Case Report
Primary Bladder MALT Lymphoma in a Patient with Rheumatoid Arthritis
Madeline Cancian1*, Timothy Y. Tran1, Ali Amin2 and Dragan Golijanin1
1Division of Urology, Warren Alpert Medical School of Brown University, USA
2Division of Pathology, Warren Alpert Medical School of Brown University, USA
*Corresponding author: Madeline Cancian, Division of Urology, Warren Alpert Medical School of Brown University, 2 Dudley Street, Suite 174 Providence, RI 02903, USA
Published: 03 Mar, 2017
Cite this article as: Cancian M, Tran TY, Amin A, Golijanin
D. Primary Bladder MALT Lymphoma in
a Patient with Rheumatoid Arthritis. Clin
Surg. 2017; 2: 1317.
Abstract
MALT lymphoma of the urinary bladder is an uncommon entity. It typically arises from an inflammatory milieu, within which chronic antigenic stimulation leads to the formation of ectopic germinal centers. Within these immune sites, B-cell clones rapidly proliferate and neoplasia results. In this report, we present a case of bladder MALT lymphoma in a patient with rheumatoid arthritis. We then review the literature and suggest an inflammatory pathway that may link this autoimmune disease with lymphomagenesis.
Introduction
Primary lymphoma of the urinary bladder is a rare clinical finding that accounts for only 0.2% of
extranodal lymphomas [1]. Approximately 100 cases of bladder lymphoma have been described [2].
Mucosa-associated lymphoid tissue (MALT) lymphoma is the most common pathologic subtype
[3].
Extranodal MALT lymphoma is oftentimes associated with chronic infection; however, it has
also been described to arise in association with autoimmune diseases [4]. Rheumatoid arthritis is a
systemic autoimmune disease associated with a base line two-fold higher risk of lymphoproliferative
malignancies which increases with autoimmune disease severity [5].
Here, we present the case of a patient with rheumatoid arthritis [RA] who developed primary
bladder MALT lymphoma. To the best of our knowledge, the association between bladder MALT
lymphoma and RA has not been described in the literature.
Case Presentation
A 71 year-old white woman with a history of rheumatoid arthritis presented to her primary
care physician with a complaint of persistent suprapubic discomfort. She had no hematuria or
other lower urinary tract symptoms. A series of urine cultures were obtained that were negative.
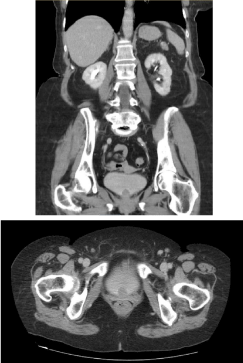
A pelvic ultrasound showed an intravesical mass, which prompted a contrast-enhanced computed
tomography (CT) scan of her abdomen and pelvis. This showed a two-centimeter filling defect
arising from the bladder floor (Figure 1). Cystoscopy confirmed a smooth nodular lesion at the
trigone that was resected in the operating room.
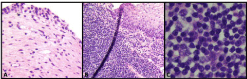
Histologic examination of this lesion revealed a dense lymphoid aggregate showing focal
residual germinal centers. This aggregate infiltrated beyond the urothelium into the lamina propria
and submucosa. The lymphoid aggregates were composed of small to medium sized lymphocytes
showing nuclear membrane irregularity, chromatin dispersion, inconspicuous nucleoli and
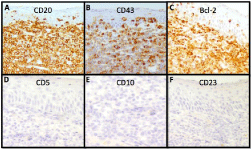
relatively abundant pale cytoplasm (Figure 2). Lymphoid cells were positive for CD20, CD43 and
BCL2and were negative for CD5, CD23 and CD10 (Figure 3). The proliferation index assessed by
KI67 was approximately 10%. Taken together, these findings were consistent with MALT lymphoma.
A positron emission tomography CT scan demonstrated no evidence of metastatic disease. The
diagnosis of primary MALT lymphoma of the bladder was made. Six weeks of oral ciprofloxacin were
given as per the recommendation of her oncologist. No systemic chemotherapy was administered.
A repeat transurethral resection was performed two months later and revealed no evidence of
malignancy. CT scan of the abdomen and pelvis was performed eight months after diagnosis and
showed no evidence of disease. Interval cystoscopies have been performed for one year and show
no evidence of recurrence.
Figure 1
Figure 2
Figure 2
Hemotoxylin and eosin stain of resected MALT lymphoma
specimen. A. Benign urothelium and lamina propria. B. Dense lymphoid
aggregation with infiltration to lamina propria. C. Small-to-medium-sized
lymphocytes with characteristic nuclear membrane irregularity, dispersed
chromatin and pale cytoplasm.
Figure 3
Figure 3
Immunohistochemistry consistent with MALT lymphoma. A.
CD20+ (B-cell marker). B. CD43+ (Often used as a pan T-cell marker,
while MALT lymphoma is usually positive). C. Bcl-2+ (over-expressed in
B-cell lymphomas). D. CD5- (positive in mantle-cell lymphomas, other
B-cell lymphomas and most T-cell malignancies; occasional CD5+ in MALT
lymphomas has been reported). E. CD10- (positive in follicular lymphoma).
F. CD23- (positive in chronic lymphocytic leukemia as well as other B and
T-cell lymphomas).
Discussion
MALT lymphoma of the bladder is a rare diagnosis. It occurs
most commonly in older patients, with a median age of 69 years.
Females are three-times more likely to be affected than males.
Hematuria is the most common symptom, reported by nearly twothirds
of patients, while one-third of patients will describe dysuria.
The pathway to developing bladder MALT lymphoma has yet to be
fully defined. However, increasing evidence suggests that extranodal
MALT lymphomagenesis is driven by chronic immune stimulation
[6]. Specifically, it has been proposed that continuous antigenic
stimulation leads to the recruitment of lymphoid tissue to ectopic
sites to form germinal centers, which are precursors for extranodal
lymphoma development. Within this extra-nodal lymphoid
aggregate, the immune stimulus promotes rapid proliferation of
antigen-specific B cell clones, resulting in errors in mitosis which
cause inactivation of tumor suppressor genes and chromosomal
rearrangements, ultimately leading to neoplasia.
Most commonly, the continuous immune reaction is caused by
chronic infection. This phenomenon has been observed at several
sites of MALT lymphoma. Over 90% of patients with gastric MALT
lymphoma are positive for Helicobacter pylori [7], 80% of ocular
adnexal lymphomas are associated with Chlamydia psittaci [8] and
spirochete infections are commonly implicated in MALT lymphoma
of the skin [9]. Indeed, amongst cases of MALT lymphoma of the
bladder, 20-33% are associated with chronic bacterial cystitis [1,10].
However, autoimmune disorders have also been linked to the
development of extra-nodal MALT lymphoma. An elevated risk of
lymphoma has been noted in patients with Hashimoto’s thyroiditis,
Sjogren’s syndrome, and relapsing polychondritis [11-13]. RA is
associated with a two-fold increased risk of lymphoproliferative
malignancy development and mortality, with the greatest risk
observed in patients with more severe disease [5]. The mechanism of
development of lymphoma in RA is not well understood. Constant
stimulation of the immune system leading to clonal selection
and predisposition of CD5+ B cells to malignant transformation,
decreased T-suppressor lymphocytes, and decreased natural killer
cell activity are some proposed mechanisms [14]. There is also
uncertainty as to whether disease modifying antirheumatic drugs
(DMARDs) contribute to the development of lymphoma, however,
studies have failed to find a correlation [5,15-16]. Similar to those
with chronic infection, it is likely that a persistent inflammatory
milieu at the site of lymphomagenesis perpetuates the development
of malignancy.
Our patient was being treated for a significant case of rheumatoid
arthritis. Amongst her medications was leflunomide, a pyrimidine
synthesis inhibitor reserved for moderate-to-severe rheumatoid
disease. Thus, a potential pathway through which this MALT
lymphoma developed is through chronic inflammation of the bladder
driven by her underlying severe autoimmune disease.
This is the first reported case of bladder MALT lymphoma in
a patient with rheumatoid arthritis. Evidence is accumulating for
the association between extranodal MALT lymphoma and chronic
inflammation, either from an infectious or autoimmune source. While
the clinical significance of this finding is still to be determined, it does
suggest that in patients with autoimmune disorders, especially those
with higher disease activity, hematuria and otherwise unexplained
lower urinary tract symptoms should prompt careful consideration
for malignancy evaluation. Although MALT lymphoma has been
described as a clinically indolent disease, its early detection and
treatment are paramount, as 10% of MALT lymphomas are reported to convert to high-grade lymphomas, which have a far more aggressive
course [17].
Conclusion
Bladder MALT lymphoma is a rare finding, however, it is more likely in female patients with chronic infections or autoimmune diseases. In these patients, a full work up should be completed in the setting of hematuria or unexplained lower urinary tract symptoms. MALT lymphoma is very treatable if caught early however a subset can convert to high-grade lymphomas with an aggressive clinical course.
References
- Bates AW, Norton AJ, Baithun SI. Malignant lymphoma of the urinary bladder: a clinicopathological study of 11 cases. J Clin Pathol. 2000; 53: 458-461.
- Vempati P, Knool MA, Alqatari M, Strauchen J, Malone AK, Bakst RL. MALT Lymphoma of the Bladder: A Case Report and Review of the Literature. Case Rep Hematol. 2015; 2015: 934374.
- Pawade J, Banerjee SS, Harris M, Isaacson P, Wright D. Lymphomas of mucosa-associated lymphoid tissue arising in the urinary bladder. Histopathology. 1993; 23: 147-151.
- Derringer GA, Thompson LD, Frommelt RA, Bijwaard KE, Heffess CS, Abbondanzo SL. Malignant lymphoma of the thyroid gland: a clinicopathologic study of 108 cases. Am J Surg Pathol. 2000; 24: 623-639.
- Baecklund E, Iliadou A, Askling J, Ekbom A, Backlin C, Granath F, et al. Association of chronic inflammation, not its treatment, with increased lymphoma risk in rheumatoid arthritis. Arthritis Rheum. 2006; 54: 692-701.
- Routsias JG, Goules JD, Charalampakis G, Tzima S, Papageorgiou A, Voulgarelis M. Malignant lymphoma in primary Sjogren's syndrome: An update on the pathogenesis and treatment. Semin Arthritis Rheum. 2013; 43:178-186.
- Witkowska M, Smolewski P. Helicobacter pylori infection, chronic inflammation, and genomic transformations in gastric MALT lymphoma. Mediators Inflamm. 2013; 2013: 523170.
- Ponzoni M, Ferreri AJ, Guidoboni M, Lettini AA, Cagni MG, Pasini E, et al. Chlamydia infection and lymphomas: association beyond ocular adnexal lymphomas highlighted by multiple detection methods. Clin Cancer Res. 2008; 14: 5794-5800.
- Schollkopf C, Melbye M, Munksgaard L, Smedby KE, Rostgaard K, Glimelius G, et al. Borrelia infection and risk of non-Hodgkin lymphoma. Blood. 2008; 111: 5524-5529.
- Hughes M, Morrison A, Jackson R. Primary bladder lymphoma: management and outcome of 12 patients with a review of the literature. Leuk Lymphoma. 2005; 46: 873-877.
- Jonsson MV, Theander E, Jonsson R. Predictors for the development of non-Hodgkin lymphoma in primary Sjogren's syndrome. Presse Med. 2012; 41(9 Pt 2): e511-516.
- Lee SC, Hong SW, Lee YS, Jeong JJ, Nam KH, Chung WY, et al. Primary thyroid mucosa-associated lymphoid tissue lymphoma; a clinicopathological study of seven cases. J Korean Surg Soc. 2011. 81: 374-379.
- Lichauco JJ, Lauer S, Shigemitsu HH, Bello JA, Bhattacharyya PK, Barland P, et al, Orbital mucosa-associated lymphoid tissue (MALT)-type lymphoma in a patient with relapsing polychondritis. Arthritis Rheum. 2001; 44: 1713-1715.
- Georgescu L, Quinn GC, Schwartzman S, Pagent SA. Lymphoma in patients with rheumatoid arthritis: association with diesease state or methotrexate treatment. Semin Arthritis Rheum. 1997; 26: 794-804.
- Askling J, Baecklund E, Granath F, Geborek P, Fored M, Backlin C, et al. Anti-tumor necrosis factor therapy in rheumatoid arthiritis and risk of malignant lymphomas: relative risk and time trends in Swedish Biologics Register." Ann Rheum Dis. 2009; 68: 648-653.
- Simon TA, Thompson A, Gandhi KK, Hochberg MC, Suissa S. Incidence of malignancy in adult patients with rheumatoid arthritis: a meta-analysis. Arthritis Res Ther. 2015; 17: 212.
- Rossi D, Gaidano G. Molecular heterogeneity of diffuse large B-cell lymphoma: implications for disease management and prognosis. Hematology. 2002; 7: 239-252.