Research Article
Iatrogenic Accessory Nerve Palsy after Laterocervical Lymph Node Biopsy–Clinico-Surgical Considerations on 42 Cases and Review of the Literature
Stefano Ferraresi*, Elisabetta Basso, Maria Angela Pino and Piero Dipasquale
Department of Neurosurgery, Ospedale di Perugia - Santa Maria della Misericordia, Rovigo, Italy
*Corresponding author: Stefano Ferraresi, Department of Neurosurgery, Ospedale di Perugia - Santa Maria della Misericordia, Rovigo, Italy
Published: 06 May, 20177
Cite this article as: Ferraresi S, Basso E, Pino MA,
Dipasquale P. Iatrogenic Accessory
Nerve Palsy after Laterocervical
Lymph Node Biopsy–Clinico-Surgical
Considerations on 42 Cases and
Review of the Literature. Clin Surg.
2017; 2: 1466.
Abstract
Aim of the Authors is to cast a light on a very severe iatrogenic injury, with long-lasting and
potentially devastating consequences for the victims and a very low level of awareness among
the general surgeons, who are usually the actors of the damage. The main features of the typically
involved patients, their clinical presentation and theresults obtainable after surgeryand rehabilitation
are investigated at light ofthe timing of injury and the type of repair.
Keywords: Iatrogenic injury; Posterior cervical triangle; Spinal accessory nerve
Introduction
Thespinal accessory nerve (SAN) is involved in the thoracoscapular physiology and essential for
the complex function of the shoulder [1,2]. It innervates a powerful and widely implanted muscle,
the trapezius, the integrity of which is essential to have a stable scapula, inasmuch it ensures the
scapulohumeral rhythm in synergic function with the serratus anterior muscle. The stabilization
of the scapula is roughly divided into a lower stabilizer, the serratus anterior muscle and an upper
stabilizer, the trapezius. Its proximal insertions make possible the full action of the supraspinatus
muscle which results in complete abduction. This last movement is lost after a complete accessory
nerve injury [3-8].
Iatrogenic causes play a consistent role in SAN injuries, and are mainly due to a radical neck
dissection when complete tumour clearance is desired.
However, this paper focuses on a very particular subset of these injuries, namely thoseoccurring
in course of lymph node biopsy in the posterior triangleof the neck [1-9]. This damage is peculiar
because unintentionally overlooked [11] and easily avoidable with an accurate surgical preparation
and selection [10]. The literature is full of reports focusing on the severity [12-15] of this unfortunate
event and unexpectedly large series witness the relative ease with which this kind of injury happens
[16,17].
Materials and Methods
Between 1996 and 2014, forty-two patients came to our attention for iatrogenic accessory nerve
palsy due to a surgical biopsy in the posterior triangle of the neck, after the emission of the branch
for the sternocleidomastoid muscle.
The age ranged from 18 to 62 years and the male to female ratio was 1: 1.6.
The biopsy, for the majority of them (39/42), was done in local anesthesia.
In 4 cases a diagnosis of lymphoma ensued, in two cases atubercolous chronic infection was
present, but in 36 cases the biopsy was either not conclusive or related to a minor a specific viral
infection, so being such an injury not warranted at the end (Table 1).
Ten patients, at time of the first observation, had an incomplete lesion and were simply
encouraged to wait for a full recovery. Among these, neuropathic pain was moderate if ever present.
In the remaining 32 complete palsies, 8 patients were referred to us very lately, more than 18
months from the time of injury (Table 2).
Thirty patients only had nerve surgery at the end while two of the very late presentations (more than two years afterinjury-Table 2) had instead a pure secondary surgery according to Numanoglu and Rode [18].
Out of them, four cases (4/30) required a simple neurolysis: in
two casesa silk ligature was found but after its removal the accessory
nerve was found in continuity. The other two had scarred nerves
probably related to thermal injury (Table 3).
The surgical approach consisted in a two-flap extension of
the original incision, which is usually quite small. The exposure is
designed to identify the proximal stump of the SAN, where it turns
behind the sternocleidomastoid muscle, and encompassesthe middle
thirdof the trapezius muscle in its anterior border,half-way from
the tip of the mastoid to its anterior insertion on the clavicle. The
anterior border of the trapezius may be heavily scarred and the soft
tissue/musculofascial interface may be not so readily recognizable.
The use of magnifying loupes is strongly recommended because the
dissection may take considerable time as the surgeon distinguishes
the cervical plexus and other sensory branches to the skin of the neck
and the periaural regions. The deeper dissection may bepainstaking
and eventual intramuscular motor branches are very short and
easily missed with the bare eye. Thedifficult task, depending on how
extensive and awkward was the dissection during the former surgery,
is the identification ofthe distal motor branches in the trapezius [19].
This is especially important where a consistent length of the SAN
has been respected at the time of lymph node removal. If, at the
beginning of surgery, one can be confident regarding the possibility
to find the proximal stump, which runs between the sternomastoid
and cleidomastoid bellies of the SCM, not the same can be expected
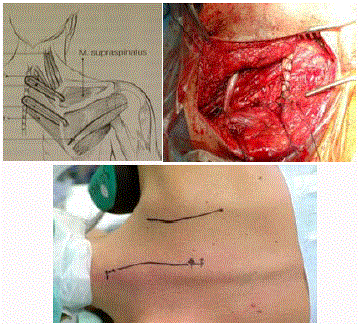
for the distal stump, because at times this results into an impossible
task (Figure 1A-1C). In two of our patients it was not possible, and
sohappened in other cases described elsewhere [20]. Only after
identification and coaptation of the proximal and distal stumps, the
microscope isbrought into the field and the suture is done with one or
two 9-0 stitches and fibrin glue.
After mobilization, if the proximal and distal stumps can be
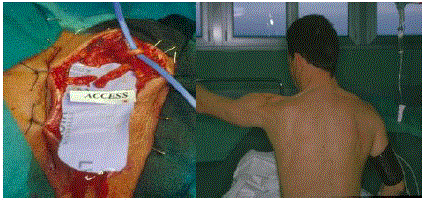
brought together without significant tension (Figure 2A), then a direct
anastomosis is the choice and thishappened in 8/30 cases (25%).
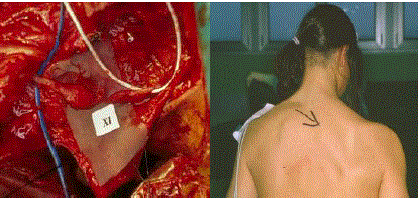
In the other cases a graft procedure was required (Figure 3A). The
great part was carried out with local nerves (greater auricular), while
less often a sural graft was harvested (Table 4).
When the final gap was short (about 1 cm) a segment of vein
stuffed with muscle (one case), or a neural tube (Neuragen – two
cases) could be used.
Selection of grafts
Finding a local graft, namely a branch of the greater auricular
nerve, is a clear advantage over the sural graft. This because we would
be dealing with another incision and, moreover, because the harvest
of a small sural nerve graft exposes the patient to a high incidence of
painful neuroma. This because the proximal stump, after a short graft,
would lie immediately under the skin, over the fascial compartment.
When more graft material is harvested, on the contrary, the
proximal stump lies deep in the calf, normally buried within the
muscle. In this way the neuroma formation is very unlikely to be a
source of pain.
Forthe sake of precision, we haveto remark how, from time to
time, also the local nerves are source of complaints. In fact, occasional
patients refer of a painful and annoying sensation in the infra and retroauricolar area, although this probably is more related to the
original damagethan as a result of their use as a graft.
Figure 1(a-c)
Figure 2
Figure 2
A) A direct suture is feasible. B) Same patient. Note the deformity
of the shoulder muscles and the levator scapulae in relief like a tense strand.
Figure 3
Figure 3
A) Nerve interruption with neuroma. After resection a graft will be
required. B) Same patient: note the striking atrophy of the trapezius muscle.
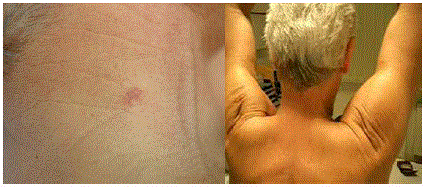
Figure 4
Figure 4
A) Note the exceedingly small scar of the first surgery. B) Same
patient after reconstructive surgery: excellent result.
Table 1
Table 2
Table 3
Table 4
Results
As expected, none of the patients where the intraoperative
impressionhad been unfavorable (4 cases), showed a useful result.
This, coupled with the two patients in whom the distal stumps were
not found, enhances the cohort of patients with a complete failure.
Excluding the obvious good results obtained by neurolysis (4 cases,
Table 3), the final results of nerve repair are burdened by a 9/26 (35%)
of M0 score. The patients with neural tubes and the vein graftscored
M0.
This last result could have been originally biased by thesenior
surgeon (SF). In fact, in three very late cases, a “second choice
material” such as the neural tubes and the vein graft were intentionally
chosen to avoid a sural nerve graft in patients deemed to have a very
low potential of regeneration, because of the exceedingly long time
elapsed.
Thevery high percentage of bad results (35%), which differs
from what reported in other papers [21-23], may so find a reliable
explanation.
Nevertheless, as already stated, a constant difficulty comes from
finding useful distal motor branches at the anterior border of the
trapezius, and this especially when the first surgery has entailed a
heavy scarring of the area.
Of the other cases, 13/17 had a very good result (trapezius scoring
M3 or more) so configuring an overall l50% of good results after this
kind of surgery.
One clear advantage, in the patients receiving a nerve repair, is
the disappearance of neuropathic pain, so suggesting a role of the
proximal stump in the pain symptoms.
At muscular effort and endurance, however, only those who
had an excellent to good BMC score (M3 and M4) experienced
a significant improvement in shoulder pain and function. A full
restoration to normal was described mainly among those having
eithera spontaneous recovery orafter surgery with neurolysis.
Discussion
Some important considerations emerge from the analysis of our
cases and namely:
a. Local anesthesia, and the consequent obvious lack of
curarization, does not protect from iatrogenic injury. The only
difference is that usually the patient experiences a sharp pain during
the removal of the lymph node. As a matter of fact, in the iatrogenic
cases of our series, the nerve stimulator was never used.
b. Those patients operated in general anesthesia feel much
more postoperative pain than expected, and keeping the arm at rest,
as it is normally advised, they delay the possibility of an immediate
diagnosis and repair, which would be the ideal situation.
c. The vast majority of patients present severe pain and
devastating muscle atrophy which, in turn, prompts a very particular
postural attitude, with hyperactivity of the levator scapulae and
striking atrophy of the shoulder (Figure 2B and 3B).
d. The patients showing a deficit of the trapezius muscle are
almost always addressed to rehabilitation therapy which, however,
very rarely has been of help in making the correct diagnosis. On the
opposite, asking for severe endeavour of the periscapular muscles, it
significantly contributes to increase pain and disability.
e. For the overmentioned reasons, the vast majority of our
patients attain a very late diagnosis, the majority of them between 5
to 8 monthsfrom the offending event.
f. The early recovery of some activity in the trapezius muscle
is a good prognosticsign. We have a consistent number of patients
who recovered very wellwithout surgery.
g. Generally speaking, one should not start surgery planning
a neurolysis, since those cases having a continuity of the nerve are
generally deemed to have a spontaneous recovery. However, on some
occasions, the pain is overwhelming and in these casesaneurolysis
is warranted. A strong ligature around the nerve or other type of
persistent damage, in fact, can well sustain the neuropathic pain
(Figure 4A and 4B).
h. If the patient shows no recovery at EMG at 3-6 months or if
he/she has excruciating pain, is addressed to surgery: usually a nerve
section is found although from time to time (severe scarring, silk ligatures around the nerve) a simple neurolysis is all what is needed
and the immediate postoperative effect is striking.
i. The surgical exploration generally is followed either by
a direct repair or by a 3 cm graft: this depends on the pathologic
anatomy and not from the time interval between injury and repair.
j. After surgical repair all the patients have experienced a clear
improvement in the pain around the shoulder, with the only exception
of those in whom the distal stump is not found, the proximal stump
developing a neuroma. A possible role of the proximal stump of the
accessory nerve seems so to be involved in the genesis of neuropathic
pain.
k. A nerve repair is certainly worth in the majority of cases.
However only slightly more than 50% of the whole group of patients
experiences a long term recovery ofshoulder function and less muscle
atrophy.
l. Afull restoration to normal has been principally observed
in the group who had neurolysis.
m. We have not clearly defined a time limit to perform or not
the repair, but the results of a nerve repair done later than 18 months
after iatrogenic injury stronglyfavour a secondary surgery such the
Eden-Lange [24] modified by Bigliani [6] or the Henry/Szubinskj as
first-time procedures [25-30].
n. People undergoing successful secondary procedures,
however, are less free of pain than those having successful nerve
repair, this further confirming an intrinsic cause of neuropathic pain
in the posterior cervical area.
Conclusion
This nerve ishighly vulnerable along its superficial course.
This study confirms how very commonly the diagnosis of this
iatropathic injury goes unnoticed for a long time before diagnosis
andmanagement are instituted.
Nerve exploration is certainly worth but for several reasons the
recovery after nerve repair is not complete in all the patients, reaching
roughly only the half of them.
Recognition and referral are seldom, if ever, made by the surgeon
responsible for the injury, leading to a marked delay in treatment.
From examination of our records, 42 patients with lesions of the
spinal accessory nerve due to lymph node biopsy have on average an
8 months delay between iatrogenic damage and referral.
Most diagnoses were due to unexplained pain and loss of
shoulder function while only secondarily because of the deformity in
the shoulder attitude.
The clinical picture is obvious. There is a characteristic downward
and lateral displacement of the scapula, with narrowing of the
inferior scapulohumeral angle and loss of function, with severe pain
commonly present.
The course of the nerve in relation to the sternocleidomastoid
muscle is constant and damage to the spinal accessory nerve is
usuallydistal to the branch for this muscle in the so-called posterior
triangle of the neck (PCT).
Despite the delay, the results of repair are surprising
especiallyconcerningearly relief of pain, implying a neuropathic
source which even precedes the recovery of muscle function.
References
- Birch R, Bonney G, Dowell J, Hollingdale J. Iatrogenic injuries of peripheral nerves. J Bone Joint Surg Br. 1991;73(2):280-2.
- Bradley PJ, Ferlito A, Silver CE, Takes RP, Woolgar JA, Strojan P, et al. Neck treatment and shoulder morbidity: still a challenge. Head Neck. 2011;33(7):1060-7.
- Camp SJ, Birch R. Injuries to the spinal accessory nerve: a lesson to surgeons. J Bone Joint Surg Br. 2011;93(1):62-7.
- Cesmebasi A, Spinner RJ. An anatomic-based approach to the iatrogenic spinal accessory nerve injury in the posterior cervical triangle: How to avoid and treat it. Clin Anat. 2015;28(6):761-6.
- Galano GJ, Bigliani LU, Ahmad CS, Levine WN. Surgical treatment of winged scapula. ClinOrthopRelat Res. 2008;466(3):652-60.
- Goeransson H, Leppaenen OV, Vastamaeki M.Patient outcome after surgical management of the spinal accessory nerve injury: A long-term follow-up study. SAGE Open Med. 2016;4.
- Grossman JA, Ruchelsman DE, Schwarzkopf R. Iatrogenic spinal accessory nerve injury in children. J Pediatr Surg. 2008;43(9):1732-5.
- Kelley MJ, Kane TE, Leggin BG. Spinal accessory nerve palsy: associated signs and symptoms. J Orthop Sports PhysTher. 2008;38(2):78-86.
- Kim DH, Cho YJ, Tiel RL, Kline DG. Surgical outcomes of 111 spinal accessory nerve injuries. Neurosurgery. 2003;53(5):1106-12.
- King RJ, Motta G. Iatrogenic spinal accessory nerve palsy. Ann R CollSurg Engl. 1983;65(1):35-7.
- Kuntz AL, Weymuller EA Jr. Impact of neck dissection on quality of life.Laryngoscope. 1999;109(8):1334-8.
- London J, London NJ, Kay SP. Iatrogenic accessory nerve injury. Ann R CollSurg Engl. 1996;78(2):146-50.
- Midwinter K, Willatt D. Accessory nerve monitoring and stimulation during neck surgery. J Laryngol Otol. 2002;116(4):272-4.
- Morris LG, Ziff DJ, DeLacure MD. Malpractice litigation after surgical injury of the spinal accessory nerve: an evidence-based analysis. Arch Otolaryngol Head Neck Surg. 2008;134(1):102-7.
- Nakamichi K, Tachibana S. Iatrogenic injury of the spinal accessory nerve. Results of repair. J Bone Joint Surg Am. 1998;80(11):1616-21.
- Nason RW, Abdulrauf BM, Stranc MF. The anatomy of the accessory nerve and cervical lymph node biopsy. Am J Surg. 2000;180(3):241-3.
- Novak CB, Mackinnon SE. Patient outcome after surgical management of an accessory nerve injury. Otolaryngol Head Neck Surg. 2002;127(3):221-4.
- Numanoglu A, Rode H. Cervical lymph node biopsy -- watch the nerves! S Afr Med J. 2006;96(1):51-2.
- Okajima S, Tamai K, Fujiwara H, Kobashi H, Hirata M, Kubo T. Surgical treatment for spinal accessory nerve injury. Microsurgery. 2006;26(4):273-7.
- Olarte M, Adams D. Accessory nerve palsy. J NeurolNeurosurg Psychiatry. 1977;40(11):1113-6.
- Park SH, Esquenazi Y, Kline DG, Kim DH. Surgical outcomes of 156 spinal accessory nerve injuries caused by lymph node biopsy procedures. J Neurosurg Spine. 2015;23(4):518-25.
- Romero J, Gerber C. Levator scapulae and rhomboid transfer for paralysis of trapezius. The Eden-Lange procedure. J Bone Joint Surg Br. 2003;85(8):1141-5.
- Lopatecki M, Loritz T, Rudigier J. Measuring intracranial pressure after trepanation in traumatized patients. AktuelleTraumatol. 1993;23(5):217-22.
- Terrell JE, Welsh DE, Bradford CR, Chepeha DB, Esclamado RM, Hogikyan ND, et al. Pain, quality of life, and spinal accessory nerve status after neck dissection. Laryngoscope. 2000;110(4):620-6.
- Vastamäki M, Solonen KA. Accessory nerve injury. ActaOrthop Scand. 1984;55(3):296-9.
- Wiater JM, Bigliani LU. Spinal accessory nerve injury. ClinOrthopRelat Res. 1999;(368):5-16.
- Williams WW, Twyman RS, Donell ST, Birch R. The posterior triangle and the painful shoulder: spinal accessory nerve injury. Ann R CollSurg Engl. 1996;78(6):521-5.
- Woodhall B, Odom GL, Bloor BM, Golden J. Studies on cerebral intravascular pressure; further data concerning residual pressure in components of the circle of Willis and in blind arterial segments. J Neurosurg. 1953;10(1):28-34.
- Wright TA. Accessory spinal nerve injury. ClinOrthopRelat Res. 1975;(108):15-8.
- Yavuzer G, Tuncer S. Accessory nerve injury as a complication of cervical lymph node biopsy. Am J Phys Med Rehabil. 2001;80(8):622-3.