Review Article
Cardiac Surgery for Arrhythmias
Alberto Pozzoli1, Ottavio Alfieri1, Paolo Della Bella2 and Patrizio Mazzone2*
1Department of Heart Surgery, San Raffaele University Hospital, Italy
2Department of Arrhythmia and Electrophysiology Laboratories, San Raffaele University Hospital, Italy
*Corresponding author: Patrizio Mazzone, Department of Arrhythmology and Cardiac Pacing Unit San Raffaele University-Hospital, Via Olgettina 60, 20132 Milan, Italy
Published: 07 Mar, 2017
Cite this article as: Pozzoli A, Alfieri O, Bella PD, Mazzone
P. Cardiac Surgery for Arrhythmias. Clin
Surg. 2017; 2: 1351.
Abstract
Surgical treatment for cardiac arrhythmias was initiated in 1968 with the first successful surgical division of an accessory atrioventricular connection for the Wolff-Parkinson-White Syndrome. Subsequent surgical procedures were the left atrial isolation procedure and the right atrial isolation procedure for the automatic atrial tachycardias, corridor procedure and the Maze procedure, developed by James Cox, to cure atrial fibrillation. More, the right ventricular disconnection for arrhythmogenic right ventricular tachycardia, the encircling endocardial ventriculotomy, transitional procedures and the Dor procedure for ischemic ventricular tachycardia should be mentioned. Nowadays, due to the significant improvements of the endovascular percutaneous ablations, the Maze IV, thoracoscopic surgical pulmonary isolation and the Dor procedure remain the most performed surgical interventions. The last four decades of research in the field of cardiac arrhythmia surgery provided the basis and source of knowledge for the development of these transcatheter techniques and hybrid approaches.
Introduction
Preamble
During the past 35 years, surgery has played a pivotal role in the elucidation of the anatomic
and electrophysiological abnormalities causing cardiac tachyarrhythmias. The development of
electrophysiological cardiac mapping and the surgical techniques which followed provided the
knowledge to cure the majority of pharmacologic refractory arrhythmias. This knowledge mainly
derived from the approaches to Wolff-Parkinson-White syndrome and to the atrioventricular
nodal reentry tachycardia [1]. Despite the exponential progress in interventional electrophysiology,
arrhythmia surgery remains fundamental into the 21st century. The first extensive electrophysiological
mapping of human atrial fibrillation (AF) and the subsequent development of the Maze procedure
(late ‘80s) to cure AF was a miliar stone for the surgical and cardiovascular treatment of all cardiac
arrhythmias [2-4].
Wolff-Parkinson White Syndrome
Gaskell was the first to demonstrate that electrical activity propagated from the atrium to the ventricle via myocardial tissue rather than nerves, reported within his studies on the turtle heart in 1883 [5]. Thereafter was Kent to identify accessory atrioventricular connections responsible for the WPW syndrome (“Kent bundles”) [6]. The conduction system of the heart was characterized in a systematical analysis in 1906 in Germany (atrioventricular node, His bundle, bundle branches and the Purkinje system).It was with Dirk Durrer in 1967, in Amsterdam, that was performed the first intraoperative mapping in a patient with the WPW syndrome, he demonstrated electrical conduction across the atrioventricular groove in the region of ventricular pre excitation [7]. The same year Dr. Howard Burchell of the Mayo Clinic was able to abolish ventricular pre excitation by injecting procaineamide into the AV groove, at that site [8]. However, the first surgical attempt was made in 1968 at the Duke University, the successful division of a right free-wall accessory pathway [9]. There was literally an explosion of surgical procedures to divide accessory pathways during the ‘80s (more than thousands per year). However, they become very shortly history of arrhythmia surgery after the introduction of the radiofrequency catheter techniques in 1990.
Atrioventricular Nodal Reentry Tachycardia
Prior to 1982, the only available surgical therapy for refractory atrioventricular nodal reentry tachycardia was the interruption of His bundle, protecting the ventricles from the AV node reentry, but requiring permanent pacemaker implantation due to advanced blocks [10]. Likewise WPW, the subsequent advent in 1982 of the trans catheter radiofrequency fulguration of the AV node immediately replaced the open-heart surgical method.
Figure 1
Automatic Atrial Tachycardia
These tachycardias have a focal origin and are due to automaticity, they are not deriving from a mechanism of reentry. The right atrium or left atrium is the most common foci. At the very beginning of this specialty, the intraoperative anesthesia was so strong to suppress the onset of these automatic atrial arrhythmias. Therefore, it was impossible to perform a precise surgical intervention. The only available approach was to blindly ablate the atrioventricular node in association to the His bundle and insert a ventricular pacemaker. It was Williams to understand the possibility to confine this automaticity to the left atrium, isolating it from the right atrium and the ventricles, without imparing the physiological atrio-ventricular function. The left atrial isolation was eventually performed after experimental studies on humans and reported for the first time in 1980 [11]. This study was fundamental as an essential step towards the development of the Maze procedure. In particular, the authors understood the essential role of the coronary sinus in the atrioventricular conduction, because its walls are themselves made of conductive fibers (not only surrounded by), becoming a mandatory target to achieve the complete isolation of the left atrium. It provided also the evidence that physiologic cardiac output is maintained if the right atrium and the right ventricle beat synchronized. During the ‘80s, the general improvements in anesthesia techniques and the development of computerized intraoperative mapping systems made it possible to map the sites of origin of automatic atrial tachycardias [12]. From a patho physiological standpoint, most of these supraventricular arrhythmias are multicentric also on the right, requiring the isolation of the entire atrium sparing the sinus node.
Non ischemic Ventricular Tachycardia
The vast majority of non-ischemic ventricular tachycardias are caused by congenitalarrhythmogenic right ventricular dysplasia, firstly described by Fontaine and Guiraudonin 1979 [13]. Clinically, patients have sustained ventricular tachycardia originating from one or more injured areas of the right ventricle, impairing the right ventricular function. The pioneeristic experimental analyses of James Cox and coworkers were again the very first to study this pathologic condition [14]. After some initial attempts on the right ventricular outflow tract, they performed an isolation of the free wall of right ventricle, guided by mapping, to confine this insidious arrhythmia within the ventricular wall itself, with the rest of the heart being able to contract in sinus rhythm. Only a small series of patients was treated, because the advent of heart transplantations and of implantable defibrillators eliminated this dramatic approach, after 1985. The physiology learned from this operation contributed to a better understanding of the interplay between the right ventricle, the septum, and the left ventricle in patients with heart failure.
Ischemic Ventricular Tachyarrhythmias
The reentrant basis of ischemic ventricular tachyarrhythmias, due to myocardial ischemic injuries, was confirmed by the experimental studies made by Cox and colleagues [15,16]. The advent of myocardial revascularization during the ‘60s was expected to improve the myocardial ischemia and so far, the ventricular arrhythmia. Simple resection of damaged myocardium and/or the revascularization alone were not able to cure or even control the ventricular tachycardia, resulting in excessive operative mortality [17]. The lack of knowledge in the genesis and perpetuation of ischemic ventricular tachyarrhythmia was particularly evident at that time. Several groups began to approach the problem in a more direct surgical manner. In 1969, Cox’s group and Kaiser and colleagues independently reported intraoperative mapping to localize the area of ischemic injury [18]. The first attempts to ablate in the area of the tachycardia guided by intraoperative mapping were done in 1975 by Wittig and by Gallagher [19,20]. After Guiraudon, who reported in five patients the encircling endocardialventriculotomy, it was Josephson to describe the endocardial resection procedure, from which derived the mainstay for the surgical management till mid-1980s, when new surgical resection and repair of left ventricular aneurysms, reshaping the ventricle, came out [21-23]. The functional improvement of the left ventricle was excellent when compared to the technique of simply resecting the aneurysm and closing the ventricle. In particular, this approach was able to cure ischemic ventricular tachycardia without the need for intraoperative mapping. Nowadays, the interventional catheter techniques provide good outcomes without being too invasive [24].
Atrial Fibrillation
Atrial fibrillation is associated with significant morbidity and
mortality due to its three detrimental sequelae and symptoms, which
are not trivial:
• Irregularly irregular heartbeat, which causes patient
discomfort and anxiety
• Loss of synchronous atrioventricular contraction, impairing
hemodynamics and leading to different degrees of congestive heart
failure
• Stasis of blood flow in the left atrium, which increases the
vulnerability to thrombo embolisms.
Although the ventricular response rate can usually be controlled
medically, it is important to recognize that the atria are still
fibrillating, and therefore, the detrimental sequelae associated with
AF persist. However, in many patients the pharmacologic therapy
results suboptimal and several surgical procedures were introduced
in the 1980s to ablate AF. After the left a trial isolation procedure
Scheinman introduced in 1982 the catheter fulguration of the
His bundle as a means of controlling the irregular cardiac rhythm
associated any kind of refractory supraventricular tachyarrhythmias
[25]. Catheter fulguration was eventually abandoned, favoring less traumatic radiofrequency techniques which are still used nowadays.
However, this approach requires the insertion of a permanent
ventricular pacemaker, which restores a physiologic ventricular
rhythm while the atria are fibrillating. Obviously, this procedure
is suboptimal and does not prevent the hemodynamic impairment
due to loss of atrioventricular synchrony and thromboembolisms. In
1985, Guiraudon and coworkers described the corridor procedure
for the treatment of AF, an open-heart technique that isolated a strip
of atrial septum (the “corridor”) which included the sinoatrial node
and the atrioventricular node, thus allowing the sinus node to pace
the ventricles [26]. Meanwhile both the atria continued to fibrillate,
isolated from the septal corridor and loosing the atrioventricular
synchrony. This technique was soon abandoned due to its intrinsic
limitations. In this cultural context, the experimental studies on the
canine models performed by Dr. Cox tested several lesion sets to
understand the response of both the atria. One of the first attempts
was to isolate the posterior wall of the left atrium and the orifices
of the four pulmonary veins (PVs) from the rest of the heart, with
a long incision. The final result was not effective as expected, but it
had particular relevance since many years later, 1998, Haissaguerre
documented here the origin of paroxysmal AF. It was evident at that
time that the surgical cure of AF would require a more complete
understanding of the underlying electrophysiology. However,
these studies showed the three mechanisms essential to understand supraventricular arrhythmias:
• A portion of atrial muscle harboring one or more macroreentrant
circuits
• The passive atrial conduction in the portions close to the
macro-reentrant circuit(s)
• The role of the atrioventricular node for the atrioventricular
conduction.
These three electrophysiological characteristics define a spectrum
of atrial arrhythmias, extending from atrial flutter, through several
forms of transitional arrhythmias, to permanent AF [27]. The Maze
concept was developed in the laboratory at Washington University,
St Louis, to provide the surgical treatment of AF. The real objective
of the Maze procedure can be described as creating atrial lesions
that preclude the ability of the atria to fibrillate by preventing the
possibility of macro-reentrant circuits formation. After several
experimental attempts, Cox developed the Maze I and II, but they
were too complex to be performed from a technical standpoint and
the decreased left atrial contraction was an issue. The modifications
of these techniques ended in the Maze III procedure which soon
became the surgical technique of choice for the cure of refractory
atrial flutter and AF [27-29]. Between 1987 and1992, more than 30
patients had the Maze I procedure while only 15 patients had the
Maze II procedure. From then on, due to the reasons mentioned
before, the Maze III became the standard of care and more than 300
patients had undergone the Maze III procedure for the treatment
of atrial flutter and/or AF. The operative mortality rate was around
3%. The most common complications following the Maze procedure
were the postoperative arrhythmias, usually atrial flutter or AF,
which occurred in 37% of patients. The careful closure of the left
atrial appendage during the procedure was an essential surgical step,
to abate the risk of postoperative strokes. In fact, one of the benefits
of the Maze is that it essentially abolishes the strokes associated
with AF. The long-term stroke rate following the Maze procedure
is 0.1% per year [30]. Overall, 15% of patients required pacemakers
postoperatively, mostly due to sick sinus syndrome (abnormal
sinoatrial nodes “unmasked” by eliminating the patient’s AF). The
long-term follow-up of these patients revealed a 15-year cure rate of
over 95% and other groups that have adhered to the concept of the
Maze achieved similar results [31-33]. Groups that have chosen to
modify the procedure without following the concepts of the Maze
procedure have suffered poorer results [4,34]. Overall, the need for
postoperative pacemakers is higher when the procedures were done
in full sternotomy, owing to extensive dissections, in comparison to
the rate observed with the minimally invasive approach, reported in
2000 employing cryoenergy [32,35]. Despite the obvious efficacy of
the Maze procedure, it has never been widely adopted by surgeons
and cardiologists because of its complexity. In 2004, Damiano and
coworkers replaced most of the incisions originally done in the Maze
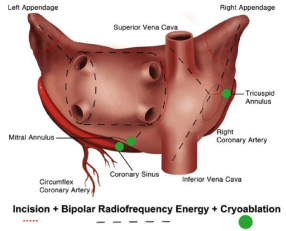
III procedure with cryoenergy and bipolar radiofrequency, developing
the so-called ablative Maze IV (Figure 1). This technique was faster,
simpler and it was possible a minimally invasive approach without
decreasing the efficacy [36]. These ablation-assisted procedures have
greatly expanded the field of AF surgery in the past decade, expecially
the treatment of AF concomitant to structural valve disease [37].
However, nowadays several ablation technologies exist, each with
its consequent advantages and disadvantages. Whatever the ablative
technology, it must meet the bidirectional conduction block across
the line of the ablative lesion as primary endpoint, becoming the benchmark for technology validation [38-40]. The laboratories of Dr.
Damiano showed that a transmural lesion is required, because even
small gaps in ablation lines (Figure 2) can conduct both sinus and
fibrillatory impulses, leading to reconnections [41]. Since the Maze
IV incorporates the lesions of the Cox-Maze III, the clinical results
showed that this modified procedure has significantly shortened
operative time while maintaining the high success rate of the original
Cox-Maze III procedure and normalizing the left ventricular function
in every patient affected by tachycardiomyopathy [42-43]. The late
outcomes of the Cox Maze IV remain good after 5 years, around 80%
with the use of antiarrhythmics. More, the type of preoperative AF or
the addition of a concomitant procedure did not affect late success.
These results remain superior to those reported for catheter ablation
and other forms of surgical ablation, especially for patients with
persistent or long-standing AF [44].
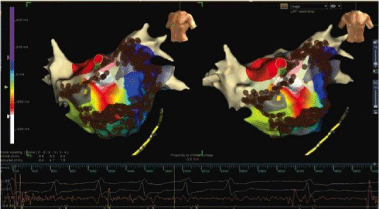
Figure 2
Figure 2
Electrophysiological findings and ablations. Pulmonary vein gaps
leading to reconnection treated with re isolation and sinus rhythm restoration.
Figure 3
Figure 3
Left atrial lesion set performed with unipolar radiofrequency
concomitant to mitral valve disease. The initial left atrial lesion set included
encircling of the pulmonary veins, a single interpulmonary line and two linear
ablations towards the appendage and the mitral annulus.
Significant Contributions to Atrial Fibrillation Surgery by Japanese Surgeons
Cryoenergy Maze II procedure
In Japan, Kosakai et al. modified the original Maze II procedure
using cryoablation instead of several cut-and-sew incision lines. This
Maze procedure was introduced in 1992, the first Maze procedure
in Japan. The authors demonstrated high efficacy for eliminating
persistent AF associated with mitral valve disease; the rate of
elimination of persistent AF at that time using this strategy reported
by authors was, at least at the very beginning, around 80% [3].
Left atrial Maze procedure and PVs box isolation
Sueda et al. reported the efficacy of the left atrial Maze procedure
for treating persistent AF associated with solitary mitral valve disease
[45]. The authors performed the left atrial Maze procedure consisting
of whole isolation of the PVs and cryoablation towards the mitral
valve isthmus and orifice of the left atrial appendage with excision
of the left atrial appendage for mitral valve disease. This procedure,
when combined with tricuspid isthmus cryoablation, is effective for
preventing atrial flutter and eliminating persistent AF associated with
both mitral and tricuspid valvular disease [45]. Because common atrial
flutter via the right atrial isthmus is a risk factor after treatment with
the left atrial Maze procedure, ablation should be administered at the
right atrial isthmus to prevent further postoperative atrial flutter. This
combined procedure was named the mini-Maze procedure by Cox
and was nearly as effective as the full-Maze procedure for managing
longstanding persistent AF.
Ablative Strategies to Treat Atrial Fibrillation Concomitant to Valve Surgery
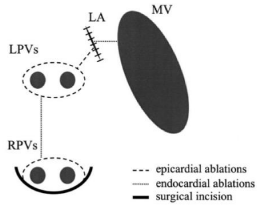
Largely because of the availability of the radiofrequency catheters and the initial reports of Haissaguerre’s reports, heart surgeons were inspired to use the radiofrequency catheters intraoperatively, to ablateendo/epicardially (Figure 3) the AF in patients who were already undergoing surgery for mitral valve disease [3,46-47]. However, the ablative lesions were frequently not transmural, thereby offering at best only a temporary electrical efficacy, with a significant failure rate. The ablative devices designed to solely encircle the PVs, even when completely transmural, had efficacy in around 50% of the patients with AF. The best scenario to approach AF concomitant to mitral valve surgery is when the maze concept, at least on the left atrium, is performed (Figure 4). Nowadays, excellent ablative devices such as bipolar radiofrequency and cryoenergy, rather than the old cutand- sew technique, permit to avoid suture lines in the posterior left atrium. This surgical technique presently adds around 20 minutes to the overall procedure maintaining similar efficacy in patients with or without mitral valve or other cardiac disease.
Figure 4
Thoracoscopic Surgical PVs Isolation for Paroxysmal Lone Atrial Fibrillation
The technical difficulty of achieving transmural electrical isolation hampers the results of transcatheter PVs isolation, especially long-term success, and in one third of the patients, more than one procedure is required to obtain stable sinus rhythm [48]. Minimally invasive surgical PVs isolation was introduced in 2005 as an alternative to transcatheter procedures [49]. The surgical approach has the advantage of being less technically demanding than a conventional full Maze open-chest operation, delivering a continuous lesion with bipolar radiofrequency in the setting of epicardial, offpump ablation. This technique has rapidly evolved to a complete thoracoscopic procedure. Short- and mid-term results of Surgical PVI have shown promising results, with around 70% freedom from AF and AAD after a single procedure [50]. Unfortunately, the current literature reports a large variety in patient selection, lesion sets and the long-term outcomes remain largely unknown. We showed that at long-term follow-up, freedom from recurrent AF without the use of AADs was 69% and freedom from recurrent AF with AAD was 84% in patients with paroxysmal AF undergoing surgical PVs isolation. No unforeseen late complications were detected [51]. This study showed slightly better results in a ‘lone’ paroxysmal AF population and demonstrated that the previously reported results are maintained at long-term follow-up. The use of bipolar radiofrequency ablation clamp (integrating an automatic transmurality algorithm with impedance feedback) and systematic verification of exit block offer certainty regarding effective isolation of the PVs, even in the thickest region surrounding PVs.
Final Thoughts
The contribution of arrhythmia surgery to the electrophysiological knowledge of cardiac arrhythmias is absolutely remarkable. All that we know today has been precisely documented more than 30 years ago, by the pioneeristic experimental studies of Dr. James Cox at Washington University, in Saint Louis. He and his coworkers provided, developed and eventually performed almost all the treatments for cardiac arrhythmias. The advent of catheters has taken great advantage from all these studies, trying to replicate the lesion sets proposed, most of the time simplifying them, according to the different pathologies. Nowadays, arrhythmia surgery for “standalone” procedures (thoracoscopic PVs isolation and Maze IV) is surely limited by invasivity in comparison to catheters. More, the lack of the ideal energy source, yielding complete transmurality when applied epicardially on the beating heart (heat sinking of the circulating blood), is limiting its widespread application and confining it to very selected cases of highly symptomatic lone AF. Arrhythmia surgery is much more adopted for the treatment of concomitant AF to valvular disease (mitral more than aortic).In the future, as we learn more about the mechanisms of AF and develop improved diagnostic technologies capable of precisely locating the areas responsible for arrhythmias, it will become easier to tailor specific lesion sets and adopt the right energy sources to individual patients. The advent of hybrid procedures and the Arrhytmia team in which surgeons and electrophysiologists work together for the perfect ablative lines is surely the key. The technological improvements of transcatheter ablative technologies and the mapping systems will likewise boost this complex and fascinating field.
References
- Jackman WM, Wang XZ, Friday KJ, Roman CA, Moulton KP, Beckman KJ, et al. Catheter ablation of accessory atrioventricular pathways (Wolff-Parkinson-White syndrome) by radiofrequency current. N Engl J Med. 1991; 324: 1605-1611.
- Canavan TE, Schuessler RB, Boineau JP, Corr PB, Cain ME, Cox JL. Computerized global electrophysiological mapping of the atrium in patients with Wolff-Parkinson-White syndrome. Ann ThoracSurg. 1988; 46: 223-231.
- Kosakai Y, Kawaguchi AT, Isobe F, Sasako Y, Nakano K, Eishi K, et al. Cox-maze procedure for chronic atrial fibrillation associated with mitral valve disease. J Thorac Cardiovasc Surg. 1994; 108: 1049-1055.
- Melo J, Adragao PR, Neves J, et al. Electrosurgical treatment of atrial fibrillation with a new intraoperative radiofrequency ablation catheter. Thorac Cardiovasc Surg. 1999; 47: 370-372
- Gaskell WH (1883) On the Innervation of the Heart, with especial reference to the Heart of the Tortoise. J Physiol. 4: 43-230.
- Kent AF. Researches on the Structure and Function of the Mammalian Heart. J Physiol. 1893; 14: 2-254.
- Durrer D, Roos JP. Epicardial excitation of the ventricles in a patient with Wolff-Parkinson-White syndrome (type B): Temporary ablation at surgery. Circulation. 1967; 35:15.
- Burchell HB, Frye RL, Anderson MW. Atrial-ventricular and ventricular-atrial excitation in Wolff-Parkinson-White syndrome (type B): Temporary ablation at surgery. Circulation. 1967; 36: 663.
- Cobb FR, Blumenschein SD, Sealy WC. Successful surgical interruption of the bundle of Kent in a patient with Wolff-Parkinson-White syndrome. Circulation. 1968; 38: 1018.
- Sealy WC, Gallagher JJ, Kasell JH. His bundle interruption for control of inappropriate ventricular responses to atrial arrhythmias. Ann Thorac Surg. 1981; 32:429
- Williams JM, Ungerleider RM, Lofland GK, Cox JL. Left atrial isolation: new technique for the treatment of supraventricular arrhythmias. J Thorac Cardiovasc Surg. 1980; 80: 373-380.
- Witkowski FX, Corr PB. An automated simultaneous transmural cardiac mapping system. Am J Physiol. 1984; 247: 661-668.
- Fontaine G, Guiraudon G, Frank R. Management of chronic ventricular tachycardia. In OS Narula(ed.): Innovations in DiagnosisandManagement of Cardiac Arrhythmias. Williams and Wilkins, Baltimore, 100C Heart Rhythm. 1979; 1: 5.
- Cox JL. Surgery for cardiac arrhythmias. In (ed.): Current Problems in Cardiology. Vol. VIII, No.4. Chicago, Year Book Medical Publishers, 1983.
- Cox JL, Daniel TM, Sabiston DC Jr. Desynchronized activation in myocardial infarction – a re-entry basis for ventricular arrhythmias. Circulation. 1969; 39: 63.
- El-Sherif N, Scherlag BJ, Lazzara R, Hope RR. Re-entrant ventricular arrhythmias in the late myocardial infarction period. 1. Conduction characteristics in the infarction zone. Circulation. 1977; 55: 686-702.
- Boineau JP, Cox JL. Rationale for a direct surgical approach to control ventricular arrhythmias: relation of specific intraoperative techniques to mechanism and location of arrhythmic circuit. Am J Cardiol. 1982; 49: 381-396.
- Kaiser GA, Waldo AL, Harris PD, Bowman FO Jr, Hoffman BF. New method to delineate myocardial damage at surgery. Circulation. 1969; 39: 183-189.
- Wittig JH, Boineau JP. Surgical treatment of ventricular arrhythmias using epicardial, transmural, and endocardial mapping. Ann Thorac Surg. 1975; 20: 117-126.
- Gallagher JJ, Oldham HN Jr, Wallace AG. Ventricular aneurysm with ventricular tachycardia. Report of a case with epicardial mapping and successful resection. Am J Cardiol. 1975; 35: 696.
- Guiraudon G, Fontaine G, Frank R, Escande G, Etievent P. Encircling endocardial ventriculotomy: a new surgical treatment for life-threatening ventricular tachycardias resistant to medical treatment following myocardial infarction. Ann Thorac Surg. 1978; 26: 438-444.
- Jatene AD. Surgical treatment of left ventricular aneurysm. In AE Baue, AS Geha, GL Hammond, H Laks, KS Naunheim (eds.): Glenn’s Thoracic and Cardiovascular Surgery. 5th edn. Norwalk, CT,Appleton & Lange Publishers. 1829.
- Dor V, Saab M, Coste P, Kornaszewska M, Montiglio F (1989) Left ventricular aneurysm: a new surgical approach. Thorac Cardiovasc Surg 37: 11-19.
- Della Bella P, Baratto F, Tsiachris D, Trevisi N, Vergara P, Bisceglia C, et al. Management of ventricular tachycardia in the setting of a dedicated unit for the treatment of complex ventricular arrhythmias: long-term outcome after ablation. Circulation. 2013; 127: 1359-68.
- Scheinman MM, Morady F, Hess DS. Catheter-induced ablation of the atrioventricular junction to control refractory supraventricular arrhythmias. JAMA. 1982; 248: 851
- Guiraudon GM, Campbell CS, Jones DL. Combined sinoatrial node atrio-ventricular node isolation: A surgical alternative to His’ bundle ablation in patients with atrial fibrillation. Circulation 1985; 72: 220.
- Cox JL, Canavan TE, Schuessler RB. The surgical treatment of atrial fibrillation: II. Intraoperative electrophysiologic mapping and description of the electrophysiologic basis of atrial flutter and atrial fibrillation. J Thorac Cardiovasc Surg. 1991; 101: 406-426.
- Cox JL, Boineau JP, Schuessler RB, Jaquiss RD, Lappas DG (1995) Modification of the maze procedure for atrial flutter and atrial fibrillation. I. Rationale and surgical results. J Thorac Cardiovasc Surg 110: 473-484.
- Cox JL, Jaquiss RD, Schuessler RB, Boineau JP. Modification of the Maze procedure for atrial flutter and atrial fibrillation. II. Surgical technique of the Maze III procedure. J Thorac Cardiovasc Surg. 1995; 110:485-495.
- Ad N, Cox JL. Stroke prevention as an indication for the Maze procedure in the treatment of atrial fibrillation. Semin Thorac Cardiovasc Surg. 2000; 12: 56-62.
- Gaynor SI, Prasad S, Cox JL. The long-term outcome of patients with coronary disease and atrial fibrillation undergoing the Cox-Maze procedure. Presented at the 83rd Annual Meeting of the American Association for Thoracic Surgery. 2003; Boston.
- McCarthy PM, Gillinov AM, Castle L, Chung M, Cosgrove D. The Cox-Maze procedure: the Cleveland Clinic experience. Semin Thorac Cardiovasc Surg. 2000; 12: 25-29.
- Schaff HV, Dearani JA, Daly RC, Orszulak TA, Danielson GK. Cox-Maze procedure for atrial fibrillation: Mayo Clinic experience. Semin Thorac Cardiovasc Surg. 2000; 12: 30-37.
- Arcidi JM, Doty DB, Millar RC. The Maze procedure: the LDS Hospital experience. Semin Thorac Cardiovasc Surg. 2000; 12: 38-43.
- Cox JL. The minimally invasive Maze-III procedure. Operative Techniques. Thorac Cardiovasc Surg. 2000; 5: 79-92.
- Lawrance CP, Henn MC, Miller JR, Sinn LA, Schuessler RB, Maniar HS, et al. A minimally invasive Cox maze IV procedure is as effective as sternotomy while decreasing major morbidity and hospital stay. J ThoracCardiovasc Surg. 2014; 148: 955-961.
- Gammie JS, Haddad M, Milford-Beland S, Welke KF, Ferguson TB. Atrial fibrillation correction surgery: lessons from the Society of Thoracic Surgeons National Cardiac Database. Ann Thorac Surg. 2008; 85: 909-914.
- Benussi S, Galanti A, Zerbi V, Privitera YA, Iafelice I, et al. Electrophysiologic efficacy of irrigated bipolar radiofrequency in the clinical setting. J Thorac Cardiovasc Surg. 2010; 139: 1131-1136.
- Pozzoli A, Benussi S, Anzil F, Taramasso M, Privitera YA. Electrophysiological efficacy of Epicor high-intensity focused ultrasound. Eur J Cardiothorac Surg. 2012; 42: 129-134.
- Pozzoli A, Taramasso M, Colombo DF, Ancona F, Cianflone D, Della Bella P, et al. Electrophysiological efficacy of temperature-controlled bipolar radiofrequency. Eur J Cardiothorac Surg. 2015; 47: 188-192.
- Melby SJ, Lee AM, Zierer A, Kaiser SP, Livhits MJ. Atrial fibrillation propagates through gaps in ablation lines: implications for ablative treatment of atrial fibrillation. Heart Rhythm. 2008; 5: 1296-1301.
- Lall SC, Melby SJ, Voeller RK, Zierer A, Bailey MS. The effect of ablation technology on surgical outcomes after the Cox-maze procedure: a propensity analysis. J Thorac Cardiovasc Surg. 2007; 133: 389-396.
- Pozzoli A, Taramasso M, Coppola G, Kamami M, La Canna G, Della Bella P. Maze surgery normalizes left ventricular function in patients with persistent lone atrial fibrillation. Eur J Cardiothorac Surg. 2014; 46: 871-876.
- Henn MC, Lancaster TS, Miller JR, Sinn LA, Schuessler RB. Late outcomes after the Cox maze IV procedure for atrial fibrillation. J Thorac Cardiovasc Surg. 2015; 150: 1168-1176.
- Sueda T, Nagata H, Shikata H, Orihashi K, Morita S. Simple left atrial procedure for chronic atrial fibrillation associated with mitral valve disease. Ann Thorac Surg. 1996; 62: 1796-1800.
- Benussi S, Pappone C, Nascimbene S, Oreto G, Caldarola A, Stefano PL, et al. A simple way to treat chronic atrial fibrillation during mitral valve surgery: the epicardial radiofrequency approach. Eur J Cardiothorac Surg. 2000; 17: 524-529.
- Kress DC, Sra J, Krum D, Goel A, Campbell J. Radiofrequency ablation of atrial fibrillation during mitral valve surgery. Semin Thorac Cardiovasc Surg. 2002; 14: 210-218.
- Weerasooriya R, Khairy P, Litalien J, Macle L, Hocini M, et al. Catheter ablation for atrial fibrillation: are results maintained at 5 years of follow-up? J Am Coll Cardiol. 2011; 57: 160-166.
- Wolf RK, Schneeberger EW, Osterday R, Miller D, Merrill W. Video-assisted bilateral pulmonary vein isolation and left atrial appendage exclusion for atrial fibrillation. J Thorac Cardiovasc Surg. 2005; 130: 797-802.
- De Maat GE, Pozzoli A, Scholten MF, Hillege HL, Van Gelder IC, Alfieri OR, et al. Surgical minimally invasive pulmonary vein isolation for lone atrial fibrillation: midterm results of a multicenter study. Innovations (Phila). 2013; 8: 410-415.
- De Maat GE, Pozzoli A, Scholten MF, Van Gelder IC, Blaauw Y. Long-term results of surgical minimally invasive pulmonary vein isolation for paroxysmal lone atrial fibrillation. Europace. 2015; 17: 747-752.