Research Article
Quantification of Serum Levels of Two Potential Biomarkers and Clinical Features in Osteoarthritis Patients
Takashi Kuroiwa1,2, Hiroshi Ageta2, Daiki Ikeda1, Mitsuhiro Morita1, Kunihiro Tsuchida2 and Harumoto Yamadaz1*
1Department of Orthopaedic Surgery, Fujita Health University Hospital, Toyoake, Aichi, Japan
2Division for Therapies against Intractable Diseases, Institute for Comprehensive Medical Science, Fujita Health University, Toyoake, Aichi, Japan
*Corresponding author: Harumoto Yamadaz, Department of Orthopaedic Surgery, Fujita Health University Hospital, 1-98 Dengakugakubo, Kutsukake, Toyoake, Aichi, 470-1192, Japan
Published: 30 Mar, 2017
Cite this article as: Kuroiwa T, Ageta H, Ikeda D, Morita M,
Tsuchida K, Yamadaz H. Quantification
of Serum Levels of Two Potential
Biomarkers and Clinical Features in
Osteoarthritis Patients. Clin Surg. 2017;
2: 1378.
Abstract
Introduction: Our previous proteomics analysis of human articular cartilage demonstrated that
levels of leukocyte cell-derived chemotaxin 2 (LECT2) and peroxiredoxin 6 (PRDX6) in the cartilage
differed significantly between osteoarthritis (OA) and control patients. LECT2 and PRDX6 levels
were significantly increased and decreased, respectively, in articular cartilages from OA patients. In
the present study, we evaluated the clinical significance of these findings by measuring serum levels
of LECT2 and PRDX6 in OA patients.
Materials and Methods: OA samples were obtained from 73 patients with severe hip and knee OA.
Control samples were obtained from 16 patients with femoral neck fracture (Fr). Serum LECT2 and
PRDX6 levels were measured by enzyme-linked immunosorbent assay.
Results: Serum LECT2 and PRDX6 levels between OA and Fr patients showed the same trends
with our previous study. Intriguingly, serum PRDX6 level was markedly elevated in OA patients
3 months after surgery as compared to before surgery. In addition, serum LECT2 and blood urea
nitrogen (BUN) levels were correlated, while serum PRDX6 level was correlated with BUN, sodium,
and potassium levels.
Conclusion: Our results indicate that PRDX6 will not only be a candidate biomarker for diagnosis
of OA before operation, but also a biomarker for the evaluation of healing process of OA.
Keywords: Osteoarthritis; LECT2; PRDX6
Introduction
Osteoarthritis (OA) is a common disease with a high prevalence among the middle-aged and
elderly that is characterized by degeneration and destruction of articular cartilage, subchondral
changes, and bone proliferation. The 2005 Research on Osteoarthritis Against Disability study
carried out in Japan reported OA in 61.9% of individuals aged 60 years and older (n = 2,282); the
prevalence of knee pain and radiographic knee OA with pain is 32.8% and 26.1%, respectively, in
the general population [1], and the worldwide prevalence of OA was 250/100,000 in 2010 [2]. As the global population ages, the increase in the number of OA patients will have a major effect on medical costs.
Risk factors for OA include advanced age, obesity, gender, and genetic predisposition as well
as local biomechanical factors such as joint instability and trauma and excessive mechanical stress
[3]. However, the pathological mechanism of OA is not well understood. It is thought to first
develop in cartilage, but is associated with secondary synovitis, bone proliferation, and cartilaginous
degeneration. Fibrillation of the cartilage surface occurs in the initial lesion [4] accompanied by biochemical changes such as degeneration of the cartilage matrix, which generates low-molecular
weight substances. Proteases such as matrix metalloproteinase and aggrecanase produced by
chondrocytes are regulated by cytokines such as interleukin 1 (IL-1), IL-6, and tumor necrosis factor
α as well as growth factors such as transforming growth factor β and basic fibroblast growth factor
that are also produced by chondrocytes [5]. Following the cartilage destruction caused by these molecules, secondary synovitisas well as the healing of bone and cartilages induced simultaneously.
OA is typically diagnosed by imaging, including simple radiography and magnetic resonance
imaging (MRI) [6]. The quantitative evaluation of cartilage has recently become possible with MRI; however, the procedure is time-consuming, costly, and is invasive owing to the use of contrast medium. Biochemical changes in the
cartilage in OA begin earlier than diagnosis by conventional imaging
techniques; as such, a more sensitive evaluation method is needed for
early diagnosis [7]. In recent years, biomarkers have been developed and tested for this purpose [8]. Biomarkers have already been applied to the diagnosis of other bone and joint diseases, including
osteoporosis and rheumatoid arthritis (RA) [9,10]. Similarly, serum
or urine biomarkers can potentially be used to measure joint structure
metabolism in the assessment of OA [11].
In 2006, Burden of disease, Investigative, Prognostic, Efficacy of
intervention, and Diagnostic (BIPED) biomarker classification was
proposed for OA with the aim of identifying appropriate markers
for specific purposes [12]. To data there are no OA biomarkers that
are used in general clinical practice, although various studies have
addressed this issue using proteomics and protein arrays to probe
blood, urine, and synovial fluid samples [13]. Collagen degradation
products and complement are useful OA biomarkers; abnormal increases in the expression and activity of complement and the
major cartilage components collagen type II and agree can have been
detected in the synovium and synovial fluid in OA patients [14,15].
In our previous study, we carried out a comprehensive proteomics
analysis of OA patient cartilage using isobaric tags for relative and
absolute quantization (iTRAQ), and identified 76 proteins that
were differentially expressed between OA patients and control subjects. Of these, leukocyte cell-derived chemotaxin 2 (LECT2)
and peroxiredoxin6 (PRDX6) had not been previously reported as
OA biomarkers, and western blot analysis revealed that LECT2 and
PRDX6 levels were significantly increased and decreased, respectively,
in articular cartilages from OA patients [16].
LECT2 is a secretory protein produced in the liver [17] that is
associated with various diseases, including obesity and skeletal
muscle insulin resistance [18], atherosclerotic inflammatory reaction
[19], hepatocellular carcinoma [20], and amyloidosis [21]. PRDX6
is an enzyme with peroxidase and phospholipase A2 activities that
has antioxidant properties [22] and is expressed in various tissues,
including the lungs. PRDX6 has been implicated in the repair of cell
membranes in pulmonary microvascular endothelial cells following
oxidative stress [23].
To identify additional tools for the pathological evaluation of
OA, in the present study we quantified the serum levels of LECT2
and PRDX6 by enzyme-linked immunosorbent assay (ELISA) and
examined the relationship between patients’ physical and blood test
data.
Table 1
Table 1
Clinical data of study subjects. p<0.05, OA vs. Fr group (Student’s t and Mann-Whitney U tests).
SE: Standard Error; Alb: Albumin; ALT: Alanine Transaminase;
AST: Aspartate Transaminase; BMI: Body Mass Index; BUN: Blood Urea Nitrogen; Ca: Calcium; Cl: Chloride; CPK: Creatine Phosphokinase; Cr: Creatinine; CRP:
C-reactive Protein; eGFR: Estimated Glomerular Filtration Rate; γ-GTP: γ-glutamyltranspeptidase; Hb: Hemoglobin; HbA1c: Hemoglobin A1c; HDL: High Density
Lipoprotein-cholesterol; K: Potassium; Na: Sodium; Plt: Platelet; TChol: Total Cholesterol; TG: Triglyceride; TP: Total Protein; WBC: White Blood Cells.
Table 2
Table 2
Correlations among clinical data and serum LECT2 and PRDX6 levels in pre-operative OA patients. Correlations among clinical data and serum LECT2 and
PRDX6 levels in pre-operative OA patients were analyzed using Spearman’s rank correlation for all possible combinations.
Alb: Albumin; ALP: Alkaline Phosphatase;
ALT: Alanine Transaminase; Amy: Amylase; AST: Aspartate Transaminase; BMI: Body Mass Index; BS: Blood Sugar; BUN: Blood Urea Nitrogen; Ca: Calcium Cl:
Chloride; CPK: Creatine Phosphokinase; Cr: Creatinine; CRP: C-Reactive Protein; dBP: Diastolic Blood Pressure; eGFR: Estimated Glomerular Filtration Rate; γ-GTP:
γ-Glutamyltranspeptidase; Hb: Hemoglobin: HbA1c: Hemoglobin A1c; HDL-cholesterol: High Density Lipoprotein-cholesterol; HR: Heart Rate; K: Potassium; Na:
Sodium; P: Phosphorus; Plt: Platelet; TChol: Total Cholesterol; sBP: Systolic Blood Pressure; TBil: Total Bilirubin; TG: Triglyceride; TP: Total Protein; UA: Uric Acid;
WBC: White Blood Cells.
Materials and Methods
This study was approved by the steering committee and was
carried out under the guidelines for clinical studies of Fujita Health
University. Written, informed consent was obtained from all
participants.
Background
In our previous study, we performed comprehensive proteomics
analysis in human articular cartilage from OA or femoral neck
fracture patients 16. To evaluate the clinical significance of these
findings, we collected blood samples from patients with OA or
femoral neck fracture for the control group. The study population
included 89 Japanese subjects (11 male and 78 female; aged 31-91
years) (Table 1) who visited the department of orthopaedic surgery at
Fujita Health University (Aichi, Japan) from April 2015 to December
2015 for surgery. OA samples were obtained from 73 patients with
severe hip or knee OA who underwent total hip or knee replacement.
Patients with osteonecrosis of the femoral head, revision surgery,
and RA cases were excluded. Control samples were obtained from
16 patients with femoral neck fracture (Fr) who underwent bipolar
femoral head replacement surgery or open reduction internal fixation
surgery. Patient characteristics including gender, age, height, weight,
body mass index (BMI), heart rate, systolic blood pressure, diastolic
blood pressure, and past medical history (hypertension (HT) and
diabetes mellitus (DM))were recorded during hospitalization.
Blood sampling
Blood samples were obtained during hospitalization and
immediately centrifuged at 3300 rpm at 4°C for 10 min and stored
at−80°C. The following biochemical parameters were recorded for
each sample: white blood cell count; levels of hemoglobin (Hb),
platelet, total protein (TP), albumin (Alb), total bilirubin, aspartate
transaminase (AST), alanine transaminase, alkaline phosphatase,
γ-glutamyltranspeptidase (γ-GTP), amylase(Amy), creatine
phosphokinase (CPK), triglyceride, total cholesterol (TChol), highdensity
lipoprotein cholesterol, blood urea nitrogen (BUN), creatinine
(Cr), uric acid, sodium (Na), potassium (K), chloride (Cl), calcium
(Ca), phosphorus, hemoglobin A1c, blood sugar, and C-reactive
protein (CRP); and estimated glomerular filtration rate. During the perioperative period, we obtained and examined the blood samples among hospitalization (pre-operation), before discharge (2 weeks
after surgery) and the first consultation (3 months after surgery).
Radiographic finding
Anterior-posterior X-rays of both hips, both knees, and lumbar
spine were obtained for all patients in the standing position. The
severity of OA was radiographically determined according to the
Kellgren–Lawrence (KL) grading system [24].
ELISA
LECT2 and PRDX6 concentrations in serum were determined
using CUS LECT2 (CSB–EL012855HU; Cusabio) and PRDX6 (CSB–
EL018659HU; Cusabio) ELISA kits according to the manufacturer’s
protocol. The lower detection limits of each kit were defined as 0, and
upper limits were defined as values above the measurement range.
Inter-assay precision (precision between assays, 8 samples were tested
for three assays to assess) was demonstrated by coefficient of variation
expressed as a percentage (CV%). Inter-assay precisions of LECT2
and PRDX6 analysis were 15% and 10%, respectively.
Statistical analysis
Data were analyzed using SPSS v.23 (IBM, Tokyo, Japan).
Anthropometric and biochemical data are presented as median ±
standard error. Non-parametric variables were compared between
groups with the Wilcoxon signed-rank, Mann-Whitney U, and
Kruskal-Wallis tests. Correlations between variables were assessed
with Spearman’s rank correlation test. |r| >0.3 and p<0.05 were
considered statistically significant.
Results
We previously carried out a proteomics analysis of human
articular cartilage using the iTRAQ method and found that LECT2
and PRDX6 are differently expressed between OA and Fr patients.
In the current study, we measured the levels of LECT2 and PRDX6
inhuman serum by ELISA and examined these parameters with
respect to clinical data.
The OA group was composed of 73 patients (seven male and 66
female), and the Fr group of 16 patients (four male and 12 female).
Patients’ clinical characteristics are shown in Table 1. Mean ages were
similar between the OA and Fr groups (70.2 ± 1.17 vs. 76.2 ± 3.48,
p=0.05). However, there were statistically significant differences in
weight (55.9 ± 1.21 vs. 46.9 ± 2.87, p=0.002) and BMI (24.2 ± 0.49
vs. 19.4 ± 0.93, p<0.001; Table 1). Four females were KL grade3, and
all other patients were KL grade 4 in the OA group. T he two groups
differed in terms of Hb, TP, Alb, CPK, CRP, AST, γ-GTP, BUN, Cr,
TChol, Na, Cl, and Ca levels (p<0.05).
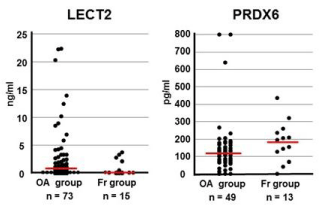
Prior to operation, LECT2 and PRDX6 levels tended to increase
and decrease, respectively, in serum from OA patients. However, their
levels did not show statistically significant difference between the OA
and Fr groups (LECT2: 0.71 ± 0.58 vs. 0 ± 0.36, p=0.44, PRDX6:119.1
± 23.8 vs. 195.4 ± 32.4, p=0.10, Figure 1). To determine whether the
operative procedure affected these parameters, we compared the
serum levels of LECT2 and PRDX6 in the OA group pre-operation
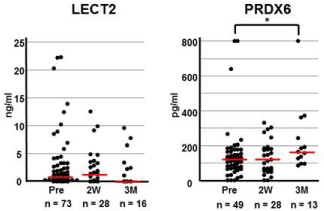
(Pre), 2 weeks after surgery (2W) and 3 months after surgery (3M).
Serum LECT2 levels were similar at Pre, 2W, and 3M. In contrast,
serum PRDX6 level was higher at 3M than at Pre (p=0.02, Figure
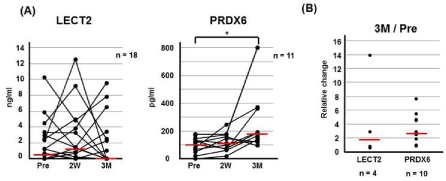
2). The same trends were observed in individual patients (p=0.002,
Figure 3(a)). In PRDX6 analysis on individual patient, the mean value
of the increasing rate (3M/Pre) and CV% between Pre and 3M were 2.7 and 41.7%, respectively. Inter-assay precision of PRDX6 analysis was 10%, indicating that this change was significant (Figure 3(b)).
We investigated whether serum LECT2 and PRDX6 levels were
correlated with clinical (anthropometric and biochemical) data in
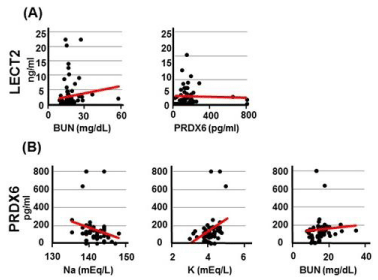
the Pre OA group (Table 2). We found that serum LECT2 and BUN
levels were correlated (r=0.310, p=0.008). There were no differences
between LECT2 and PRDX levels (r=0.153, p=0.294, Figure 4(a)). On
the other hand, the serum level of PRDX6 was correlated with BUN (r=0.316, p=0.027), Na (r=−0.359, p=0.011), and K (r=0.364, p=0.010, Figure 4(b)) levels.
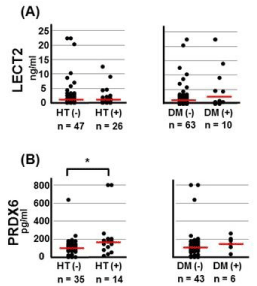
OA patients with and without HT differed in terms of serum
PRDX6 level (p=0.02); on the other hand, there was no difference
between OA patients with and without DM. A history of HT or DM
had no effects on serum LECT2 level (Figure 5(a)).
Figure 1
Figure 1
Comparison of serum LECT2 and PRDX6 levels between preoperative
OA and Fr patients. Serum LECT2 and PRDX6 levels were
measured by ELISA. Horizontal red bars indicate median values for each
group. LECT2: OA, n= 73 and Fr, n= 15 (p=0.44, Mann-Whitney U test).
PRDX6: OA, n=49 and Fr, n=13 (p=0.10; Mann-Whitney U test).
Figure 2
Figure 2
Changes in serum LECT2 and PRDX6 levels in OA patients after
operation. Serum LECT2 and PRDX6 levels were measured pre-operation
(Pre), 2 weeks after surgery (2W), and 3 months after surgery (3M).
Differences between the three groups were evaluated with the Kruskal-Wallis
test. Serum PRDX6 level was elevated in the 3M as compared to the Pre
group (p=0.02; Mann-Whitney U test). Horizontal red bars indicate median
values for each group.
Figure 3
Figure 3
Changes in serum LECT2 and PRDX6 levels in individual patients.
(A) Each line represents each patient; horizontal red bars show median
value for each group. Pre, pre-operation; 2W, 2 weeks after operation; 3M,
3 months after operation. SerumLECT2 and PRDX6 levels were compared
with the Kruskal-Wallis test (LECT2, p=0.44 and PRDX6, p=0.07). PRDX6
but not LECT level differed significantly between Pre and 3M groups with the
Wilcoxon signed-rank test (p=0.002 and p=0.53, respectively). (B) The rate
of change between Pre and 3M. Zero value at Pre and 3M was not included
in this data plot. Each plot represents each patient; horizontal red bars show
mean value for each group.
Figure 4
Figure 4
Correlation between clinical data and serum LECT2 (A) and
PRDX6 (B) levels in pre-operative OA patients. Correlations between clinical
data and serum LECT2 and PRDX6 level were analyzed with Spearman’s
rank correlation test for all possible combinations. LECT2 was positively
correlated with BUN (r=0.310, p=0.008) level. PRDX6 was positively
correlated with BUN (r=0.316, p=0.027) and K (r=0.364, p=0.010) level and
negatively correlated with Na (r =−0.359, p=0.011).
Figure 5
Figure 5
Serum LECT2 (A) and PRDX6 (B) levels in pre-operative OA
patients with/without hypertension (HT) and diabetes mellitus (DM). OA
patients with and without HT differed significantly with respect to PRDX6
levels (p= 0.02; Mann-Whitney U test).
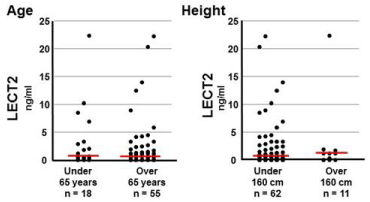
Figure 6
Figure 6
Comparison of serum LECT2 levels by age and height in preoperative
OA patients. Age, LECT2: median ± standard error (<65 years,
0.92 ± 1.34; >65 years, 0.71 ± 0.63); Mann-Whitney U test: p=0.23. Height,
LECT2: median ± SE (<160 cm, 0.68 ± 0.59; >160 cm, 1.18 ± 1.98); Mann-
Whitney U test: p=0.79. Horizontal red bars show median values for each
group.
Discussion
In the present study, we evaluated the utility of serum LECT2 and
PRDX6 levels as candidate biomarkers of OA. This is the first report
to quantify the serum concentrations of LECT2 and PRDX6 in OA
patients. Although there were no statistically significant difference
in serum LECT2 and PRDX6 levels between OA and Fr patients,
the same trends with our previous study were observed. Serum
biomarkers are modulated or metabolized in various tissues, while
changes in cartilage or synovial fluid may not be reflected in serum
[25]. It is therefore possible that we were unable to detect significant differences due to the effects of normal joints or other tissues on
serum content. Interestingly, we found that serum PRDX6 levels were
significantly elevated 3 months after as compared to before surgery.
We also found that serum LECT2 level was correlated with BUN, and
that serum PRDX6 level was correlated with BUN, Na, and K levels.
In addition, serum PRDX6 levels were also higher in OA patients with
than in those without HT.
Prognosis analysis of acute liver failure have revealed that serum
LECT2 levels are lower in patients who died of acute liver failure than
in survivors [26] and are elevated in patients with obesity and fatty
liver [27]. Although there are no reports on the relationship between
LECT2 and OA, in a mouse RA model, the exacerbation of arthritis
resulting from LECT2 deficiencywas improved by LECT2 over
expression [28], suggesting that LECT2 may have an inhibitory effect
on inflammation in RA. OA is also characterized by inflammation,
although it is less severe than in RA [29]. Our previous finding that
in human cartilage, LECT2 expression was higher in OA than in Fr
patients suggested that LECT2 levels are upregulated as a biological
defense mechanism to inhibit inflammation. Although it was not
statistically significant, we observed a similar trend in serum samples
from OA patients in the present study. We expect that the difference
between OA and Fr patients would be significant using a larger
sample size. We also found that serum LECT2 and BUN levels were
correlated. Urea nitrogen is a waste product of protein metabolism
and reflects renal functioning as serum Cr level [30]. Thus, although
serum LECT2 and Cr levels were not correlated, LECT2 may
nonetheless be associated with renal function.
Serum PRDX6 level is approximately five times higher in breast
cancer patients than in healthy individuals [31]. We previously
reported that PRDX6 is down regulated in the cartilage of OA
patients. Similarly, in the present study, PRDX6 levels were lower in
the serum of OA as compared to Fr patients, although the difference
was not statistically significant. PRDX6 is an antioxidant enzyme with
both peroxidase and phospholipase A2 activities. Reactive oxygen
species (ROS) production is increased in OA [32,33]. Interestingly,
PRDX5 levels are upregulated and protects against ROS in OA
cartilage [34], and PRDX5 knockdown stimulates osteoarthritic
chondrocyte apoptosis and inhibits the scavenging of endogenous
ROS [35]. The PRDX family is classified into three subgroups (2-Cys,
atypical 2-Cys, and 1-Cys) according to the number and position of
cysteine residues that participate in catalysis. PRDX5 is an atypical
2-Cys whereas PRDX6 is a 1-Cys member [36]. Thus, differences
between these two proteins in terms of effects on cartilage may be attributable to structural differences. In the present study, we found that serum level of PRDX6 was markedly elevated 3 months after as
compared to before surgery in OA patients; this may be explained by
the fact that the ROS-induced reduction in PRDX6 expression was
restored by surgery.
In this study, serum level of PRDX6 was correlated to BUN, Na,
and K levels. Renal failure is associated with BUN and K concentrations
in blood, and the prevalence of OA is higher among cases of renal
failure [37]; indeed, approximately, 37% of OA patients have renal
impairment as compared to only 27% of the general population [37].
Thus, although we did not find any correlation between serum levels
of PRDX6 and Cr, PRDX6 may be associated with renal function.
We also found that serum PRDX6 level was higher in OA patients
with than in those without concurrent HT. Although the relationship
between OA and HT remains unclear, one U.S. study reported that
40% of OA patients have HT [37]. On the contrary, in Korea, OA
prevalence was significantly lower in OA patients with HT than
in those with OA only after adjusting for patient background, and
longer duration of HT attenuated this correlation [38]. Thus, the
presence of HT may affect the level of PRDX6, which has a protective
effect against OA.
Conclusion
This study is the first to report the measurement of serum LECT2 and PRDX6 by ELISA in OA patients. LECT2 and PRDX6 levels tended to increase and decrease, respectively, in serum from OA patients. Furthermore, we found that serum PRDX6 level was significantly elevated 3 months after as compared to before surgery. In addition, serum PRDX6 level was correlated with BUN, Na, and K levels. Given that serum level of PRDX6 was linked to the healing of OA, PRDX6 may be useful as a candidate biomarker not only for diagnosis of OA but also for monitoring OA improvement.
Acknowledgment
We thank members of the orthopaedic surgery department at Fujita Health University Hospital for providing of clinical data. We also thank Mrs. Yamaguchi and Mrs. Tajima for technical assistance. This work was partly supported by the Program for the Strategic Research Foundation at Private Universities and an Intramural Research Grant (26-8) for Neurological and Psychiatric Disorders of NCNP. All authors declare that there is no conflict of interest.
References
- Muraki S, Oka H, Akune T, Mabuchi A, En-yo Y, Yoshida M, et al. Prevalence of radiographic knee osteoarthritis and its association with knee pain in the elderly of Japanese population-based cohorts: the ROAD study. Osteoarthritis Cartilage. 2009;17:113743.
- Murray CJ, Vos T, Lozano R, Naghavi M, Flaxman AD, Michaud C, et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2197-2223.
- Felson DT, Lawrence RC, Dieppe PA, Hirsch R, Helmick CG, Jordan JM, et al. Osteoarthritis: new insights. Part 1: the disease and its risk factors. Ann Intern Med. 2000;133(8):635-46.
- Panula HE, Hyttinen MM, Arokoski JP, Langsjo TK, Pelttari A, Kiviranta I, et al. Articular cartilage superficial zone collagen birefringence reduced and cartilage thickness increased before surface fibrillation in experimental osteoarthritis. Ann Rheum Dis. 1998;57:237-45.
- Loeser RF. Molecular mechanisms of cartilage destruction: mechanics, inflammatory mediators, and aging collide. Arthritis Rheum. 2006;54:1357-60.
- Hayashi D, Roemer FW, Guermazi A. Imaging for osteoarthritis. Ann Phys Rehabil Med. 2016;59(3):161-9.
- Ishijima M, Watari T, Naito K, Kaneko H, Futami I, Yoshimura-Ishida K, et al. Relationships between biomarkers of cartilage, bone, synovial metabolism and knee pain provide insights into the origins of pain in early knee osteoarthritis. Arthritis Res Ther. 2011;13:22.
- Bay-Jensen AC, Reker D, Kjelgaard-Petersen CF, Mobasheri A, Karsdal MA, Ladel C, et al. Osteoarthritis year in review 2015: soluble biomarkers and the BIPED criteria. Osteoarthritis Cartilage. 2016;24:9-20.
- Diem SJ, Joffe H, Larson JC, Tsai JN, Guthrie KA, LaCroix AZ, et al. Effects of escitalopram on markers of bone turnover: a randomized clinical trial. J Clin Endocrinol Metab. 2014;99(9):E1732-7.
- Taylor P, Gartemann J, Hsieh J, Creeden J. A systematic review of serum biomarkers anti-cyclic citrullinated Peptide and rheumatoid factor as tests for rheumatoid arthritis. Autoimmune Dis. 2011;2011: 815038.
- Garnero P, Piperno M, Gineyts E, Christgau S, Delmas PD, Vignon E. Cross sectional evaluation of biochemical markers of bone, cartilage, and synovial tissue metabolism in patients with knee osteoarthritis: relations with disease activity and joint damage. Ann Rheum Dis. 2001;60:619-26.
- Bauer DC, Hunter DJ, Abramson SB, Attur M, Corr M, Felson D, et al. Classification of osteoarthritis biomarkers: a proposed approach. Osteoarthritis Cartilage. 2006;14(8):723-7.
- Blanco FJ. Osteoarthritis year in review 2014: we need more biochemical biomarkers in qualification phase. Osteoarthritis Cartilage. 2014;22:2025-32.
- Elsaid KA, Chichester CO. Review: Collagen markers in early arthritic diseases. Clin Chim Acta. 2006;365(1-2):68-77.
- Wang Q, Rozelle AL, Lepus CM, Scanzello CR, Song JJ, Larsen DM, et al. Identification of a central role for complement in osteoarthritis. Nat Med. 2011;17(12):1674-9.
- Ikeda D, Ageta H, Tsuchida K, Yamada H. iTRAQ-based proteomics reveals novel biomarkers of osteoarthritis. Biomarkers. 2013;18(7):565-72.
- Yamagoe S, Yamakawa Y, Matsuo Y, Minowada J, Mizuno S, Suzuki K. Purification and primary amino acid sequence of a novel neutrophil chemotactic factor LECT2. Immunol Lett. 1996;52:9-13.
- Lan F, Misu H, Chikamoto K, Takayama H, Kikuchi A, Mohri K, et al. LECT2 functions as a hepatokine that links obesity to skeletal muscle insulin resistance. Diabetes. 2014;63(5):1649-64.
- Hwang HJ, Jung TW, Hong HC, Seo JA, Kim SG, Kim NH, et al. LECT2 induces atherosclerotic inflammatory reaction via CD209 receptor-mediated JNK phosphorylation in human endothelial cells. Metabolism. 2015;64:1175-82.
- Okabe H, Delgado E, Lee JM, Yang J, Kinoshita H, Hayashi H, et al. Role of leukocyte cell-derived chemotaxin 2 as a biomarker in hepatocellular carcinoma. PLoS One. 2014;9(6):e98817.
- Khalighi MA, Yue A, Hwang MT, Wallace WD. Leukocyte chemotactic factor 2 (LECT2) amyloidosis presenting as pulmonary-renal syndrome: a case report and review of the literature. Clin Kidney J. 2013;6: 618-21.
- Kim TS, Sundaresh CS, Feinstein SI, Dodia C, Skach WR, Jain MK, et al. Identification of a human cDNA clone for lysosomal type Ca2+-independent phospholipase A2 and properties of the expressed protein. J Biol Chem. 1997;272(4):2542-50.
- Li H, Benipal B, Zhou S, Dodia C, Chatterjee S, Tao JQ, et al. Critical role of peroxiredoxin 6 in the repair of peroxidized cell membranes following oxidative stress. Free Radic Biol Med. 2015;87:356-65.
- Kellgren JH, Lawrence JS. Radiological assessment of osteo-arthrosis. Ann Rheum Dis. 1957;16(4):494-502.
- Li L, Jiang BE. Serum and synovial fluid chemokine ligand 2/monocyte chemoattractant protein 1 concentrations correlates with symptomatic severity in patients with knee osteoarthritis. Ann Clin Biochem. 2015;52:276-282.
- Sato Y, Watanabe H, Kameyama H, Kobayashi T, Yamamoto S, Takeishi T, et al. Serum LECT2 level as a prognostic indicator in acute liver failure. Transplant Proc. 2004;36(8):2359-61.
- Okumura A, Unoki-Kubota H, Matsushita Y, Shiga T, Moriyoshi Y, Yamagoe S, et al. Increased serum leukocyte cell-derived chemotaxin 2 (LECT2) levels in obesity and fatty liver. Biosci Trends. 2013;7(6):276-83.
- Okumura A, Saito T, Otani I, Kojima K, Yamada Y, Ishida-Okawara A, et al. Suppressive role of leukocyte cell-derived chemotaxin 2 in mouse anti-type II collagen antibody-induced arthritis. Arthritis Rheum. 2008;58:413-21.
- Bonnet CS, Walsh DA. Osteoarthritis, angiogenesis and inflammation. Rheumatology (Oxford). 2005;44(1):7-16.
- Lyman JL. Blood urea nitrogen and creatinine. Emerg Med Clin North Am. 1986;4(2):223-33.
- Liu FJ, Wang XB, Cao AG. Screening and functional analysis of a differential protein profile of human breast cancer. Oncol Lett. 2014;7(6):1851-1856.
- Regan E, Flannelly J, Bowler R, Tran K, Nicks M, Carbone BD, et al. Extracellular superoxide dismutase and oxidant damage in osteoarthritis. Arthritis Rheum. 2005;52(11):3479-91.
- Collins JA, Moots RJ, Winstanley R, Clegg PD, Milner PI. Oxygen and pH-sensitivity of human osteoarthritic chondrocytes in 3-D alginate bead culture system. Osteoarthritis Cartilage. 2013;21:1790-98.
- Wang MX, Wei A, Yuan J, Trickett A, Knoops B, Murrell GA. Expression and regulation of peroxiredoxin 5 in human osteoarthritis. FEBS Lett. 2002;531(2):359-62.
- Ma Y, Li R, Zhang Y, Zhou L, Dai Y. Knockdown of peroxiredoxin 5 inhibits the growth of osteoarthritic chondrocytes via upregulating Wnt/β-catenin signaling. Free Radic Biol Med. 2014;76:251-60.
- Kang SW, Baines IC, Rhee SG. Characterization of a mammalian peroxiredoxin that contains one conserved cysteine. J Biol Chem. 1998;273(11):6303-11.
- Singh G, Miller JD, Lee FH, Pettitt D, Russell MW. Prevalence of cardiovascular disease risk factors among US adults with self-reported osteoarthritis: data from the Third National Health and Nutrition Examination Survey. Am J Manag Care. 2002;8:383-91.
- Bae YH, Shin JS, Lee J, Kim MR, Park KB, Cho JH, et al. Association between Hypertension and the Prevalence of Low Back Pain and Osteoarthritis in Koreans: A Cross-Sectional Study. PLoS One. 2015;10(9):e0138790.