
the patient in the “sloppy lateral position”, permitting both abdominal
and sacral access. The retrorectal space is investigated following the
mobilization of the sigmoid colon, and the tumour is dissected from
the mesorectum. Similarly, the tumour is separated posteriorly from
the presacral fascia if possible. In case en bloc resection is mandatory,
due to tumour size or extensive infiltration, S3 should be preserved, or
a colostomy should be created for malignant tumours. To minimize
blood loss during the abdominal portion of the operation, careful
ligation of middle and lateral sacral vessels should be performed, as
well as internal vessels. However, the anterior division of the internal
iliac artery must remain intact, to avoid perineal necrosis [4,6].
The next portion is initiated through a sacrococcygeal incision,
with respect to the external sphincter. Rectal resection is completed,
in case this was not performed during the abdominal portion.
Moreover, the anal canal and the anus are removed in case this is
mandatory. Suction drains are placed where appropriate and the
perineal wound is sutured in layers [4].
Distinct benefits linked to the combined approach, may be the
better visualization of structures through the anterior incision, as
well as the improved exposure of the nerve roots via the posterior
approach. For all three approaches intraoperative digital rectal
examination is vital to avoid rectum injury, while dissociating the
tumor. For trans-sacral and combined approaches, at least unilateral
S3 and all of the S1-S2 nerve roots should be reserved and protected
[47].
Discussion
PSTs are rare tumours with variable signs and symptoms. This
fact may lead to delayed diagnosis, inappropriate treatment and
thus bad prognosis. However, modern imaging modalities have
substantially contributed to a prompt and accurate diagnosis, as well
as better preoperative planning via a detailed description of tumour
nature (solid or cystic) and extent. Moreover, preoperative biopsy
may support the appropriate management of heterogeneously cystic
and solid lesions [4].
Adjuvant chemotherapy and tyrosine kinase inhibitors and/
or epidermal growth factor inhibitors may decrease PSTs, such as
sarcomas and chordomas. Conversely, radiotherapy has an unclear
role, due to the radioresistant nature of these tumours. However, high
recurrence rates of malignant PSTs could be treated through modern
radiotherapy, although the available data are still limited. Surgical
management remains the primary and definite therapeutic approach
for PSTs and numerous approaches have been described, aiming to a
safer-more radical tumour resection. New instruments are available
for the surgeons to use nowadays, while laparoscopic techniques are
also performed [1,6].
Surgical resection of benign PSTs may offer 100% overall survival.
Nonetheless, recurrence rates range according to the completeness
of resection. Lev-Chelouche et al. [10] from Israel presented data
on 21 benign PST cases, where no recurrence was discovered in 10
years of postoperative follow-up. Similarly, Glasgow et al. [29] from
USA recorded no recurrence in such patients after a follow-up of 22
months. The same results were reported recently from Maddah et al.
[50] from Iran for 29 benign cases and mean follow-up of 56 months.
Malignant PSTs offer variable rates of survival and recurrence,
according to tumour biology, surgical resection and seeding of
operative field with cancer cells. Moreover, adjuvant chemotherapy
may contribute to better prognosis, while radiotherapy may decrease
recurrence risk. Wang et al. [51] from Taiwan analyzed 22 cases of
malignant PSTs, including chordomas and leiomyosarcomas and
estimated a 5-year survival rate of 41%. Similarly, Lev-Chelouche et al.
[10] investigated 21 cases of such lesions and presented 67% recurrence
and 50% survival rate. Furthermore, Bergh et al. [52] studied 39
patients with chordoma and reported 44% recurrence and 84% 10-
year survival rate. Interestingly, Kaiser et al. [53] concluded that local
recurrence rate may increase from 28% to 64%, when sacrococcygeal
chordomas were violated during the operation. Survival rates were
extensively evaluated by McMaster et al. [54] from USA, through
nine registries from 1973-1995 within the National Cancer Institute’s
Surveillance, Epidemiology and End Result program (NSEER). 400
chordoma cases were reviewed and 5-year and 10-year survival rates
were calculated at 74% and 32% respectively.
Conclusion
Active clinical and experimental research may aid in the development of new neoadjuvant chemotherapeutic agents, as well as new radiation modalities. Carbon ion radiotherapy may constitute a potential advancement in the field of PST therapy, as well as preoperative arterial embolization. Similarly, progress in laparoscopic and robotic surgery may open new horizons in PST surgery. Overall, presenting symptoms in PSTs are nonspecific, while the literature reveals a wide range of variability in physical findings. It is of great importance that clinicians maintain high clinical acumen and remain cognoscente of the range of symptoms linked to PSTs, in order to avoid delayed or false diagnosis. It is noteworthy that accurateprompt diagnosis and successful treatment of PSTs demand active involvement and cooperation of multiple medical specialties, with the surgeons in the leading role.
References
- Hassan I, Wietfeldt ED. Presacral tumors: diagnosis and management. Clin Colon Rectal Surg. 2009;22(2):84-93.
- Uhlig BE, Johnson RL. Presacral tumors and cysts in adults. Dis Colon Rectum. 1975;18(7):581-9.
- Jao SW, Beart RW Jr, Spencer RJ, Reiman HM, Ilstrup DM. Retrorectal tumors. Mayo Clinic experience, 1960-1979. Dis Colon Rectum. 1985;28(9):644-52.
- Toh JW, Morgan M. Management approach and surgical strategies for retrorectal tumours: a systematic review. Colorectal Dis. 2016;18(4):337-50.
- Güvençer M, Dalbayrak S, Tayefi H, Tetik S, Yilmaz M, Erginoğlu U, et al. Surgical anatomy of the presacral area. Surg Radiol Anat. 2009;31(4):251-7.
- Neale JA. Retrorectal tumors. Clin Colon Rectal Surg. 2011;24(3):149-60.
- Huber SA, Northington GM, Karp DR. Bowel and bladder dysfunction following surgery within the presacral space: an overview of neuroanatomy, function, and dysfunction. Int Urogynecol J. 2015;26(7):941-6.
- Hebert-Blouin MN, Sullivan PS, Merchea A, Leonard D, Spinner RJ, Dozois EJ. Neurological outcome following resection of benign presacral neurogenic tumors using a nerve-sparing technique. Dis Colon Rectum. 2013;56(10):1185-93.
- Aranda-Narvaez JM, Gonzalez-Sanchez AJ, Montiel-Casado C, Sanchez-Perez B, Jimenez-Mazure C, Valle-Carbajo M, et al. Posterior approach (Kraske procedure) for surgical treatment of presacral tumors. World J Gastrointest Surg. 2012;4:126-30.
- Lev-Chelouche D, Gutman M, Goldman G, Even-Sapir E, Meller I, Issakov J, et al. Presacral tumors: a practical classification and treatment of a unique and heterogeneous group of diseases. Surgery. 2003;133(5):473-8.
- Glasgow SC, Dietz DW. Retrorectal tumors. Clin Colon Rectal Surg. 2006;19(2):61-8.
- Hobson KG, Ghaemmaghami V, Roe JP, Goodnight JE, Khatri VP. Tumors of the retrorectal space. Dis Colon Rectum. 2005;48(10):1964-74.
- Dahan H, Arrive L, Wendum D, Docou le Pointe H, Djouhri H, Tubiana JM. Retrorectal developmental cysts in adults: clinical and radiologic-histopathologic review, differential diagnosis, and treatment. Radiographics. 2001;21(3):575-84.
- Bathla L, Singh L, Agarwal PN. Retrorectal cystic hamartoma (tailgut cyst): report of a case and review of literature. Indian J Surg. 2013;75(1):204-7.
- Williams L, Rojiani AM, Quisling RG, Mickle JP. Retrorectal cyst-hamartomas and sacral dysplasia: MR appearance. AJNR Am J Neuroradiol. 1998;19(6):1043-5.
- Graadt van Roggen JF, Welvaart K, de Roos A, Offerhaus GJ, Hogendoorn PC. Adenocarcinoma arising within a tailgut cyst: clinicopathological description and follow up of an unusual case. J Clin Pathol. 1999;52(4):310-2.
- Polat AV, Belet U, Aydin R, Katranci S. Anterior sacral meningocele mimicking ovarian cyst: a case report. Medical ultrasonography. 2013;15(1):67-70.
- Haga Y, Cho H, Shinoda S, Masuzawa T. Recurrent meningitis associated with complete Currarino triad in an adult--case report. Neurol Med Chir (Tokyo). 2003;43(10):505-8.
- Oren M, Lorber B, Lee SH, Truex RC Jr, Gennaro AR. Anterior sacral meningocele: report of five cases and review of the literature. Dis Colon Rectum. 1977;20(6):492-505.
- Isik N, Balak N, Kircelli A, Goynumer G, Elmaci I. The shrinking of an anterior sacral meningocele in time following transdural ligation of its neck in a case of the Currarino triad. Turk Neurosurg. 2008;18(3):254-8.
- Kole MJ, Fridley JS, Jea A, Bollo RJ. Currarino syndrome and spinal dysraphism. J Neurosurg Pediatr. 2014;13(6):685-9.
- Al-Essa AA, Malik TA, Baghdadi MK, El Tayeb AA. Adult sacrococcygeal teratomas. Saudi Med J. 2004;25:367-9.
- Simpson PJ, Wise KB, Merchea A, Cheville JC, Moir C, Larson DW, et al. Surgical outcomes in adults with benign and malignant sacrococcygeal teratoma: a single-institution experience of 26 cases. Dis Colon Rectum. 2014;57(7):851-7.
- Sabuncuoglu H, Ozdogan S, Dogan H, Ataoglu O, Timurkaynak E. Total resection of inferiorly located sacral chordoma with posterior only approach: case report and review of the literature. Turk Neurosurg. 2010;20(4):527-32.
- Gottlieb K, Lin PH, Liu DM, Anders K. Transrectal EUS-guided FNA biopsy of a presacral chordoma--report of a case and review of the literature. World J gastroenterol. 2008;14(16):2586-9.
- Sun W, Ma XJ, Zhang F, Miao WL, Wang CR, Cai ZD. Surgical Treatment of Sacral Neurogenic Tumor: A 10-year Experience with 64 Cases. Orthop Surg. 2016;8(2):162-70.
- Boujoual M, Hachi H, Merrouni MA, El Khannoussi B, Bougtab A. [Presacral giant solitary neurofibroma: a rare cause of pelvic mass in women]. Pan Afr Med J. 2014;17:288.