Research Article
Comparison between Negative Pressure Wound Therapy and Conventional Wound Care after Extensive Surgical Debridement in Patients with Fournier’s Gangrene
Mario E Trejo-Ávila*, Andrés Rodríguez-Parra, Aarón Díaz-Flores, Elisafat Arce-Liévano, Miguel Blas-Franco, Sujey Romero-Loera and Mucio Moreno-Portillo
Department of General Surgery, General Hospital “Dr. Manuel Gea González”, Mexico
*Corresponding author: Mario E. Trejo Avila, Department of General Surgery, Hospital General “Dr. Manuel Gea González”. Calzada de Tlalpan 4800, Colonia Sección XVI Tlalpan, ZIP 14080, Mexico
Published: 06 Mar, 2017
Cite this article as: Trejo-Ávila ME, Rodríguez-Parra
A, Díaz-Flores A, Arce-Liévano E,
Blas-Franco M, Romero-Loera S, et
al. Comparison between Negative
Pressure Wound Therapy and
Conventional Wound Care after
Extensive Surgical Debridement in
Patients with Fournier’s Gangrene. Clin
Surg. 2017; 2: 1335.
Abstract
Introduction: Negative pressure appears an effective tool for postoperative wound care of patients
with Fournier´s gangrene. The aim of this study is to compare the surgical outcomes of patients
with Fournier´s gangrene treated with negative pressure wound therapy (NPWT) or conventional
dressings after aggressive surgical debridement in our institution.
Materials and Methods: We retrospectively reviewed records of patients with the diagnosis of
Fournier´s gangrene treated with extensive surgical debridement (between 2013 and 2015). We
compare surgical outcomes between negative pressure wound therapy and conventional treatment.
Clinical features of 25 patients were described. We included in the analysis 11 patients with NPWT
and 11 patients with conventional treatment.
Results: Patients in the conventional wound care group had shorter length of stay (9 days vs. 20.7
days, p=0.005). NPWT patients had faster healing rates (24.5 vs. 41.8 days, p=0.024), they needed
less opioids to control pain (5 vs. 10 patients, p=0.022) and they required more fecal diversion
procedures (5 vs. 1 patient, p=0.055). One death in the NPWT group (1 vs. 0, p=0.306).
Conclusions: Patients with Fournier´s gangrene benefit with the use of negative pressure wound
therapy compared with conventional wound care.
Keywords: Conventional Dressings, Fournier´s Gangrene, Negative Pressure Wound Therapy, Vacuum Assisted closure
Introduction
Since Jean Alfred Fournier first described in 1883 young men´s disease affecting penis and scrotal region, several reports and case series have been published [1]. Today, Fournier´s gangrene (FG) is considered a type of necrotizing fasciitis of the perineal, genital and perianal region that has a rapidly progressive and potentially fatal course. This disease has also been recognized in women although is ten times less frequently [2]. Early diagnosis is important and the principles of management are: hemodynamic stability, broad-spectrum antibiotics and prompt aggressive surgical debridement [3]. Several different protocols and surgical techniques have been proposed for postoperative wound treatment: hyperbaric oxygenation, growth factors, skin grafts, different types of sterile dressings and recently the use of negative-pressure wound therapy [4]. Negative pressure dressing was first described by Felischmann et al. [4] in 1993. Morykwas and Argenta in 1997 studied the use of suction applied to polyurethane foam in wound. Several NPWT mechanisms of action have been proposed: fluid removal and environmental control of the wound, granulation tissue formation, cell proliferation, modulation of inflammation and change in bacterial levels [5- 6]. NPWT appears an effective tool for postoperative wound care of patients with FG, but large series and randomized trials are lacking. The aim of this study is to compare the surgical outcomes of patients with Fournier´s gangrene treated with negative pressure wound therapy (NPWT) or conventional wound therapy after aggressive surgical debridement in our institution.
Materials and Methods
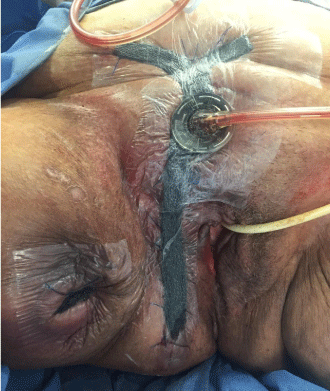
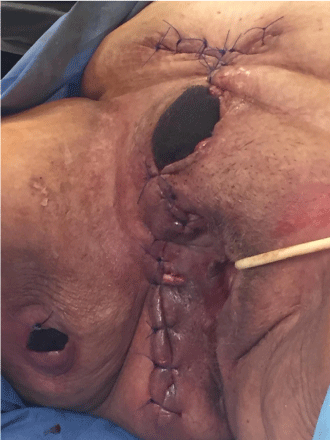
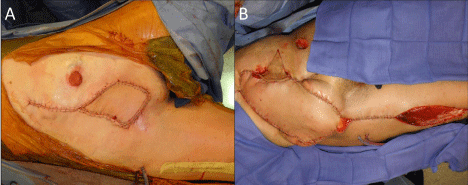
We retrospectively reviewed records of patients with the diagnosis of Fournier´s gangrene treated with extensive surgical debridement with negative pressure wound therapy or conventional wound care. All patients were treated in our Institution from January, 1 2013 to December, 31 2015. All data obtained from our Institutional Approved Database. We analyzed preoperative data: age, sex, ASA (American Society of Anesthesiologists) class, diagnosis of septic shock, white cell blood count, sepsis etiology, and localization of the primary abscess. Then we analyzed surgical variables: number of surgical debridement required, microbiology, need of intensive care unit, and in patients with anorectal etiology we studied the presence of anorectal fistulae. For data analysis, the patients were divided into two groups: the first group included those patients treated with negative pressure wound therapy (NPWT) V.A.C. (V.A.C.®, vacuum assisted closure, KCI, San Antonio, TX) and the second group included those patients treated with conventional wound care. All patients received the same initial therapy: intravenous fluid reanimation, electrolyte imbalance correction, broad-spectrum antibiotics and aggressive surgical debridement with microbial cultures. We performed the following technique in patients with NPWT [7]. The first step, after surgical necrotic tissue debridement, consisted in measure the surgical area and trims the foam (V.A.C.® GranuFoam™ Dressings) according with this area. Prior the application of the foam we have to be sure that all the necrotic tissue is debrided and no active bleeding is present. It is necessary to avoid contact of the foam with healthy skin and anus. After the foam is applied, we covered with the V.A.C.® Advanced Drape. After choose pad application side we applied the pad (V.A.C® SensaT.R.A.C™pad). Suction pressure of 125 mmHg was applied to the wounds continuously, with increased pressure up to 175 mmHg (Figure 1). Dressings were changed on demand. With every dressings change, we performed new debridement and closed the wound with stitches partially from the edges (Figure 2). After the wounds were clinically healed, no signs of infection present and enough granulation tissue existed, tertiary wound closure was performed in all patients (Figure 3). The second group included patients with conventional wound care. This conventional care consisted in sterile gauze or dressings changed once or twice daily. We performed surgical debridement on demand in the operating room. Secondary or tertiary wound closures were the goal depending on each patient. For the comparison of both groups we selected 11 patients of the conventional wound care group that had similar characteristics (age and sex) with the NPWT group. The variables that we compared were: hospital length of stay (LOS), time from initial surgical debridement to complete healing, need to control pain with opioids, need for fecal diversion and 60-day mortality. We reported the data as means (standard deviation), medians (minimum and maximum) and percentages, depending on the type of variable. The categorical variables were compared using Fisher´s exact test. The continuous variables were compared with a Student´s t-test (if normally distributed) or with a Mann-Whitney U test (if not normally distributed). All statistical tests were two-tailed and a value of p<0.05 was considered significant. The data were analyzed using SPSS v18.0 for Windows (SPSS, Inc., Chicago, IL.).
Figure 1
Figure 2
Figure 3
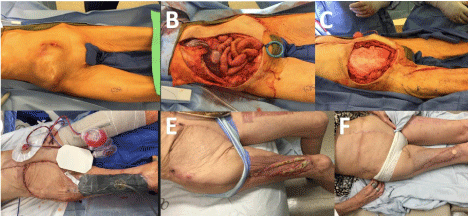
Figure 4
Results
We found a total of 25 patients (Table 1). The mean age was 51.4 years (SD=13.8), male predominance with 15 patients. The most frequent comorbidity was diabetes, present in 44% of the patients, 20% had hypertension and one patient with lupus. Twelve patients were classified as ASA I-II class and thirteen patients as ASA III-IV. Three patients had septic shock on presentation, and the rest of the patients had signs of sepsis. The mean value of white blood cells was 13.7 (SD=7.4). In 21 patients a CT scan was performed preoperatively. On regard of the etiology we found that 19 patients (76%) were anorectal and in 6 patients (24%) the origin of the infection was urogenital. Of the patients with anorectal etiology, they had abscess in the following locations: perirectal (n=8/19), horse-shoe (n=6/19), and ischiorectal (n=5/19). In this group of patients we found anorectal fistula (in 9 of 19 patients) during the first surgery, they were treated with fistulotomy alone (n=7/9) and fistulotomy + seton (n=2/7). About the number of surgical debridements, we found a median of 2, with a minimum of 1 and a maximum of 6 debridements. In 14 patients extensive debridement of the genital area was performed and in 10 patients the process extended to the anterior abdominal wall and required debridement of that area too. In 11 patients, NPWT with V.A.C. ® was used. In two of these patients we applied the V.A.C. Instill ® Therapy System with normal saline infusion. The median of dressings change was 2 (range=1-5). The most commonly isolated agent was E.coli (n=19; 76% of all patients). In two patients we found rectal carcinoma in tissue biopsies. Three of the patients required admission to the intensive care unit. And one patient (55 year old male) died of respiratory complications associated with mechanical ventilation in the intensive care unit. The results of the comparative analysis are presented in (Table 2). We found no statistical significant differences in age and sex. Patients in the conventional wound care group had shorter LOS (9 days vs. 20.7 days, p=0.005). Patients with NPWT had faster healing rates, from initial surgical debridement to complete healing (24.5 [SD=7.6] vs. 41.8 [SD=21.2] days, p=0.024), and the difference was significant. The use of opioids to control pain was less in patients with NPWT (5 [45.5%] vs. 10 [90.9%] patients, p=0.022).We found 6 patients that needed fecal diversion. Of these patients, 4 were diverted with temporary colostomy and 2 with a temporary fecal containment device (Figure 4). No statistical significant difference exist on regard of the fecal diversion in both groups (p=0.055), although more patients in the NPWT group needed diversion (5 vs. 1 patient). No significant difference in the 60-day mortality (1 vs. 0, p=0.306), the patient dead belongs to the NPWT group.
Discussion
Fournier´s gangrene (FG) is a rare, fatal and progressive necrotizing fasciitis affecting external genitalia, perineal or perianal regions, which may extend up to the anterior abdominal wall and down to thelower extremities, between the fascial planes [8]. The most common foci include the gastrointestinal tract (30-50%) and genitourinary tract (20-40%). It is caused by synergistic aerobic and anaerobic microorganisms. The most commonly isolated are Escherichia coli, Klebsiella pneumonia, Staphylococcus aureus and Bacteroidesfragilis [2-8]. Comorbid disorders are frequent, the commonest being diabetes mellitus [8]. Some authors estimate the prevalence of diabetes among FG patients between 50-70% [9]. In our study we found diabetes to be present in 44% of our population. It is considered a rare condition, with a reported incidence of 1.6/100,000 males with peak incidence in the 5th and 6th decades [2,8]. The mortality rate is high, at 20-50% in most series [2]. The mortality from FG drastically decreases once patients survived initial sepsis in the acute phase [10]. Our study showed male predominance and a mean age of 51.4 years, both in concordance with global series. Something interesting is the number of females (n=10/25) found in our population. The cornerstones of treatment of FG are broadspectrum antibiotics and urgent radical surgical debridement [2,8,10]. Multiple surgical debridements are sometimes necessary, with a mean of 3.5 debridements required per patient [11]. The objective of surgery is the removal of all necrotic tissue thus eliminating the systemic effects of necrotic material, toxins and bacteria [10,12]. This aggressive treatment produces extensive tissue defects. So the dilemma of the surgeons is how to deal with these defects. To facilitate the detection of changes in the wound site, proper dressing with simultaneous exploration is the key to wound therapy success in FG [12]. There are three approaches: the conventional dressings, the negative pressure wound treatment, and plastic reconstruction. Each of the above mentioned approaches have pros and cons [2,8,11]. The conventional method of dressing is known as wet-to-dry dressing. This method is beneficial and widely used globally because it is easy to apply and cost-effective [12]. The disadvantage of this method is the frequency of dressing changes. Some argue that conventional dressings require painful daily and additional changes due to blood and fluids from the wound, with quality-of-life impairment [4]. In our study we found that opioid drugs were less required in the NPWT group, we believe this is due to increased pain during daily dressings change in the conventional group. Vacuum-assisted closure device is a wound care system that promotes healing providing continuous sub-atmospheric pressure by a pump connected to the foam sponge placed in the wound [5,6]. Several NPWT mechanisms of action have been proposed and are divided in primary and secondary effects. Primary effects are: macro deformation, micro deformation, fluid removal and environmental control of the wound. And the secondary effects are: granulation tissue formation, cell proliferation, modulation of inflammation, change in neuropeptides and change in bacterial levels [5,6,13]. The optimal therapeutic choice remains uncertain; the vast majority of FG treatment studies are case report and case series, only few retrospective comparative studies and no randomized control trials. Cyzmek et al. [10] reported in 2009 a comparative retrospective study between conventional dressings (n=16) vs. vacuum-assisted closure (n=19). The mean age was 58.2 years and 57.2 years respectively. Length of stay was 27.8 (±27.6) for conventional dressing group and 96.8 (±77.2) days for V.A.C. group (p< 0.001). Enterostomies were performed in 43.8% of conventional and in 89.5%of V.A.C. patients. The patients in conventional group underwent an average of 4.9 surgical procedures and in VAC group had an average of 10.1 surgeries. The mortality rate was 20% among all patients. This study differs from our results on regard of LOS, number of ostomies needed, and the average of procedures performed. In 2012, Ozkan et al. [13] published a retrospective observational study. They enrolled 12 patients, in eight patients they used conventional dressings and in four they used NPWT. For fecal diversion, colostomy was performed in six patients and Flexi-Seal (Convatec, Greensboro, NC) in two cases. The mean LOS in patients with NPWT was 18 days, while it was 20 days in the others. Ozturk et al. [14] have reported that V.A.C. treatment and conventional methods yielded equivalent results in terms of wound healing and costs. And reported that the use of V.A.C. led to fewer dressing changes, less pain, fewer skipped meals, and greater mobility while reducing hands-on treatment time for the surgeon. We agree with these conclusions. Colostomy is the treatment of choice to protect wound affected by FG. Temporary fecal containment device is an alternative to colostomy with economic benefit and patient comfort for short-term faecal diversion [13]. We consider fecal diversion when the wound couldn’t be maintained clean and especially when the NPWT V.A.C. system failed because fecal contamination of the sponge or the V.A.C. system couldn’t be applied closed to the anus. Although we believe NPWT is a better choice for wound care in patients with FG, there are important limitations in our study. Data collection was retrospective, the cohort is small, we did n’t evaluate Fournier´s Gangrene Severity Index and we focused only in the postoperative results.
Table 1
Table 2
Conclusion
Patients with Fournier´s gangrene benefit with the use of negative pressure wound therapy compared with traditional wound care. In our study, patients treated with negative pressure had faster healing rates and less use of opioid medication, although they have longer length of stay. In our experience treatment of Fournier´s gangrene with negative pressure is easy and reliable.
References
- Fournier A. Gangrenefoudroyante de la verge ( overwhelming gangrene )Sem Med.1883. Dis colon Rectum.1988; 31: 984-988.
- Chennamsetty A, Khourdaji I, Burks F, Killinger KA. Contemporary diagnosis and management of Fournier´s gangrene. Ther Adv Urol. 2015; 7: 203-215.
- Ye J, Xie T, Wu M, Ni P, Lu S. Negative Pressure Wound Therapy Applied Before and After Split-Thickness Skin Graft Helps Healing of Fournier Gangrene. A Case Report. Medicine. 2015; 94: e 426.
- Tucci G, Amabile D, Cadeddu F, Milito G. Fournier´s gangrene wound therapy: our experience using VAC device. Langenbecks Arch Surg. 2009; 394: 759-760.
- Huang C, Leavitt T, Bayer LR, Orgill DP. Effect of negative pressure wound therapy on wound healing. Curr Probl Surg. 2014; 51: 301-331.
- Orgill DP, Bayer LR. Negative pressure wound therapy: past, present and future. Int Wound J. 2013; 10: 15-19.
- V.A.C.®. Therapy Forms and Brochures.
- Mallikarjuna MN, Vijayakumar A, Patil VS, Schivswamy BS. Fournier´s Gangrene: Current Practices. ISRN Surgery. 2012; 1-8.
- Benjelloun EB, Souiki T, Yakla n, Ousadden A, Mazaz K, Louchi A, et al. Fournier´s gangrene: our experience with 50 patients and analysis of factors affecting mortality. World J of Emerg Surg. 2013; 8: 13.
- Czymek R, Schmidt A, Eckmann C, Bouchard R, Wulff B, Laubert T, et al. Fournier´s gangrene: vacuum-assisted closure versus conventional dressings. The Am J Surg. 2009; 107:168-176.
- Chawla SN, Gallop C, Mydlo JH. Fournier’s gangrene: an analysis of repeated surgical debridement. Eur Urol. 2003; 43: 572-575.
- Oguz A, Gumus M, Turkoglu A, Bozdag Z, Ulger BV, Agacayak E, et al. Fournier´s Gangrene: A Summary of 10 Years of Clinical Experience. Int Surg. 2015; 100: 934-941.
- Ozkan OF, Koksal N, Altinli E, Celik A, Uzun MA, Ckman O, et al. Fournier´s gangrene current approaches. Int Wound J. 2016; 13: 713-716.
- Ozturk E, Ozguc H, Yilmazlar T. The use of vacuum assisted closure therapy in the management of Fournier’s gangrene. Am J Surg. 2009; 197: 660-665.