Case Report
Disseminated Thrombi in a Patient with Suspected Patent Foramen Ovale Post Left Lower Lobectomy
Sanjeet Singh Avtaar Singh, Sotiris Papaspyros, Karim Morcos* and Mohammed Asif
Department of Cardiothoracic Surgery, Golden Jubilee National Hospital, UK
*Corresponding author: Karim Morcos, Department of Cardiothoracic Surgery, Golden Jubilee National Hospital, Glasgow, UK
Published: 14 Apr, 2017
Cite this article as: Singh SSA, Papaspyros S, Morcos
K, Asif M. Disseminated Thrombi in a
Patient with Suspected Patent Foramen
Ovale Post Left Lower Lobectomy. Clin
Surg. 2017; 2: 1405.
Abstract
A 73-year-old man presented with a T2a N0 M0 adenocarcinoma of lung confirmed via CT guided
biopsy. He underwent a left lower lobectomy for the 30 mm mass posteriorly within the apical
segment left lower lobe. His operation was uncomplicated however he developed a post operative
pneumonia noted on CXR. He developed a DVT postoperatively which coincided with bilateral
pulmonary emboli and systemic dissemination of thrombi.
Echocardiography raised the suspicion of an undiagnosed Patent Foramen Ovale. After a period
of anticoagulation on advice from Haematology, he subsequently recovered with supportive
management. His thrombophillia screen did not reveal any abnormalities. On follow up, albeit frail,
he recovered well from his ordeals and is actively rehabilitating.
Case Presentation
A 73-year-old man presented with a T2a N0 M0 adenocarcinoma of lung confirmed via CT
guided biopsy. His past medical history included moderate chronic obstructive pulmonary disease
(COPD) (Forced Expiratory Volume at 1 second (FEV1) 1.18 ~ 48% predicted & transfer factor
of 4.3~57% predicted), obesity, peripheral vascular disease, hypertension, atrial fibrillation – on
Warfarin, Penicillin Allergy and previous unstable angina –15 years ago and an Ex-smoker- 90
pack year history. He has a penicillin allergy and is an ex-smoker with a 90-pack-year history. He
underwent a left lower lobectomy for the 30-mm mass posteriorly within the apical segment left
lower lobe. It was noted that he had fragile perivascular tissues intraoperatively but there were
no complications and was extubated in the operating room. His post-operative period had been
complicated by a pneumonia (chest infection with consolidation on chest radiograph (CXR) a week
postoperatively) as he became tachypnoeic with peripheral capillary oxygen saturation (SPO2)
<90%. He was haemodynamically stable but had a low-grade pyrexia (37.8°C). He complained of
tightness in his left calf on postoperative day 12. A lower limb duplex ultrasound confirmed a deep
venous thrombosis within the distal femoral and popliteal vein of his left lower limb despite having
a therapeutic prothrombin ratio (INR) and receiving 5000 units of Dalteparin (prophylactic low
molecular weight heparin) daily since admission alongside anti-thrombotic (TEDTM) stockings.
His oxygenation had not improved on arterial blood gases thus a pulmonary embolus was clinically
suspected as he became tachycardic but his blood pressure remained stable at 128/71 mmHg. A
Computed Tomography Pulmonary Angiogram (CTPA) confirmed bilateral pulmonary embolus
and he was transferred to the intensive care unit for invasive monitoring. A subsequent CTPA
revealed a large mural thrombus in relation to left lower lobar artery stump which was thought to
be related to the lung resection with thrombi within the middle lobar and segmental arteries and
in several of the segmental arteries to the right lower lobe. In addition, it was felt a combination
of heart failure with superimposed infection or acute respiratory distress syndrome (ARDS) was
suggested based on ground glass appearance of CT findings of the left lung. On the delayed scan,
some thrombus was seen in a dilated right atrium which adhered to the atrial septum.
He underwent a further CTPA on POD 41 which revealed no change in the thrombus within
the post-resection left lower lobar artery, and both atria but revealed a large mobile serpentinious
thrombus within the right atrium which traversed the tricuspid valve, potentially arising from the
foramen ovale although no flow was demonstrable on Doppler. There was however progressive
bilateral small pleural effusion with progression in ground glass consolidation bilaterally within
the lungs suggesting ARDS, potentially due to micro-emboli. He was reintubated as his oxygen
requirements increased and he had difficulty tolerating continuous positive airway pressure
(CPAP). He had a caval filter inserted on POD 43 by the interventional radiologists. Unfortunately, he developed an asystolic arrest on POD 50 and was successfully
resuscitated. A Tracheostomy was fashioned on the 51st POD. He
underwent pulmonary rehabilitation with input from dietitians and
physiotherapists alike. Day 74 – Right apical pneumothorax noted
10-15% of lung field Day 76 – Right apical pneumothorax resolving.
It was felt that he would benefit from being closer to home thus he
was transferred to a district general hospital for on the 81st post op
day. He discharged from the DGH after 14 days. He was seen in clinic
4 months postoperatively looking remarkably well, mobilising with
the aid of two sticks.
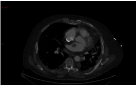
Figure 1
Figure 1
There was also thrombi seen within the left atrium raising the
possibility of a patent foramen ovale which was suggested as a possibility
on echocardiography.
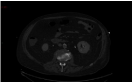
Figure 2
Figure 2
Further thrombo-embolic phenomena were seen in his kidneys
with little change to other organs.
Discussion
Lung resection carries a risk of pulmonary thromboembolism with an incidence of about 1% [1]. This well-recognised complication may indeed be fatal for the patient. Many prevention strategies have been employed including the routine use of Prophylactic Low Molecular Weight Heparin, TEDTM stockings and early mobilisation as per recommendation of the American College of Chest Physicians [2] and the European Society of Medical Oncology [3]. The patient unfortunately developed a DVT and intracardiac emboli despite the above mentioned prophylactic measures. The management of PE in the context of intracardiac thrombus [4] is controversial. Experts have not reached a consensus approach and current literature reflects this. This is probably due to the rare nature of this disease as there are no distinguishing clinical features from pulmonary embolus. Our patient was also screened for possible causes of thrombophillia but these were negative. A case series of 3 patients reported similar findings with bilateral DVTs, pulmonary embolus and intracardiac emboliiv [4]. They postulated that the thrombi likely originated from the DVT and got dislodged in the right atrium. In theory, it is also possible that thrombus in the right heart chambers may be responsible for PE if we consider that the heart is a symmetrical structure [5]. Our patient also had a suspicion of a patent foramen ovale which was noted on ECHO which increases the risk of shunting albeit he had no neurological features to suggest an intracerebral infarct. A group of investigators analysed autopsy results from in-hospital deaths over a period of thirteen years. They found that intracardiac thrombi were present in 7% of all cases. Right-sided thrombosis had similar incidence to leftsided thrombosis. Up to half of these patients also had a concomitant pulmonary embolus [5]. LA thrombosis is also associated with atrial fibrillation [6]. Inferior vena cava (IVC) filters may be of benefit in cases of large PE to prevent further embolism. This will provide the necessary time for resolution of the PE with anticoagulation of the patient. This strategy may prevent propagation of emboli in patients where thrombolysis and pulmonary embolectomy carry prohibitive risks or are not available. The International Cooperative Pulmonary Embolism Registry (ICOPER) assessed 11 patients with massive PE (defined by systolic arterial pressure <90 mm Hg) who IVC filters inserted. 10 of them survived at least 90 days with no recurrent PE. They concluded that the use of IVC filter in patients with unfavourable cardiopulmonary profile may be a viable option [7]. IVC filter placement, however, carries significant risks. Mortality has been reported at 0.1%. Other complications include device malposition (1.3%), pneumothorax (0.02%), hematoma (0.6%), air embolism (0.2%), inadvertent carotid artery puncture (0.04%), and arteriovenous fistula (0.02%). Thrombosis of the common femoral vein has a higher incidence (8.5%) and it is related to removal of the insertion sheath. Recurrent DVT (21%), IVC thrombosis (2% to 10%), IVC penetration (0.3%) and filter migration (0.3%) [8,9] have also been reported as late complications. As witnessed in the subsequent CTPA, our patient had no further PEs and did not have any clinical signs of a subsequent DVT. Silent infarcts occur in patients with Patent Foramen Ovale (PFO) despite infrequent neurological complications. A study used diffusion-weighted MRI to detect the incidence of cerebral emboli in patients who had been hospitalized after suffering a PE. Patients with a PFO were more likely to have subclinical brain infarcts than those without a PFO (5 (33.3%) of 15 versus one (2.2%) of 45 patients, P=0.003) although only one patient with a PFO had neurological symptoms. Furthermore, logistic regression analysis flagged the existence of a PFO as an independent predictor of subclinical cerebral infarcts (OR, 34.9 (3.1 to 394.3); P=0.004) [10]. Despite undergoing an eventful postoperative period, our patient survived neurologically intact and can mobilise independently. This case is unique as it employed several strategies to deal with the disseminated thrombi with a positive outcome.
References
- Kalweit G, Huwer H, Volkmer I, Petzold T, Gams E. Pulmonary embolism: a frequent cause of acute fatality after lung resection. Eur J Cardiothorac Surg. 1996;10(4):242-6.
- Gould MK, Garcia DA, Wren SM, Karanicolas PJ, Arcelus JI, Heit JA, et al. Prevention of VTE in non orthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2):e227S–e277S.
- Mandalà M, Falanga A, Roila. ESMO Guidelines Working Group. Management of venous thromboembolism (VTE) in cancer patients: ESMO Clinical Practice Guidelines. Ann Oncol. 2011;22(6):vi85–vi92.
- Brodmann M, Stark G, Pabst E, Lueger A, Pilger E. Pulmonary embolism and intracardiac thrombi--individual therapeutic procedures. Vasc Med. 2000;5(1):27-31.
- Ogren M, Bergqvist D, Eriksson H, Lindblad B, Sternby NH. Prevalence and risk of pulmonary embolism in patients with intracardiac thrombosis: a population-based study of 23 796 consecutive autopsies. Eur Heart J. 2005;26(11):1108-14.
- Singer DE, Albers GW, Dalen JE, Go AS, Halperin JL, Manning WJ. Antithrombotic therapy in atrial fibrillation: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 Suppl):429S-456S.
- Deshpande KS, Hatem C, Karwa M, Ulrich H, Aldricht TK, Kvetan V. The use of inferior vena cava filter as a treatment modality for massive pulmonary embolism. A case series and review of pathophysiology. Respir Med. 2002;96(12):984-9.
- DuraiRaj R, Fogarty S. A penetrating inferior vena caval filter... Eur J Vasc Endovasc Surg. 2006;32(6):737-9.
- Mohan CR, Hoballah JJ, Sharp WJ, Kresowik TF, Lu CT, Corson JD. Comparative efficacy and complications of vena caval filters. J Vasc Surg. 1995;21(2):235-45.
- Clergeau MR, Hamon M, Morello R, Saloux E, Viader, Hamon M. Silent cerebral infarcts in patients with pulmonary embolism and a patent foramen ovale: a prospective diffusion-weighted MRI study. Stroke. 2009; 40: 3758–62.