Case Report
Breast Cancer in a Male with BRCA2 Mutation: Is Screening Mammography Necessary in Men?
Jessica L. Buicko*,Michael A. Lopez and Beth Ann Lesnikoski
Department of General Surgery, University of Miami- Regional Medical Campus, USA
*Corresponding author: Jessica L. Buicko, Department of General Surgery, University of Miami- Regional Medical Campus, Atlantis, FL 33462 USA
Published: 10 Feb, 2017
Cite this article as: Buicko JL, Lopez MA, Lesnikoski BA.
Breast Cancer in a Male with BRCA2
Mutation: Is Screening Mammography
Necessary in Men?. Clin Surg. 2017;
2: 1302.
Abstract
We present the case of a 76 year-old male with BRCA2 and a personal history of left-sided breast cancer who presented 20 years later with right-sided breast cancer found on screening mammography. The role of screening mammography and its relevance to males with breast cancer is poorly understood. There are currently no screening guidelines for male breast cancer. Further research is needed to determine the benefit of screening males at higher risk of developing breast cancer.
Case Presentation
A 76 year-old male presented to our surgical clinic for evaluation. He has a known BRCA mutation and a personal history of left breast cancer, treated with a left sided modified radical mastectomy 20 years prior. He was undergoing intermittent mammography for surveillance. He denied ever feeling a palpable mass or experiencing any nipple discharge. Recent screening mammographyafter 4 year interval, showed a nodule in the right retroareolar region, which was 1.2 x 1.3cm in size. He underwent diagnostic workup with spot compression mammography views and ultrasound. Ultrasound showed an irregularly marginated 8 x 5 x 7mm hypoechoic lesion, with a surrounding echogenic region measuring 1.1 x 0.7cm. Ultrasound guided biopsy revealed intermediate grade invasive ductal carcinoma. He subsequently underwent a right total mastectomy with sentinel lymph node biopsy. Final pathology confirmed an estrogen receptor positive, progesterone receptor positive, Her-2-neu non-amplified, 2.0cm invasive ductal carcinoma. Sentinel lymph node was negative for carcinoma.
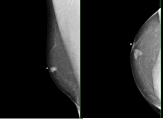
Figure 1 and 2
Figure 1 and 2
Mammogram (CC & MLO view) showing a mass with irregular borders within the right breast retro areolar region measuring 1.3 x 1.3 cm. There is a central hypoechoic region of the mass approximately 0.8 x 0.5 x 0.8 cm
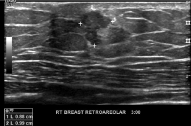
Figure 3
Figure 3
Right Breast Mass seen on ultrasound measuring 1.4 cm x 0.9 cm x 0.9 cm which was subsequently biopsied.
Discussion
Male breast cancer accounts for less that 1% of cancers diagnosed in the United States. The
female-to-male ratio for breast cancer is 100:1 [5]. Approximately 90% of male breast cancers are
invasive ductal carcinomas. The male breast lacks terminal lobules, unless exposed to high doses
of endogenous or exogenous estrogens. Lobular type accounts for less than 1.5% of male disease,
whereas it accounts for greater than 10% in women [6].
It has been found that most male breast cancers express high levels of hormone receptors. The
estrogen and progesterone receptors are more likely to be positive in male patients than in female
patients. The proportion of hormone-receptor-expressing tumors increases with age, comparable to
post-menopausal women. The proto-oncogene HER2/neu is expressed in only 5% of male patients,
and in approximately 15% of female patients [6].
There have been many documented risk factors related to male breast cancer. Some of the more
common include Klinefelter syndrome, BRCA1 and BRCA2, a family history in a first degree male
or female relative, hyperestrogenism, advanced age, and a history of chest radiation [5]. In contrast
to women with breast cancer, white race is not a risk factor in males [7].
Germ line mutations in the high penetrance BRCA2, and with a lower frequency BRCA1, have
been shown to predispose men to primary breast cancer. Men with a high-risk family history have
shown a 60-76% incidence of BRCA2 mutation and a 10-16% BRCA1 mutation [6]. The median
age of male breast cancer in BRCA2 positive men is 58.8, and is 69.7 years in those without the
mutation. The frequency of mutation of these genes is highly variable. In both men and women,
and prevalence has been shown to depend on mainly on ethnicity and country. In Iceland, BRCA2
mutations carries the highest likelihood of both male breast cancer
and prostate cancer, and warrants genetic testing for male family
members [8,9]. Male patients with a BRCA2 mutation have an 8%
estimated lifetime risk of developing breast cancer [10]. This is
comparable to subgroups of women in the US who are routinely
encouraged to undergo mammography screening, such as African
American, Latina and Asian women. In total, 4-14% of male patients
with breast cancer not selected on basis of family history will have
a BRCA2 mutation [10]. In regards to other genetic risk factors,
there is mixed data in regards to the association of PALB2, CYP17,
and CHEK2 in their risk of male breast cancer [7]. In female breast
cancer, it has been established that there is an association of leisuretime
physical activity (LTPA) with risk reductions of about 25%
among post-menopausal normal weight women compared with no
association for obese women [11]. There is some evidence in small
studies that exercise reduces the risk of breast cancer in males but
this has not been corroborated by larger studies [12]. Arem et al. [11]
evaluated the association of physical activity and risk of male breast
cancer and found that compared to low-LTPA, medium, and high
LTPA were not associated with male breast cancer risk (OR 1.01;
95% CI, 0.79-1.29; 0.90, 0.69-1.18, respectively). There is no current
agreement regarding what subgroup of men would benefit from
screening mammography. Male breast cancers are often detected at
a late stage, commonly when a mass is palpable or nipple discharge is
present [1]. Multiple reasons exist for the delay in diagnosis. Factors
may include lack of screening in high-risk men, poor education on
male breast cancer, and decreased breast tissue in males, which may
lead to early lymphatic spread [1,13] Historically, compared to female
breast cancer patients, there is approximately a 22-month delay
between onset of symptoms and diagnosis, which has a dramatic
impact on treatment and survival [3]. Women, specifically those who
are BRCA 1 and BRCA 2 positive, are more likely to be diagnosed at
an earlier stage because of guidelines that dictates screening protocols
[1]. For women who are BRCA positive, the National Comprehensive
Cancer Network (NCCN) recommends yearly breast MRIs between
25 and 29 years old and then adding yearly mammograms starting
at age 30. In regards to males at high risk, the NCCN recommends
considering mammogram at age 40 [14]. Brenner et al proposed that
any male patient with a BRCA mutation should undergo screening
mammography [4]. Arguments against intensive screening included
false-positive results, unnecessary imaging, and unneeded surgery.
In a Dutch multicenter screening study for women with hereditary
breast cancer, the false positive rate of mammography and MRI was
5.5 vs. 14.0% (P< .001) [15]. No such data exists for men.
Conclusion
At this time there are no specific screening guidelines for male breast cancer. Although there is convincing evidence for an increased risk of cancer associated with certain gene mutations, specifically BRCA2, no recommendations have been made to detect disease in its early stages. This has unfortunately led to the late diagnosis of cancer in men that may have been managed before its progression to a terminal state. A thorough history can prompt genetic screening, which should encourage early screening of the patient, and both male and female relatives. Further data is needed to determine if men at increased risk, for example those with Klinefelter syndrome or history of chest radiation, would benefit from screening.
References
- Friedman L, Gayther S, Kurosaki T, D Gordon, B Noble, G Casey, et al. Mutation analysis of BRCA1 and BRCA2 in a male breast cancer population. Am J Hum Genet. 1997; 60: 313-319.
- Serarslan A, Gursel B, Okumus Meydan D, Sullu Y, Gonullu G. Male Breast Cancer: 20 Years Experience of a Tertiary Hospital from the Middle Black Sea Region of Turkey. Asian Pac J Cancer Prev. 2015; 16: 6673-6679.
- Heinig J, Jackish C, Rody A, Koch O, Buechter D, Schneider HP. Clinical management of breast cancer in males: a report of four cases. Eur J Obstet Gynecol Reprod Biol. 2002; 102: 67-73.
- Brenner R, Weitzel J, Hansen N, Boasberg P. Screening-detected breast cancer in a man with BRCA2 mutation: Case Report. Radiology. 2004; 230: 553-555.
- Nguyen C, Kettler M, Swirsky M, Scott C, Krause R, Hadro JA, et al. Male breast disease: pictorial review with radiologic-pathologic correlation. Radiographics. 2013; 33: 763-779.
- Ottini L, Palli D, Rizzo S, Federico M, Bazan V, Russo A. Male breast cancer. Crit Rev Oncol Hematol. 2010; 73: 141-155.
- Ruddy KJ, Winer EP. Male Breast Cancer: Risk Factors, Biology, Diagnosis, Treatment, and Survivorship. Ann Oncol. 2013; 24: 1434-1443.
- Tai Y, Domchek S, Parmigiani G, Chen S. Breast cancer risk among male BRCA1 and BRCA2 mutation carriers. J Natl Cancer Inst. 2007; 99: 1811- 1814.
- Cavanagh H, Rogers K. The role of BRCA1 and BRCA2 mutations in prostate, pancreatic, and stomach cancers. Hereditary Cancer in Clinical Practice. 2015; 13: 1-7.
- Dietz JR, Partridge AH, Gemignani ML, Javid SH, Kuerer HM. Breast Cancer Management Updates: Young and Older, Pregnant, or Male. Ann Surg Oncol. 2015; 22: 3219-3224.
- Arem H, Brinton LA, Moore SC, Gapstur SM, Habel LA, Johnson K, et Al. Physical Activity and Risk of Male Breast Cancer. Cancer Epidemiol Biomarkers Prev. 2015; 24: 1898-1901.
- Brinton LA, Richesson DA, Gierarch GL, Park Y, Hollenbeck AR, Schatzkin A, et al. Prospective evaluation of risk factors for male breast cancer. J Natl Cancer Inst. 2008; 100: 1477-1481.
- Wang J, Kollias J, Marsh C, Maddern G. Are males with early breast cancer treated differently from females with early breast cancer in Australia and New Zealand? Breast. 2009; 18: 378-381.
- Breast Cancer Screening and Diagnosis. National Comprehensive Care Network Clinical Practice Guidelines in Oncology. Version 1.2015.
- Kriege M, Brekelmans C, Boetes C, Muller SH, Zonderland HM, Obdeijn IM, et al. Dutch MRI Screening (MRISC) Study Group. Differences between first and subsequent rounds of the MRISC breast cancer screening program for women with a familial or genetic predisposition. Cancer. 2006; 106: 2318-2326.