Research Article
Reduced Port Laparoscopic Retroperitoneal Liposarcoma Resection
Olivier Nicod1, Michel Degueldre2 and Giovanni Dapri1,3*
1Department of Gastrointestinal Surgery, European School of Laparoscopic Surgery, Saint-Pierre
UniversityHospital, UniversitéLibre de Bruxelles, Brussels, Belgium
2Department of Gynecology and Obstectrics, Saint-Pierre University Hospital, Brussels, Belgium
3Laboratory of Anatomy, Faculty of Medicine and Pharmacy, University of Mons, Mons, Belgium
*Corresponding author: Giovanni Dapri, Department of Gastrointestinal Surgery, European School of Laparoscopic Surgery, Saint- Pierre University Hospital 322, Rue Haute1000 Brussels, Belgium
Published: 03 May 2017
Cite this article as: Nicod O, Degueldre M, Dapri
G. Reduced Port Laparoscopic
Retroperitoneal Liposarcoma
Resection. Clin Surg. 2017; 2: 1459.
Abstract
Liposarcomas originate from the primitive mesenchymal cells of the soft tissue of the body’s
extremities and retroperitoneum. Retroperitoneal liposarcomas are suspected once they become
clinically symptomatic due to their size and repercussion to the adjacent organs. They are commonly
diagnosed at 40-60 years of age with a slight male predominance. Contrast enhanced computed
tomography scan and magnetic resonance imaging allow establishing the staging. Image-guided
core needle biopsy can help in the diagnosis, but the specimen’s examination remains the gold
standard.
The authors report a 47-year-old female presenting the suspicion of a middle-size liposarcoma in the
left paracolic gutter, safely removed by 3 trocars laparoscopy and confirmed at the histopathological
examination.
Keywords:Retroperitoneal liposarcoma; Laparoscopy; Reduced port
Introduction
Liposarcomas account for 10-12% of all soft tissue sarcomas [1-3]. They seem to originate from
the primitive mesenchymal cells. These cells come from the soft tissue of the body’s extremities or
the retroperitoneum. Retroperitoneal Liposarcomas(RPLS) represent the most frequent histologic
type affecting the adults with 41% of these tumors [4]. Usually they are being suspected once they
become clinically symptomatic due to their size and repercussion to the adjacent organs. They are
commonly diagnosed at 40-60 years of age with a slight male predominance. Contrast Enhanced
Computed Tomography scan (CECT-scan) and magnetic resonance imaging (MRI) allow to
establish the staging and to evaluate the possibility of resection [5-6]. Image-guided core needle
biopsy can help in the diagnosis [7], but the specimen’s examination remains the gold standard.
Due to the low response to chemotherapy, optimal treatment remains the surgical resection with
safe margins [5]. Therefore large laparotomies are usually performed due to the large size of these
tumors once they are being discovered.
The authors report a case of a woman presenting the suspicion of a middle-size liposarcoma in the
left paracolic gutter, safely removed by 3 trocars laparoscopy and confirmed at the histopathological
examination.
Case Presentation
A 47-year-old female with a 20,42 kg/m2 body mass index, consulted her family doctor for a
persistent cough and fever since a week. She was known with chronic constipation. She denied any
other gastro-intestinal symptoms, and did not present any urologic, gynecological nor neurological
complaints. General physical examination was unremarkable. Blood test showed an inflammatory
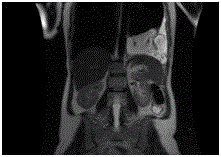
syndrome and an increased CA-125 tumor marker. An abdominal MRI showed a heterogeneous
retroperitoneal mass of 88 mm per 65 mm. This mass was located between the left Gerota’s fascia
and the descending colon containing fat tissues and calcifications (Figure 1). The abdominal CECTscan
confirmed this hypothesis. A reduced port diagnostic laparoscopy including tumor removal
was proposed to the patient and has been accepted.
Surgical techniquem
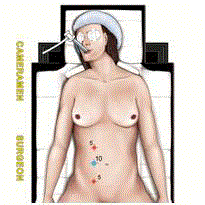
The patient was placed in supine position. Peritoneal cavity was entered at the right flank by the open laparoscopy technique. After establishment of the
pneumoperitoneum by carbon dioxide insufflations through a 10mm
trocar, two other 5 mm trocars were positioned, one in the right
hypochondrium and the other one in the right iliac fossa respectively
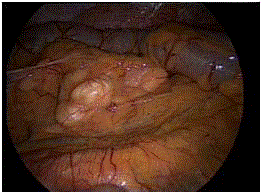
(Figure 2). At laparoscopic exploration, the mass appeared located
in the left mesocolon under the left colon and above the left kidney
(Figure 3). The “touching sensation” of the mass by atraumatic
grasping forceps revealed partially soft and calcified tissue. The
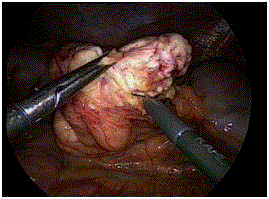
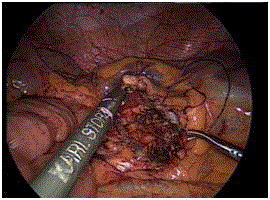
left mesocolon was opened by a coagulating hook respecting the
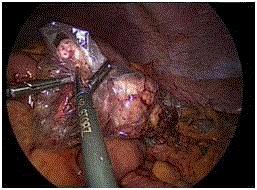
proper colic vascularization(Figure 4). The tumor was respected
together with a part of the inferior renal fatty tissue and part of
the retroperitoneal fatty tissue, ensuring an adequate safety of the
margins. After complete removal, the left mesocolon appeared to be
well vascularized, as well as the descending colon. The left mesocolon
was closed by an absorbable running suture (Figure 5). The respected
specimen was removed in a plastic bag (Figure 6) through the
enlargement of the 10 mm trocar. No drain was left in the abdomen.
After withdrawal of the three trocars, the 10 mm trocar was closed in
plans by absorbable sutures.
Figure 1
Figure 2
Figure 3
Figure 4
Figure 5
Figure 6
Results
Operative time was 105 minutes. Estimated blood loss was 20
cc. Postoperative course was uneventful. Flatus and intestinal transit
were restored at day one postoperative. Liquid nutrition was started
and the patient was discharged at day two.
Anatomopathology revealed a well-differentiated liposarcoma
weighting 170 grams with a major proportion of fatty tissue
proliferation, with calcifications and atypical cells expressing the
MDM2 receptor. This is corresponding with the grade 1 of the
French FNCLCC (Fédération National des Centres de LutteContre le Cancer) histological evaluation system. The patient was followed up
regularly at office visits and at 6 months she was doing well.
Discussion
Minimally invasive surgery (MIS) has to be considered as a
useful diagnostic method in front of a patient with an uncertain
diagnosis. It allows the surgeon to explore the entire abdominal
cavity and to evaluate the location and extension of the tumor.
Moreover by collecting liquid samples and frozen biopsies, it permits
to detect the presence of a peritoneal carcinomatosis, which remains
a contraindication to standard resection. This latter aspect is a
substantial advantage of diagnostic laparoscopy because it avoids a
useless laparotomy.
The optical system enables to localize precisely the mass, and
thanks to the latest evolution of 3D cameras [8], the contiguity of
the tumor with the surrounding organs can be assessed. Obviously,
after having completed the diagnostic laparoscopy, the decision
to continue to operate by laparoscopy or to perform a standard
laparotomy can be undertaken. If laparoscopy is chosen, various
advantages can be added to the patient like minimal postoperative
pain, faster intestinal peristalsis restoration, reduced hospital stay,
low surgical sites infection rate, low incisional hernia rate and finally
enhanced cosmetic outcomes.
On the other hand, laparotomy may appear superior to
laparoscopy concerning the “touching sensation” of the tumor,
but nowadays laparoscopic atraumatic grasping forceps enable the
surgeon to get a similar sensation like the one obtained with the
proper hands. Furthermore, the surgeon’s experience in laparoscopic
oncologic surgery can help in this matter. Surely, the laparoscopic
resection must always be performed respecting the rules of oncologic
surgery, like free margins, «R0» removal, no tumor’s diffusion, and
tumor’s supplying vessels first.
Laparoscopic surgery is usually performed placing 5 to 7 trocars
into the abdomen [9]. The current trend is to reduce the number and
the size of the trocars, heading towards reduced port laparoscopic
surgery [10]. We adopted this latter technique considering the
patient’s characteristics, the size and the location of the tumor. The
laparoscopic resection is feasible and can be proposed for small and
middle size tumors, whereas laparotomy is certainly adopted in front
of giant tumors. After laparoscopic resection, the retrieval of the mass
can be done through a trocar enlargement, like in our patient, or
through an additional suprapubic access.
Our operative time was similar to other laparoscopic approaches
reported in literature [9] and it was shorter than the average time
for open surgery regardless of the size of the masses[11]. The patient
was discharged at day 2, which is another advantage of laparoscopy
considering the average of 11 days reported in literature [11].
RPLS can be classified into four histologic types (welldifferentiated,
myxoid/round cell, pleomorphic, and dedifferentiated)
and three grades (grade I, II and III) according to the WHO
classification [12].As Dalal “et al.”[13], among the four subtypes,
well-differentiated (46%) are the most common forms, followed by
myxoid/round cell (28%), dedifferentiated (18%) and pleomorphic
(8%). Recently, Gronchi“et al.”[14]reported an overall 5-year survival
for well-differentiated subtypes of 90%, comparing to 30-50% for
the pleomorphic subtypes. The rates of myxoid/round cell and
de-differentiated subtypes are 60-90% and 75%, respectively. In a
large cohort of 72 patients with primary RPLS, Neuhaus“et al.”[15]
found that R0 excision and histologic grade I am the only variables
significantly associated with a decreased rate of local recurrence.
The 5-year rate of metastasis is approximately 3%, when the size
of the tumor is less than 2.5 cm. This rate climbs to 55-60% if the
tumor mesures more than 20 cm [16].
Even though there is no specific scientific data, the consensus
agrees that a close clinical follow-up is mandatory. It may include
an abdominal MRI and CECT-scan every 3 to 6 months in the early
years after surgery and afterwards, yearly controls [17]. Follow-up is
advised for at least 10 years, allowing controlling local recurrences
that sometimes predate symptoms by years [17]. These recurrences
are the cause of 75% the mortality of well-differentiated liposarcomas
[17]. Our patient was consulted at 1, 3 and 6 months postoperatively
and, so far, she did not present any clinical or radiological tumor
recurrence.
Conclusion
Small, well-differentiated and isolated retroperitoneal liposarcomas can be safely removed by reduced port laparoscopy, which is a step forward to enhance all the advantages of MIS.
References
- Dong M, Bi J, Liu X, Wang B, Wang J. Significant partial response of metastatic intra-abdominal and pelvic round cell liposarcoma to a small-molecule VEGFR-2 Tyrosine kinase inhibitor apatinib: a case report. Medicine (Baltimore). 2016;95(31):e4368.
- Taguchi S, Kume H, Fukuhara H, Morikawa T, Kakutani S, Takeshima Y, et al. Symptoms at diagnosis as independent prognostic factors in retroperitoneal liposarcoma. Mol Clin Oncol. 2016;4(2):255-260.
- Molina G, Hull MA, Chen YL, DeLaney TF, De Amorim Bernstein K, Choy E, et al. Preoperative radiation therapy combined with radical surgical resection is associated with a lower rate of local recurrence when treating unifocal, primary retroperitoneal liposarcoma. J Surg Oncol. 2016;114(7):814-20.
- Leao P, Vilaça S, Oliveira M, Falcao J. Giant recurrent retroperitoneal liposarcoma initially presenting as inguinal hernia: review of literature. Int J Surg Case Rep. 2012;3(3):103-6.
- Trans-Atlantic RPS Working Group. Management of primary retroperitoneal sarcoma (RPS) in the adult: a consensus approach from the Trans-Atlantic RPS Working Group. Ann Surg Oncol. 2015;22:256-63.
- Thomas JM. Retroperitoneal sarcoma. Br J Surg. 2007;94(9):1057-8.
- Wilkinson MJ, Martin JL, Khan AA, Hayes AJ, Thomas JM, Strauss DC. Percutaneous core needle biopsy in retroperitoneal sarcomas does not influence local recurrence or overall survival. Ann Surg Oncol. 2015;22(3):853-8.
- Buchs NC, Morel P. Three-dimensional laparoscopy: a new tool in the surgeon's armamentarium. Surg Technol Int. 2013;23:19-22.
- Nomura R, Tokumura H, Matsumura N. Laparoscopic resection of a retroperitoneal liposarcoma: a case report and review of the literature. Int Surg. 2013;98(3):219-22.
- In: Dapri G, Mori T. Reduced Port Laparoscopic Surgery. Springer. 2014.
- Gemici K, Buldu Ä°, Acar T, Alptekin H, Kaynar M, Tekinarslan E, et al. Management of patients with retroperitoneal tumors and a review of the literature. World J Surg Oncol. 2015;13:143.
- Fan Z, Tian XF, Tang SX, Zhang YY, Pan JY, Wang S. Myxoid liposarcoma in the abdominal wall: a case report. Medicine (Baltimore). 2014;93(28):e239.
- Dalal KM, Kattan MW, Antonescu CR, Brennan MF, Singer S. Subtype specific prognostic nomogram for patients with primary liposarcoma of the retroperitoneum, extremity or trunk. Ann Surg. 2006;244(3):381-91.
- Gronchi A, Collini P, Miceli R, Valeri B, Renne SL, Dagrada G, et al. Myogenic differentiation and histologica grading are major prognostic determinants in retroperitoneal liposarcoma. AM J Surg Pathol. 2015;39(3):383-93.
- Neuhaus SJ, Barry P, Clark MA, Hayes AJ, Fisher C, Thomas JM. Surgical management of primary and recurrent retroperitoneal liposarcoma. Br J Surg. 2005;92(2):246-52.
- McCallum OJ, Burke JJ, Childs AJ, Ferro A, Gallup DG. Retroperitoneal liposarcoma weighing over one hundred pounds with review of the literature. Gynecol Oncol. 2006;103(3):1152-54.
- Bonvalot S, Raut CP, Pollock RE, Rutkowski P, Strauss DC, Hayes AJ, et al. Technical considerations in surgery for retroperitoneal sarcomas: position paper from E-Surge, a master class in sarcoma surgery, and EORTC-STBSG. Ann Surg Oncol. 2012;19(9):2981-91.