Research Article
Hemodynamic Effects of Commercial Flights. Potential Implication on Patients
Hernan Cohen Arazi*, Silvina Waldman, Norberto Casso and Mauricio Abello
Department of Cardiology, CEMIC Saavedra, Argentina
*Corresponding author: Hernán Cohen Arazi, Department of Cardiology, CEMIC Saavedra, Peron 2351. Derqui, Buenos Aires Argentina, Barrio La Delfina, Argentina
Published: 31 Mar, 2017
Cite this article as: Arazi HC, Waldman S, Casso N,
Abello M. Hemodynamic Effects
of Commercial Flights. Potential
Implication on Patients. Clin Surg.
2017; 2: 1389.
Abstract
Background: The main cause of in-flight mortality is a cardiac event. Most of the available
information arises from small retrospective studies. The aim of the study is to analyze the
cardiovascular response during a commercial flight in healthy volunteers.
Methods: Twelve healthy volunteers were studied. Prior to the flight all subjects were studied with
physical evaluation, electrocardiogram (EKG), capillary saturation monitoring, echocardiogram,
stress test echocardiogram, 24 h EKG Holter, 24 h blood pressure monitoring, and blood tests which
included pro brain natriuretic peptide (pro BNP) and C reactive protein (CRP). During the flight,
all subjects had a 24 h EKG Holter, an echocardiogram was performed in cruise altitude, blood
pressure and capillary saturation was measured every 15 mins. One hour after landing blood pro
BNP and CRP was dosed.
Results: There was a significant difference in the saturation observed between base and at 120
mins (98.4% + 0.5 vs. 93.1% +1.7, p<0.001). Blood pressure decreased from baseline to 120 mins
(119 mmHg + 11.6/ 76.6 + 11 mmHg and 110 mmHg+ 7/ 70.2 mmHg+ 5, respectively, p=0.03).
No differences were observed in the measurements of heart rate, CRP and proBNP. During the
flight, 2 subjects showed transient regional wall-motion abnormalities, one of whom showed apical
ballooning pattern.
Conclusion: We observed important decrease in arterial oxygen saturation during the flight and
the arterial blood pressure was significantly lower compared to baselines measures. Two healthy
volunteers showed cardiovascular dysfunction during the commercial flight. Haemodynamic
changes should be considered in patients with cardiac conditions, after thoracic surgery and
cardiovascular treatment.
Introduction
Every year, nearly 40 million people flight around the world [1]. Moreover, the average passenger
age is rising due to an increased life expectancy in western countries, estimating that by 2030, half
of the passengers in commercial flights will be over 50 years of age [2]. This translates into a higher
number of people with cardiac diseases or with history of cardiac surgery that travel in commercial
flights. Due to this matter, the question if flying is safe or if special care is needed appears to be a
frequent issue, not only in hospitals but also in outpatient settings [3].
Retrospective data shows that the higher percentage of in flight mortality is due to cardiac events
[1]. Reflecting this concern, in the 2001 Federal Aviation Administration stated that any flight that
weighted over 7,500 pounds (3,400 kg) has to provide an external defibrillator in the emergency
plane kit [1,4]. Moreover, since 2004, basic pulmonary cardiac resuscitation training is mandatory
for in-flight assistants [4].
Commercial flights maintain a relative cabin altitude between 5,000 and 8,000 feet during
routine flight. At this altitude, the barometric pressure (BP) decreases from a normal sea level value
of 760 mm Hg to around 560 mm Hg, with lower baseline arterial partial pressure of oxygen (a OP),
higher oxygen consumption and higher sympathetic tone – which predispose to arrhythmia, and
deep vein thrombosis secondary to blood stasis [5-7]. These conditions could be more important
and frequent in people who had a cardiac surgery.
Most information comes from retrospective studies, and there is no study evaluating the effect
of commercial flights in the cardiac physiology. There are no references of these changes after heart
or thoracic surgery. The aim of this study is to evaluate changes in cardiac physiology in a group of healthy volunteers. It could be a basement to analyze the situation when a physician needs to consider the patient risk for air travel.
Methods
This is a prospective, observational study, in which a cohort of
healthy volunteers served as self-control. The objective was to assess
changes in cardiovascular physiology during air travel. Male or
female subjects, between 18 and 40 years were selected if they had
no history of cardiovascular disease, hypertension, diabetes mellitus,
or dyslipidemia. Subjects with history of arrhythmias, acute coronary
syndrome, cardiac surgery, blood pressure> 135/85 mmHg, resting
hypoxemia (SaO2 <97% at rest at sea level, breathing room air) and
history of panic attacks or other anxiety disorders were excluded.
Also subjects with abnormal electrocardiogram (AV block, bundle
branch block, of pre-excitation syndrome, atrial fibrillation, or signs
of atrial or ventricular overload) and poor acoustic window for
echocardiogram were excluded from the study.
All volunteers who met the inclusion criteria and had no exclusion
criteria had a complete medical history, physical examination, 12-
lead electrocardiogram and an exercise echocardiogram. Moreover,
Doppler echocardiogram, 24 h ambulatory blood pressure
monitoring, 24 h holter EKG, pulse oximetry and a blood test
(including C Reactive Protein (CRP) and proBNP) was performed
before the study and in-flight, to each participant.
The study included data derived from three different trips,
including 4 volunteers in each one. One of the subjects was reevaluated
in a second trip.
Blood pressure was measured before boarding the plane (T0)
and at different periods that were correlated with holter monitoring
(it was placed on the ground before boarding): at takeoff (T1), 20
minutes after takeoff (T2), reaching 8,000 meters (T3), at 60 minutes
(T4), at 75 minutes (T5), 105 (T6) and 120 minutes (T7), at the time
that the descent (T8) started and during landing (T9). The destination
was the same for all the flights (Rio Gallegos, Santa Cruz, Argentina),
with the same duration (180 minutes) and the same cruise altitude
8000 meters. The echocardiogram was performed at 60 minutes, with
an altitude of 8,000 meters. Flight anxiety and stress was assessed with
Body Sensation Questionnaire (BSQ; a 17-item questionnaire with
increasing levels of somatic arousal scored 17-85) prior to take off.
The study was conducted according to the Declaration of Helsinki,
Good Clinical Practice Guidelines and under Argentinean data
protection law. Every volunteer signed informed consent.
Statistics
The data was analyzed using STATA statistical program 9.0.
Values are expressed as mean±standard deviation and
interquartile range or average, as appropriate.
A comparative analysis between the baseline results and the
measurements obtained in-flight was made; Student test and chi2
were used to assess statistical significance considering a p-value <0.05
as statistically significant.
Figure 1
Figure 2
Results
Twelve male individuals (35±4 years) with no history of cardiac
disease or coronary risk factor were included.
The cabin pressurization and weather conditions were similar in
the 3 flights.
There were no significant differences in heart rate at different
times of flight (HR baseline = 70.5 (61.5-73) beats/minute, takeoff=72
(69-80) beats/min, p=0.27, 71 (65-75.5 bpm) to 8,000 meters of
altitude. p=0.56 (compared to baseline).
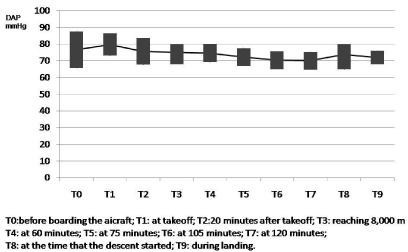
Significant differences were observed in the blood pressure
values; the maximum SBP and DBP were observed at the time of
takeoff (130.6+19 mmHg and 79.8+6.8 mmHg, respectively). The
lower SBP and DBP, were recorded at cruise altitude (at 120 mins:
110+7 mmHg: 70.2+5 mmHg, respectively) compared to baseline
(119+11.6 mmHg, p=0.049 and 76.6+11 mmHg, p=0.03, respectively)
and landing (122.2+14 mmHg, p=0.018 and 72+2.5 mmHg, P=0.043,
respectively) (Figure 1 and 2).
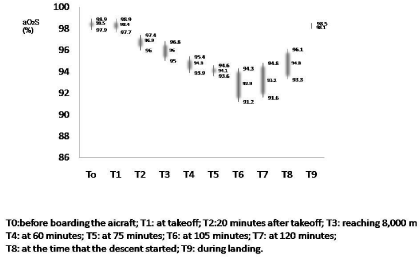
A decrease in arterial oxygen saturation level was observed during
the flight when compared with baseline, with lowest saturation levels
recorded at 120 minutes in cruise altitude (98.4 vs. 93.1+1.7+0.5,
p<0.001).
The echocardiogram performed in flight, showed no significant
differences with the baseline diameters of the left ventricle (LV) and
right ventricular function. The LV ejection fraction at baseline was
66% (65-72) and at in flight was 62% (55-69), (p=0.29). However, a
decrease in LV ejection fraction was observed in two subjects, (76-46%
in one and from 68-35% in another), generating severe ventricular
dysfunction One of these subjects was re-evaluated again with a
second stress echocardiography with exercise stress testing, within
two weeks of the flight, and no ventricular dysfunction was observed.
However, this subject repeated severe ventricular dysfunction (equal
percentage drop in LV ejection fraction), with an apical ballooning
pattern in the second flight.
We were unable to accurately measure pulmonary systolic
pressure in all subjects during flight consequently, it is not reported.
There were no differences in CRP (basal and post flight: 0.73+0.23,
p=0.15), or BNP values (pg 60 before and after the flight).
We analyzed differences between the two individuals who had
ventricular dysfunction and the other subjects. A lower maximum HR
(103.5+123.9+2.1 vs. 11.5 beats/min, p=0.03) and a lower 120 minutes
mean arterial pressure (DBP + 1/3 the differential) was observed in the
two individuals with ventricular dysfunction (58.5+4.6 without LV
dysfunction and 48.5+1.2 mmHg in subjects showed LV dysfunction,
p=0.04) compared to the other subjects without LV dysfunction.
We attempted to determine whether the two patients who
had ventricular dysfunction also presented pre-flight parameters
significantly different from the rest of the population included.
The basal TAS was lower in these two patients (107+2.8 mmHg)
compared to the rest of the subjects (121.4+11.2 mmHg), p=0.06;
and left ventricular diastolic diameter (LVDD) was higher (51.5 vs.
48.4+0.7 mm+1.7 mm, respectively, p=0.04) compared to the rest of
the evaluated population.
The basal EKG holter recorded frequent supraventricular
extrasystolia (2250 beats) in only one patient, which appeared to be
significantly higher during the flight (4298 beats).
Discussion
There are few clinical studies related to changes in cardiovascular
physiology during commercial flights, and most of the available
information is derives from studies using simulation tests in
specialized laboratories [3]. Although the aircraft cabins are
pressurized, they do not provide identical environment to that of sea
level. Besides it is assumed that the cabin pressurization is constant
throughout the journey, variations may exist. The pressurization
changes affect oxygen concentration. Changes in pressure generate
lower arterial blood pressure of oxygen (aO2P). In healthy individuals
at rest, PaO2 is about 98 mmHg at sea level, and may decrease to less
than 60 mmHg at the height a routine commercial flight achieves [3].
Consequent hypoxia, may have effects on vascular hemodynamics
causing vasodilation, increased heart rate and blood pressure [8],
increased myocardial contractility [9] and cardiac index and elevated
pulmonary artery pressure [10,11]. The postulated mechanism for
the increased cardiac output caused by slight levels of hypoxemia is
tachycardia, which reaches a peak in the first 5 minutes to descend to
baseline levels within 20 minutes on land. These effects are not usually
apparent until severe levels of hypoxia (aO2P<40 mmHg and arterial
oxygen saturation <80%) occur [12].
The respiratory system responds to hypoxia increasing the
respiratory rate and tidal volume [13]. Previous studies have
demonstrated an increase in sympathetic activity at altitudes between
1,500 and 3,000 meters high [12]. The sympathetic tone and its
association with hypoxemia, cause a rise in systemic and pulmonary
arterial pressure, with a consequent augment in the left ventricle
diastolic pressure. The increased cardiac output is associated with an
incremental risk of myocardial ischemia, which could further elevate
the left ventricular diastolic pressures. Myocardial ischemia and high
ventricular filling pressure may trigger ventricular arrhythmias [8].
Our study was consistent with other publication that showed
hypoxemia during flight [13]. However, the subjects did not suffer
significant variations in heart rate and a decrease in BP was observed
during flight except for the landing and takeoff periods.
The most relevant finding was that two subjects showed
significant ventricular dysfunction on echocardiography coincidently
with lower values of FC and TAM. Because of the characteristics
of the study we could not determine whether these changes were
the cause or the consequence of decreased LV Fey. In one of the 2
subjects we found a comparable echocardiographic pattern to the
one that is observed in takotsubo syndrome, an acute and transient
cardiomyopathy, characterized by asymmetric impaired ventricular
motility [14]. Ischemia by coronary microvascular dysfunction,
reperfusion injury, microinfarction, fat metabolism disorders
induced by catecholamines or stunning have been suggested to be
involved in the pathophysiology of this syndrome. Catecholamine
levels are two to three times higher in patients with Takotsubo
compared to those who suffer myocardial infarction, describing a
situation of severe emotional stress as the main precipitant [15]. Air
travel might raise anxiety. Ten to 40% of the population refers fear of
flying, situations that can lead to a significant adrenergic discharge
[16]. In our study the two patients with left ventricular dysfunction
had no ischemia in echocardiography stress tests and no evidence of
excessive adrenergic state was detected (they had lower FC and TA
and equal stress score compared to the other subjects). Importantly,
the decline in LV ejection fraction during the flight was not associated
with elevation of proBNP. Troponin was not determined. Another
possible explanation could be the decrease in the microcirculation
in the coronary bed, as it has been demonstrated a decrease in the
sublingual microcirculatory flow in subjects ascending to a maximal
altitude of 77950 m above the sea level [17].
The number of subjects included did not allow us to determine
predictors of hemodynamic patterns. In individuals over 40 years
old, a progressive decrease in PaO2 of 5 mm Hg occurs per every
decade [18]. Patients with chronic lung disease, who present PaO2
at sea level of 60 mmHg and very depressed forced expiratory
volume in spirometry, can achieve risky levels of aO2P even with
the cabin pressurization [19]. Experimental studies in patients with
documented coronary heart disease have shown a significant decrease
in coronary flow reserve which is mainly observed above 2500 m of
altitude [20].
A previous study in men aged between 50-64 years postulated that
hypoxia secondary to high altitude produced atrial and ventricular
ectopy caused alpha and beta adrenergic stimulation [21]. Our
hypothesis was that air travelling could be associated with increased
QTc dispersion and ventricular late potentials, predisposing to
ventricular arrhythmias [22]. Subjects in our study had no ventricular
arrhythmias during the flight.
After the thorax has been opened to perform cardiac surgery it
is inevitable that some air will remain once the wound is closed. It
takes 3 to 10 days for air to be reabsorbed. If any significant amount
of air remains in the pericardial space or in the thoracic cavity it may
expand by up to 60% during a flight. It may be painful or dangerous.
In patients without any of these complications, flying should be
safe after 10-14 days [3,23]. However the Canadian Cardiac Society
Guidelines on Flying recommends that a hemoglobin level of 9 g/l is
a threshold value below which travelling is inadvisable for passengers
who have undergone CABG [24].
The association between inflammation and flight status has not
been studied. Our group suggests that flight stress, prolonged rest
with blood stasis, and hemodynamic changes could be associated with an increased inflammatory tone with high levels of C-reactive
protein. We did not find any difference in CRP, probably because of
the short period between the flight and the sampling or due to the lack
of real time relationship.
Limitations
For economic and safety reasons, a small population of healthy individuals was assessed and does not represent the true risk in the population with cardiovascular disease. Travelling time was less than 4 h, so alterations that occur on longer flights can be assessed.
Conclusion
During the flight variations in HR, TA and oximetry are observed, in healthy and young individuals. Impaired left ventricular function on echocardiography was detected in two subjects. This finding can have greater significance in those patients with history of cardiovascular disease and in those who have undergone a heart surgery. Larger studies are needed to confirm this data.
References
- Possick SE, Barry M. Air travel and cardiovascular disease. J Travel Med. 2004;11(4):243-8.
- Sand M, Bechara FG, Sand D, Mann B. Surgical and medical emergencies on board European aircraft: a retrospective study of 10189 cases. Crit Care 2009;13(1):R3.
- Aerospace Medical Association, Medical Guidelines Task Force. Medical Guidelines for Airline Travel, 2nd edn. Aviat Space Environ Med. 2003;74:A1-19.
- Page RL, Joglar JA, Kowal RC, Zagrodzky JD, Nelson LL, Ramaswamy K, et al. Use of automated external defibrillators by a U.S. airline. N Engl J Med. 2000;343(17):1210-6.
- Higgins JP, Tuttle T, Higgins JA. Altitude and the heart: is going high safe for your cardiac patient? Am Heart J. 2010;159(1):25-32.
- Morton RL, Duster MC, High-altitude cardiopulmonary diseases. Emedicine form webMD. 2015.
- Silverman D, Gendreau M. Medical issues associated with commercial flights. Lancet. 2009;373(9680):2067-77.
- Heistad DD, Abboud FM, Dickinson W. Richards Lecture: Circulatory adjustments to hypoxia. Circulation. 1980;61(3):463-70.
- Vatner SF, Rutherfor JD. Control of the myocardial contractile state by carotid chemo-and baroreceptor and pulmonary inflation reflexes in conscious dogs. J Clin Invest 1978;61(6):1593-601.
- Von Euler US, Liljestrand G. Observations on the pulmonary arterial blood pressure in the cat. Acta Physiol Scand. 1946;12(4):301-20.
- Cutaia M, Rounds S. Hypoxic pulmonary vasoconstriction: physiologic significance,mechanism and clinical relevance. Chest. 1990;97(3):706-18.
- Phillips BA, McConnell JW, Smith MD. The effects of hypoxemia on cardiac output. A dose-response curve. Chest. 1988;93(3):471-5.
- Cottrell JJ, Lebovitz BL, Fennell RG, Kohn GM. In flight arterial saturation: continuous monitoring by pulse oximetry. Aviat Space Environ Med. 1995;66(2):126-30.
- Komamura K, Fukui M, Hirotani S, Iwasaku T, Masuyama T. Takotsubo cardiomyopathy: pathophysiology, diagnosis and treatment. World J Cardiol. 2014;6(7):602-9.
- Abraham J, Mudd JO, Kapur NK, Klein K, Champion HC, Wittstein IS. Stress cardiomyopathy after intravenous administration of cathecolamines and beta-receptors agonists. J Am Coll Cardiol. 2009; 53(15):1320-5.
- Dowdall N. Customer health: a new role for occupational physicians. Occup Med (Lond). 2003;53(1):19-23.
- Martin DS, Goedhart P, Vercueil A, Ince C, Levett DZ, Grocott MP, et al. for the Caudwell Extreme Everest Research Group. Changes in sublingual microcirculatory flow index and vessel density on ascent to altitude. Exp Physiol. 2010;95(8):880–91.
- Smith D, Toff W, Joy M, Dowdall N, Johnston R, Clark L, et al. Fitness to fly for passengers with cardiovascular disease. Heart. 2010;96:(2):1-16.
- Dillard TA, Ewald FW Jr. The use of pulmonary function testing in piloting, air travel, mountain climbing, and diving. Clin Chest Med. 2001;22(4):795-816.
- Wyss CA, Koepfli P, Fretz G, Seebauer M, Schirlo C, Kaufmann PA. Influence of altitude exposure on coronary flow reserve. Circulation. 2003;108(10):1202-7.
- Kujaník S, Snincák M, Galajdová K, Racková K. Cardiovascular changes during sudden ascent in a cable cabin to the moderate altitude. Physiol Res. 2000;49(6):729-31.
- Bettes TN, McKenas DK. Medical advice for commercial air travelers. Am Fam Physician. 1999;60(3):801-8, 810.
- David Smith, William Toff, Michael Joy, Nigel Dowdall, Raymond Johnston, Liz Clark, et al. Fitness to fly for passengers with cardiovascular Disease. Heart 2010;96(2).
- Simpson C, Ross D, Dorian P, Essebag V, Gupta A, Hamilton R, et al. CCS Consensus Conference 2003: Assessment of the cardiac patient for fitness to drive and fly e executive summary. Can J Cardiol 2004;20(13):1313-23.