Research Article
Autologous Breast Reconstruction Implant Augmentation (A.B.R.I.A.): Indications and Outcomes
Ryan E. Rebowe*, J. Clayton Crantford, Austin Bond and Ivo A. Pestana
Department of Plastic and Reconstructive Surgery, Wake Forest University Baptist Medical Center, USA
*Corresponding author: Ryan E. Rebowe, Department of Plastic and Reconstructive Surgery, Wake Forest University Baptist Medical Center, Winston Salem, North Carolina 27103, USA
Published: 17 Apr, 2017
Cite this article as: Rebowe RE, Crantford JC, Bond
A, Pestana IA. Autologous Breast
Reconstruction Implant Augmentation
(A.B.R.I.A.): Indications and Outcomes.
Clin Surg. 2017; 2: 1406.
Abstract
Background: Increasing numbers of young women with normal body mass index (BMI) are receiving
autologous tissue breast reconstruction, often requiring a prosthetic implant to achieve their desired
volume and symmetry. Our aim is to describe the technique of autologous breast reconstruction
implant augmentation (A.B.R.I.A.) and its indications and outcomes to satisfy soft tissue implant
coverage, volume, and contour problems in this breast reconstruction patient population.
Methods: A single surgeon’s experience with consecutive patients undergoing A.B.R.I.A. over a
four year period was conducted. Outcomes related to patient demographics, indications for surgery,
operative procedure, implant type, size, location and timing of placement, and complications were
analyzed. In addition, a PubMed MeSH database search for related topics to identify technique
variables that may influence outcome of the procedure was completed.
Results: Six patients who underwent eight breast reconstructions were included in the study. There
were no implant or flap complications. The literature review identified seven reports reviewing 2341
breast reconstructions, 226 of which utilized A.B.R.I.A. including our series; these flaps had a flap
loss and implant failure rate of 0.4% and 3.1% respectively. The rate of infection, seroma, partial flap
loss, and hematoma are 3.5%, 1.8%, 6.1%, and 3.1%.
Conclusion: A.B.R.I.A combines the benefits of both prosthetic and autologous reconstruction
including a straight forward secondary procedure, excellent implant soft tissue coverage, hidden
donor site scarring, improved abdominal contour, and minimal to no donor site functional loss. This
study demonstrates that autologous breast reconstruction implant augmentation has an acceptably
low complication rate and can be a safe and effective option for optimizing breast reconstruction
with autologous tissue.
Introduction
Operations utilizing autologous tissue from the abdominal donor site have been an integral
part of breast reconstruction since their description in the late 1970’s. There are many reasons for
the popularity of these procedures. The abdominal soft tissues are the ideal replacement for the
breast due to its reliable blood supply, ease in molding, and natural ptosis. Moreover, advancement
of microsurgical techniques to allow harvest of perforator flaps has improved these operations
significantly resulting in decreased rates of abdominal donor site morbidity [1-3]. Despite
this, many women are not considered candidates for abdominal based flaps due to a paucity of
abdominal tissue. If these patients desire the natural ptosis and longevity of an autologous flap,
they have traditionally been offered a latissimus dorsimyocutaneous (LDMC) flap, with or without
an implant, or alternative microvascular free tissue transfer sites such as the thigh or the gluteal
areas. These reconstructive options come with the donor site morbidity that abdominally based flaps
seek to avoid such as a potentially visible posterior thorax scar and functional deficits in the upper
extremity. Furthermore, the tissue they provide is not as ideal in terms of natural look and ease of
molding [4,5].
At our institution, women who desire the natural ptosis and permanence of an autologous
reconstruction but have a relative lack of donor site soft tissues are candidates for autologous breast
reconstruction implant augmentation (A.B.R.I.A.). This technique employs standard techniques for
autologous breast reconstruction followed by implant augmentation similar to that employed for
cosmetic breast augmentation. Our aim is to describe the technique of A.B.R.I.A. and its indications
and outcomes to satisfy soft tissue implant coverage, volume and contour problems in this breast
reconstruction patient population. In conjunction, a literature review was completed to identify technique variables that may influence outcomes of this procedure.
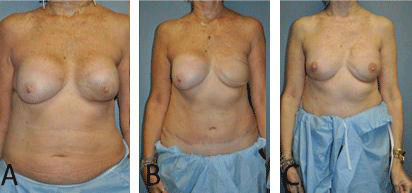
Figure 1
Figure 1
57 year old woman with left-sided ductal carcinoma in situ prior
to undergoing free tissue transfer. Note site of previous lumpectomy on left
breast. B) Same patient status post muscle sparing free TRAM to the left
breast. C) Same patient after placement of bilateral subpectoral Allergan
Style 10 implants. For a complete pictorial of this patient’s reconstructive
process, please see Supplementary Material 1.
Materials and Methods
Patients
All cases performed by the senior author (IAP) from December
2011 to January 2014 were reviewed. These cases were reviewed for
variations in technique such as type and volume of implant placed,
plane of implant placement, timing of implant placement, type of
microsurgical anastomosis if present, and length of follow up. All
complications including hematoma, seroma, infection, implant
extrusion, and flap failure were analyzed. This study was approved by
the Institutional Review Board at our institution.
Literature review
Surgical technique: Women who are appropriate candidates for
free tissue transfer are identified in clinic and a frank preoperative
discussion is held regarding risks, benefits, and alternatives to the
procedures. We first perform an abdominally based microvascular
transfer based according to the standard previously described
technique [6,7]. The choice of muscle sparing free transverse rectus
abdominus myocutaneous flap (msfTRAM) vs. deep inferior
epigastric perforator (DIEP) flap is made at the time of surgery based on the individual perforator anatomy of each patient. The patient is
allowed to completely recover and heal following their microvascular
free tissue transfer (Figure 1A/B).
Once the patient is totally healed, operative planning for implant
placement is begun. This time allows for the abdominal flap to take the
natural shape of a breast, allowing us to utilize standard tissue based
planning techniques when selecting an implant. In doing so, we take
into account breast width, breast height, and intermammary distance.
Nipple to inframammary fold (IMF) distance and sternal notch to
nipple distance are estimated based on the purported placement of
the nipple reconstruction. A style and approximate size of implant
is selected in clinic with the patient to ensure she is comfortable with
the selection [8-10].
The patient is then taken back to the operating room for
placement of the implant (Figure 1C). We utilize previous incisions
for implant placement, taking care to note the position of the
previous microvascular pedicle. At the time of surgery, the quality
and amount of flap tissue is assessed. If the flap tissue is insufficient to
provide adequate coverage for the implant, the prosthesis is inserted
in the sub-muscular plane. Otherwise we are comfortable placing the
implant underneath the flap itself. No a cellular dermal matrix (ADM)
is utilized in this secondary procedure, nor do we leave drains.
Following placement of the implant, further reconstructive
goals are discussed with each patient. We offer each patient with
contour irregularities further revision procedures. We discuss all
options with each patient including fat grafting, liposuction, scar
revision, or local flap reconstruction. Nipple tattooing and surgical
nipple reconstruction are offered to all women undergoing breast
reconstruction. As with any breast reconstruction method, the
number of total surgeries will depend on each individual patient.
Statistical analysis
All comparisons of complication rates were analyzed for
statistical significance using chi-squared tests. P values less 0.05 were
considered statistically significant.
Figure 2
Figure 2
Abdominal fascial defects and reconstruction.
(A) Fascial defect closed primarily at the superior aspect with ADM inlay to bridge the remaining defect.
(B) A 600 cm2 ADM underlay and vacuum assisted closure dressing were placed during the laparotomy 10 days prior to definitive abdominal wall reconstruction.
Table 1
Results
Our results
Six patients with a mean age of 49.5 years and a mean BMI of
22.1 underwent breast reconstruction with 8 autologous flaps (Table
1). Muscle sparing free TRAM flaps were used in 63% (5/8) of cases
followed in frequency by DIEP flaps (2/8) and a single pedicled
TRAM. Indications for the use of autologous tissue include previous
radiation (4/6), failed implant reconstruction, and level of activity.
All patients underwent delayed augmentation with a mean implant
volume of 190 mL. Placement of implants was distributed equally
between sub-flap (4/8) and sub-pectoral (4/8) pockets. Median
time from breast reconstruction to implant augmentation was 5.5
months. There were no implant/flap complications. Following
completion of the reconstruction with the implant, further revision
of the reconstructed breast or contralateral breast may be indicated.
Two patients, comprising three reconstructions, did undergo further
fat grafting for contour irregularities. We did regularly perform
symmetry procedures on the contralateral breast for symmetry.
One patient each received contralateral mastopexy, augmentation,
mastopexy and augmentation, contralateral expander/implant
reconstruction, and bilateral axillary liposuction. Four patients went
on to have nipple and areola reconstruction or tattoo.
Average follow up was 8.5 months. An example patient is shown
in Figure 2.
Discussion
Since their introduction in the 1970’s, abdominally based
flaps have become the preferred method for autologous tissue
reconstruction of the breast. While surgical advancements have
provided various methods of tissue harvest, all abdominal flaps
share certain characteristics that make this tissue the ideal breast
replacement. These factors include a reliable axial blood supply,
ease of molding, and the aesthetically pleasing body contouring
provided by the donor site. Utilizing all of these advantages allows
the reconstructive surgeon to provide the patient with a permanent
breast mound replacement with natural feel and ptosis [1,2].
However, not all women have sufficient abdominal tissue to
perform a total autologous reconstruction. Though the desires, age,
and co-morbidity profile of a particular woman may allow her the
benefits of an abdominally based reconstruction, her relative paucity
of abdominal fat and overlying skin would limit the ability of the
reconstructive surgeon to provide a satisfactory outcome utilizing
only this tissue. While alternative flaps such as the superior gluteal
artery perforator (SGAP) flap, inferior gluteal artery perforator
(IGAP) flap, transverse upper gracilis (TUG) flap, or profunda
artery perforator (PAP) flap are autologous tissue options, women
without sufficient tissue for abdominal free tissue transfer rarely have
sufficient tissue in these sites [11-13]. Stacked flaps are available to
these women and have a generally good success, but they do require a
longer operating time and increased number of vascular anastomoses
which can increase the risk associated with the procedure including
medical complications and flap loss [14,15]. While the LDMC flap
can be utilized for autologous reconstruction, the transverse scarring
across the superior thorax and functional deficit following muscle
harvest make this flap a less ideal candidate in younger women and
can be associated with lower satisfaction scores [5,16]. Therefore,
it may be beneficial to perform the autologous reconstruction and
subsequently, or simultaneously, augment the planned flap with a
prosthetic implant. At our institution, we have termed this process
autologous breast reconstruction implant augmentation (A.B.R.I.A.).
We have found that A.B.R.I.A. can combine the benefits of
both autologous and implant based reconstruction. The autologous
reconstruction will provide the natural appearance and feel of a
breast mound while also providing ample soft tissue for coverage
of the implant. The implant will allow for increased projection, improve contour abnormalities, and further shape the autologous
reconstruction. This process has proved a reliable way to perform
reconstructions that are safe, stable, and aesthetically pleasing to both
patient and surgeon.
To further discern the indications and risks of A.B.R.I.A., a
literature review was performed to discern the current trends as well
as the specific risks of this technique. A Pubmed Medical Subject
Headings (MeSH) search was performed on available Pubmed and
Medline literature. Inclusion criteria included articles in English
language journals pertaining to human subjects. Only articles with
more than one subject and follow up greater than 6 months were
included. Furthermore, only articles that listed complications as they
specifically pertain to the technique variables described above were
included. Exclusion criteria included articles in non-English language
journals, case studies, articles that described only technique without
patient examples, or articles that did not include sufficient follow up
or complication information for their provided cases. In order for a
complication to be included in the analysis, it must be reported in the
majority of the papers reviewed.
A Pubmed MeSH search for “Mammaplasty,” which includes
all articles related to augmentation and reconstruction, returned
9,112 results. Separate Pubmed searches that included the MeSH
term “Mammaplasty” as well as “Autologous” or “Implant” returned
984 results and 1,867 articles respectively. A pubmed search for
all headings together produced 268 results. Articles that met our
inclusion criteria were reviewed as were the references of the included
articles to ensure all pertinent information was included. Overall, we
found 12 articles that pertained to A.B.R.I.A. Seven of these articles
met our previously described criteria for inclusion.
These seven articles included description of 226 breast
reconstructions (Table 2). In 149 (72%) of these breast reconstructions,
the implant was placed in the subpectoral plane while in the remaining
58 (28%) cases the implant was placed underneath the autologous
reconstruction (sub-flap). Twenty-one (9.2%) of the reconstructions
were performed utilizing a pedicled TRAM flap. 205 (90.8%) of the
reconstructions utilized either the muscle sparing free transverse
rectus abdominus myocutaneous flap (msTRAM) or the deep inferior
epigastric perforator flap (DIEP). One hundred (48.8%) of these free
tissue transfers utilized the internal mammary artery (IMA) pedicle,
and 105 (51.2%) utilized the thoracodorsal (TD) pedicle. Ninetynine
(43.9%) of these reconstructions were performed in a delayed
fashion, with implant or expander placement at a later time following
autologous reconstruction, while 127 (56.1%) reconstructions were
performed with simultaneous flap and implant placement.
All included reconstructions recorded information regarding
complication rates. The rate of flap loss and implant removal
(describing reconstructive failure in both cases) was 0.4% and 3.1%,
respectively. The rate of seroma and hematoma were equally low at
1.8% and 3.1% each, while the rate of partial flap loss was slightly
higher at 6.1%. Documented infections can range between an acute
infection of the implant itself (necessitating removal) to erythema
of the flap or surrounding skin that was responsive to antibiotic
therapy. As most papers reviewed failed to give a firm definition of
observed infection, we report here the total rate of infection listed in
the literature as 3.5%.
To determine which differences in technique, if any, make a
difference in outcome, we compared the incidence of complications
in each reported subgroup of procedure variations. The only
difference in technique that is consistently reported to affect the rate
of complication is the timing of placement of the implant. Those that
performed the implant placement in a delayed fashion compared to
flap coverage reported different outcomes than those authors that
reported performing implant placement and flap inset at the same
time. The results of this comparison are shown in Table 3 [17-23]. As
shown, the overall rate of infection is the only complication that is
affected by any statistically significant amount when these two groups
are compared.
Unfortunately, combining two procedures can also expose
patients to the combined risks of each separate surgery. The
autologous reconstruction may expose her to vascular compromise
of the flap and its sequelae. Similarly, utilizing a breast prosthesis
can lead to an increased risk of hematoma, seroma or infection.
Fortunately our report indicates that ABRIA is safe overall, with very
low risk of flap loss or implant removal (0.4% and 3.1% relatively).
Likewise, the incidence of hematoma, seroma, and partial flap loss is
also extremely low (3.1%, 1.8%, and 6.1% respectively). While the rate
of reported infection was extremely low (3.5%), all of these infections
occurred when the flap and implant were placed simultaneously.
This information would corroborate the conclusions of Roehl et
al., [22] who found an increased risk of long term complications,
including a significant number with capsular contracture, in those
patients in whom implants and autologous tissue reconstructions
were performed simultaneously. The rate of infection reported in
the literature is comparable to the latissimus dorsi flap and implant
reconstruction, with reported infection rates of 1.2-5.4%. [24-26] At
this time, we would not hesitate to perform A.B.R.I.A. on appropriate
patients, but we will warn any patients whom are contemplating a
combined procedure that this may expose them to an increased risk
of infection of the implant or the surrounding soft tissue envelope.
As with any surgical procedure, patient selection is critical.
Previous authors have outlined the ideal candidates for combining
prosthetic implant and abdominal autologous tissue reconstruction.
Kronowitz et al. described this procedure as most useful in young
women with medium to large size breasts [20]. Serletti and Moran
note than women who benefit most from the combined procedure
have minimal abdominal donor tissue and large, ptotic breasts [18].
Likewise, multiple authors note that the procedure is particularly
helpful in matching symmetry to a previously reconstructed or
natural contralateral breast [18,21]. There is also significant evidence
that combining implants and abdominal autologous tissue can
provide a stable long term reconstruction for women with previously
irradiated breasts [27]. Miller et al. has previously provided the most
specific guidelines on patient selection. They enumerated five patient
preferences that were key to their decision to pursue A.B.R.I.A.
These included insufficient abdominal tissue for total autologous
reconstruction, desire for immediate breast reconstruction, and
patient comfort with prosthetic implants, wish to avoid tissue
expansion, and preference for abdominal donor site [17].
In our practice, we have found that utilizing A.B.R.I.A. in
young patients with low BMI provides the best aesthetic outcome
and risk profile for combining these procedures. The guidelines for
patient selection set forth by Miller et al. have certainly proven true
in our practice environment. In particular, avoiding the scar and
functional deficit of the latissimus dorsi flap is a critical component
of the decision for both the surgeon and the patients. Furthermore,
appropriate patient education about the risks involved with utilizing
prosthetic implants in the setting of reconstruction has played a
critical role in our patient’s overall comfort with the procedure.
The variations in surgical techniques have not proven to play a
significant role in patient outcomes, though the lack of reporting of
these variables in relation to surgical outcomes and complications
is a limitation in our report. There does not seem to be a particular
affinity for placing the implant in the sub-muscular or sub-flap
pocket, nor does this seem to influence the final outcomes. Similarly,
there does not seem to be a preference for which recipient vessels
are utilized for the microanastomoses. While avoiding pressure on
the microvascular pedicle has traditionally been a concern in any
autologous reconstruction, it does not appear that placement of the
implant has any impact on these vessels, regardless of which recipient
vessels are chosen. The demonstrated rate of total flap loss and partial
flap necrosis was 0.4% and 6.1%, both of which are very well within
previously reported acceptable limits.
As with any study, our research does have its own set of
limitations. The data from our institution is a small data set without
sufficient power for drawing wide conclusions. When combined with
our literature review, we believe that we have provided a set of data
with sufficient power to guide surgical decisions at our institution
and for other surgical teams. This review is limited, however, to the
amount and type of previously reported data. The lack of standard
definition of complications, in particular infection and partial flap
loss or fat necrosis, is a limitation of literature reviews in general and
must be recognized when counseling patients utilizing information
garnered from this report.
Table 2
Table 3
Conclusion
We have found that combining lower abdominal autologous tissue reconstruction with implant based reconstruction can provide stable long term reconstructions to a certain subset of individuals. Typically these are young patients with low BMI and limited abdominal donor tissue that wish to avoid the donor site morbidity of the latissimus dorsi flap. Complications rates combining abdominal autologous and prosthetic implant reconstruction are acceptably low and in general do not seem to be influenced by technique variables. When utilizing these techniques during the same procedure, there is an increased risk of infection that the patient must be aware of for true informed consent. Overall, autologous breast reconstruction implant augmentation is a safe and reliable way to provide an aesthetically pleasing breast reconstruction.
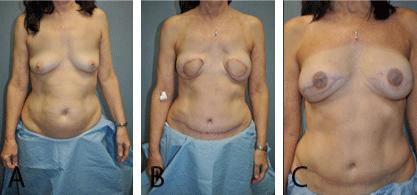
Figure 2
Figure 2
A) 54 year old woman with left breast invasive ductal carcinoma.
Patient was an A cup prior to surgery and wished to remain her size or
increase in size following surgery. B) Same patient following bilateral muscle
sparing free TRAM reconstruction C) Patient after placement of bilateral
subpectoral 150cc Allergan Style 10 Implants. For a complete pictorial of
this patient’s reconstructive process, please see Supplementary Material 2.
References
- Koshima I, Soeda S. Inferior epigastric artery skin flaps without rectus abdominis muscle. Br J Plast Surg. 1989;42(6):645-8.
- Allen RJ, Treece P. Deep inferior epigastric perforator flap for breast reconstruction. Ann Plast Surg. 1994;32(1):32-8.
- Blondeel PN. One hundred free DIEP flap breast reconstructions: a personal experience. Br J Plast Surg. 1999;52(2):104-11.
- Caterson SA, Carty MJ, Helliwel LA, Hergrueter CA, Pribaz JJ, Sinha I. Evolving options for breast reconstruction. Curr Probl Surg. 2015;52(5):192-224.
- Yang JD, Huh JS, Min YS, Kim HJ, Park HY, Jung TD. Physical and Functional Ability Recovery Patterns and Quality of Life after Immediate Autologous Latissimus Dorsi Breast Reconstruction: a 1-year Prospective Observational Study. Plast Reconstr Surg. 2015;136:1146-54.
- Chang DW. Breast Reconstruction with Microvascular MS-TRAM and DIEP Flaps. Arch Plast Surg. 2012;39(1):3-10.
- Granzow JW, Levine JL, Chiu ES, Allen RJ. Breast reconstruction with the deep inferior epigastric perforator flap: history and an update on current technique. J Plast Reconstr Aesthet Surg. 2006;59(6):571-9.
- Tebbetts JB, Adams WP. Five critical decisions in breast augmentation using five measurements in 5 minutes: the high five decision support process. Plast Reconstr Surg. 2005;116:2005-16.
- Tebbetts JB. A system for breast implant selection based on patient tissue characteristics and implant-soft tissue dynamics. Plast Reconstr Surg. 2002;109(4):1396-409.
- Adams WP Jr, Mallucci P. Breast augmentation. Plast Reconstr Surg. 2012;130(4):597e-611e.
- Granzow JW, Levine JL, Chiu ES, Allen RJ. Breast reconstruction with gluteal artery perforator flaps. J Plast Reconstr Aesthet Surg. 2006;59(6):614-21.
- Wechselberger G, Schoeller T. Transverse upper gracilis flap for breast reconstruction. Plast Reconstr Surg. 2012;130(1):182e-183e.
- Allen RJ, Haddock NT, Ahn CY, Sadeghi A. Breast reconstruction with the profunda artery perforator flap. Plast Reconstr Surg. 2012;129(1):16e-23e.
- DellaCroce FJ, Sullivan SK, Trahan C. Stacked deep inferior epigastric perforator flap breast reconstruction: a review of 110 flaps in 55 cases over 3 years. Plast Reconstr Surg. 2011; 127:1093-9.
- Stalder MW, Lam J, Allen RJ, Sadeghi A. Using the Retrograde Internal Mammary System for Stacked Perforator Flap Breast Reconstruction: 71 Breast Reconstructions in 53 Consecutive Patients. Plast Reconstr Surg. 2016;137(2):265e-277e.
- Yueh JH, Slavin SA, Adesiyun T, Nyame TT, Gautam S, Morris DJ, et al. Patient satisfaction in postmastectomy breast reconstruction: a comparative evaluation of DIEP, TRAM, latissimus flap, and implant techniques. Plast Reconstr Surg. 2010;125(6): 1585-95.
- Miller MJ, CS Rock, GL Robb. Aesthetic breast reconstruction using a combination of free transverse rectus abdominis musculocutaneous flaps and breast implants. Ann Plast Surg. 1996;37:258-64.
- Serletti JM, Moran SL. The combined use of the TRAM and expanders/implants in breast reconstruction. Ann Plast Surg. 1998;40(5):510-4.
- Spear SL, Wolfe AJ. The coincidence of TRAM flaps and prostheses in the setting of breast reconstruction. Plast Reconstr Surg. 2002;110(2):478-86.
- Kronowitz SJ, Robb GL, Youssef A, Reece G, Chang SH, Koutz CA, et al. Optimizing autologous breast reconstruction in thin patients. Plast Reconstr Surg. 2003;112(7):1768-78.
- Figus A, Canu V, Iwuagwu FC, Ramakrishnan V. DIEP flap with implant: a further option in optimising breast reconstruction. J Plast Reconstr Aesthet Surg. 2009;62(9):1118-26.
- Roehl KR, Baumann DP, Chevray PM, Chang DW. Evaluation of outcomes in breast reconstructions combining lower abdominal free flaps and permanent implants. Plast Reconstr Surg. 2010;126(2):349-57.
- Pien I, Caccavale S, Cheung MC, Butala P, Hughes DB, Ligh C, et al. Evolving Trends in Autologous Breast Reconstruction: Is the Deep Inferior Epigastric Artery Perforator Flap Taking Over? Ann Plast Surg. 2016;76(5):489-93.
- Levine SM, Patel N, Disa JJ. Outcomes of delayed abdominal-based autologous reconstruction versus latissimus dorsi flap plus implant reconstruction in previously irradiated patients. Ann Plast Surg. 2012;69(4):380-2.
- Chang DW, Barnea Y, Robb GL. Effects of an autologous flap combined with an implant for breast reconstruction: an evaluation of 1000 consecutive reconstructions of previously irradiated breasts. Plast Reconstr Surg. 2008;122(2):356-62.
- Pinsolle V, Grinfeder C, Mathoulin-Pelissier S, Faucher A. Complications analysis of 266 immediate breast reconstructions. J Plast Reconstr Aesthet Surg. 2006;59(10):1017-24.
- Chang DW, Y Barnea, GL Robb. Effects of an autologous flap combined with an implant for breast reconstruction: an evaluation of 1000 consecutive reconstructions of previously irradiated breasts. Plast Reconstr Surg, 2008. 122(2):356-62.