Review Article
Keys to Development of a Successful Mini-Thoracotomy Cardiac Surgery Program: Lessons from a 10 Year Experience
Richard C Cooka1*, Peter L Skarsgard1, Juliette Atherstone2, Jens Lohser2, Chris Durkin2, Amandeep Sidhu3, Rob Chalus3, Matt Fieldwalker3, Maria C Alaba4 and Michelle L Kent4
1Department of Cardiac Surgery, University of British Columbia, Canada
2Department of Anesthesia, University of British Columbia, Canada
3Perfusion Services, Vancouver Coastal Health, Canada
4Department of Nursing, Vancouver Coastal Health, Canada
*Corresponding author: Richard C. Cook, Department of Cardiac Surgery, University of British Columbia, St. Paul’s Hospital, 1081 Burrard St, Vancouver, British Columbia, Canada, V6Z 1Y6
Published: 07 Feb, 2017
Cite this article as: Cook RC, Skarsgard PL, Atherstone J,
Lohser J, Durkin C, Sidhu A, et al. Keys
to Development of a Successful Mini-
Thoracotomy Cardiac Surgery Program:
Lessons from a 10 Year Experience.
Clin Surg. 2017; 2: 1290.
Abstract
Surgical procedures are increasingly being performed through minimally-invasive approaches. In
addition to the obvious cosmetic benefits, patients report a quicker recovery after cardiac surgery
performed through a mini-thoracotomy, and may have a less intense inflammatory reaction, with
less blood loss, and less need for transfusion of blood products. Whether the right mini-thoracotomy
approach is able to deliver on those desired outcomes is very controversial due to a high degree of
variability in actual outcomes.
At Vancouver General Hospital, between March 2006 and October 2016, 123 consecutive patients
underwent cardiac surgical procedures through a right mini-thoracotomy approach. The mean age
was 60.7 years, and 38 of the patients were female. There was 1 peri-operative death (0.8%), with
no major CVA’s. The mean ejection fraction was 58.5%. The mean operative time was 5hours, 56
minutes. The mean operative time decreased with experience, from a mean of 6 hours, 31 minutes
for the first 10 cases, to a mean of 5 hours, 28 minutes for the last 10 cases. The mean post-operative
length of stay was 8.3 days, with a median length of stay of 6 days. The operative times differed,
depending on the type of procedure (mitral valve surgery (103 patients), tricuspid valve surgery (11
(patients), atrial septal defect closure (9 patients)). Of the patients who were taken to the operating
room for a planned mitral valve repair, 87/88 had a successful repair (98.9% planned repair rate).
Fifteen of the patients had had a prior sternotomy, and all had their operations with systemic
hypothermia and fibrillatory arrest.
The unique challenges presented by mini-thoracotomy cardiac surgery (exposure, cannulation
for cardiopulmonary bypass, myocardial protection, de-airing) will be addressed, with specific
solutions and strategies to minimize the risk of mortality or morbidity. Issues specific to patients
who have undergone a prior sternotomy-based cardiac operation will be addressed separately.
Mini-thoracotomy surgery presents the cardiac surgeon with new challenges. Excellent results can
be delivered with development of a team-based approach, and careful implementation of special
techniques and tools unique to mini-thoracotomy surgery.
Introduction
Surgical procedures are increasingly being performed through minimally-invasive approaches.
For cardiac surgical procedures, alternative approaches to the midline sternotomy include a hemisternotomy,
trans-apical approach, percutaneous femoral approach, and right mini-thoracotomy
approach. In addition to the obvious cosmetic benefits, patients report a quicker recovery after
cardiac surgery performed through a mini-thoracotomy, as opposed to cardiac surgery performed
through a sternotomy [1,2]. By avoiding the sternotomy, there also seems to be a less intense
inflammatory reaction, with less blood loss, and less need for transfusion of blood products [3].
This appears to be especially true for patients who have had prior sternotomy-based procedures.
Whether the right mini-thoracotomy approach is able to deliver on those desired outcomes is
very controversial due to a high degree of variability in actual outcomes. Several risks have been
identified, including lower rates of repair in patients undergoing mitral valve surgery, a possible
increased risk of neurologic morbidity [4,5], and in some instances, a
higher risk of death, usually due to poor myocardial protection. Our
experience at Vancouver General Hospital (VGH) would suggest
that with appropriate training and program development, all of
these potential negative outcomes can be avoided. The first step is
to recognize that right mini-thoracotomy procedures are completely
different operations than sternotomy-based cardiac operations, and
require different skill sets and tools.
This article will address the right mini-thoracotomy approach
only, and discuss the challenges which need to be overcome in order
to safely offer this type of minimally-invasive surgery to patients
with the following cardiac pathologies: mitral valve disease, tricuspid
valve disease, and atrial septal defects. The challenges of operating on
patients with prior sternotomies will also be addressed separately.
When considering minimally-invasive cardiac surgery, patient
outcomes are the most important objectives. Therefore it is essential
that the operation be conducted in a way that ensures that patients
will have the lowest possible risk of peri-operative mortality and
morbidity, and in the case of mitral valve surgery, the greatest chance
of a successful and durable repair, where that is the expected result.
Over the past 10 years, right mini-thoracotomy surgery has been
developed in a slow and deliberate fashion at VGH, in order to ensure
that patient outcomes are equal to, or better than those expected
for sternotomy-based operations. The important challenges and
solutions applied are summarized, along with a presentation of the
results from the entire group of patients who have undergone right
mini-thoracotomy surgery at VGH.
The Challenges
Exposure
Right mini-thoracotomy surgery presents several unique
challenges to the surgeon accustomed to performing cardiac surgeon
through a midline incision. The first and most obvious challenge is
visualization of the cardiac structures through a 5-6 cm incision.
This is closely accompanied by the challenge of manipulating those
structures without being able to move the heart, and without being
able to place the hands or traditional instruments through the
small incision. Tying knots deep in the chest can be a particularly
challenging and time-consuming procedure.
Cannulation for cardiopulmonary bypass (CPB)
Because the incision is small, there is not enough room for the
CPB cannula or tubing, and therefore peripheral cannulation is
required. Unlike central cannulation, where large-bore cannula can
easily be placed where necessary, peripheral cannulation requires
different strategies in order to ensure good venous drainage in all
patients, and prevent the complications secondary to suboptimal
venous drainage.
Myocardial protection
Good myocardial protection is a critical factor for successful
outcomes. Unfortunately, with retraction of the interatrial septum
with the left atrial retractor, the aortic valve is often rendered
incompetent. Therefore regular administration of intermittent
antegrade cardioplegia can be very time-consuming if the left atrial
has to be repositioned every 15-20 minutes. Placement of a retrograde
coronary sinus catheter without manual manipulation of the heart is
a skill which most sternotomy-based cardiac surgeons have not had
to learn, and can therefore be even more intimidating through a small
incision.
Technique
As with any left-sided cardiac operation, avoidance of air
embolism is critical. De-airing has to be done without the ability to
manipulate the left ventricle, and therefore a strategy of preventing
accumulation of air within the left heart is an important aspect of
mini-thoracotomy surgery.
Technique
The issue with patients who have had a prior sternotomy is
the presence of pericardial and pleural adhesions. Furthermore,
some patients have live coronary bypass grafts originating from the
ascending aorta, which makes dissection of the aorta for placement
of an antegrade cardioplegia cannula and cross clamp potentially
dangerous. Fortunately, these patients can be dealt with using a
strategy of fibrillatory arrest without clamping the aorta [4,6].
For patients with aortic valve prosthesis or ascending aortic/root
replacement, a right mini-thoracotomy approach can provide better
visualization of the mitral valve than a sternotomy-based approach.
The Solutions
Fortunately, there are now multiple good solutions to almost all of the challenges listed above. In order to take advantage of those solutions, it is best to approach right mini-thoracotomy surgery as a completely separate type of surgery, requiring its own set of unique skills, and specialized team consisting of cardiac anesthesiologists, echocardiographers, perfusionists, and nurses. Each member of the mini-thoracotomy surgery team has their own set of skills which are different from those who are only familiar with sternotomy-based cardiac operations. An article in the Harvard Business Review looked at several hospitals which were implementing minimally-invasive surgery programs, and found that the operative times were shortest at the hospital which took a team-based approach [7].
Anesthesiologists and Echocardiographers
In the case of the cardiac anesthesiologists, it is important for them
to be facile with the placement of bronchial blockers or double-lumen
endotracheal tubes so that the right lung can be collapsed. Also, some
patients require placement of a large (17-21 French) percutaneous
venous drainage catheter in the superior vena cava (SVC), via the
right internal jugular vein. Many anesthesiologists are not familiar
with managing these large bore catheters, and do not have the wire
skills to perform this task safely. At VGH, this is now done using
transesophageal echo (TEE) guidance, stiff Amplatz wires, and 3 sets
of hands in order to manage the skin incision and the exchanges of
the dilators and cannula over the long wire. Early in the experience
at VGH, two patients had to have their surgery postponed because
of inadvertent passage of the cannula into the right pleural space,
presumably from perforation of the venous system at the junction
between the right internal jugular vein, right subclavian vein, and the
SVC. Fortunately, in both instances, the patients were successfully
managed non-operatively, with removal of the cannula, placement
of a chest tube, and positive pressure ventilation. Neither suffered
any long-term morbidity, apart from blood transfusions, and both
returned to the operating room several weeks later, for sternotomybased
mitral operations.
The importance of good intraoperative TEE imaging cannot be
overestimated. Because it can be difficult to manipulate the heart
through a 5cm mini-thoracotomy incision, visualization of parts
of the heart can be poorer than it is through a sternotomy. TEE is
therefore particularly important when performing mitral valve repair
surgery. TEE is also critical for safe placement of the femoral venous
cannula, the femoral arterial cannula, and the retrograde cardioplegia
catheter, in addition to ensuring that there is no intra-cardiac air at
the end of the case.
TEE is also used to rule out major aortic pathology, such as
significant aortic atheroma, dissection, or aneurysm, which could
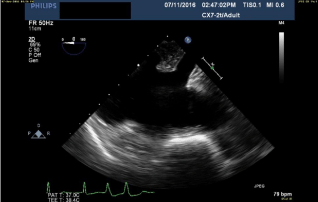
make femoral cannulation unsafe. Coronary sinus is visualized by first
obtaining the mid-esophageal 4-chamber view, and then rotating the
probe to bring the right atrium into view. The probe is then pushed
deeper in the esophagus to expose the orifice of the coronary sinus
(Figure 1).
Figure 1
Perfusionists
The input from the perfusionists at VGH has been a critical factor in the success of the program. Unlike central cannulation, venous drainage from peripherally-placed cannula can often be suboptimal. Optimal positioning of the cannula is critical in this regard. Vacuumassisted venous drainage is very helpful. Another very important development has been the introduction of the RAP femoral venous cannula 23/25fr (Sorin) cannula, which has 2 sets of side holes, and a wire-wound portion between them, which allows for retraction of the interatrial septum. The more distal holes (23fr) are placed into the SVC, and the proximal holes (25fr) sit in the IVC. When using this cannula, it is no longer necessary to place a separate venous cannula into the SVC via the right internal jugular vein, and saves approximately 30 minutes of O.R. time. However, it should be noted that we have had inadequate venous drainage using this cannula in patients > 100 kg. More recently, on the suggestion of one of our perfusionists, the pump has been moved from its usual position opposite the surgeon, to a position by the patient’s feet. This allows for a more direct route from the femoral venous cannula to the pump. When the pump is in this position, we have also minimized the length of the venous tubing, and with these 2 changes, the venous drainage has been substantially improved. Suboptimal venous drainage can obstruct the surgical field with excess blood, and may contribute to inadequate myocardial protection by allowing warm systemic blood to circulate through the heart from the right side. The perfusionists also monitor the central venous pressure (CVP). When the CVP is high, there is a theoretical risk of cerebral malperfusion, and resultant neurologic injury [8]. Another key role for the perfusionists is in maintaining higher than normal arterial perfusion pressures (i.e. >80 mmHg) when performing surgery with fibrillatory arrest (instead of cross clamping the aorta). This keeps the aortic valve closed, and minimizes the risk of inadvertent embolization of air into the aorta while the left atrium is open.
Nurses
The nurses need to be familiar with setting up and using the
specialized equipment needed for mini-thoracotomy surgery.
Needless to say, an enthusiastic nursing team can greatly impact the
conduct of the operation.
Exposure
At VGH, patients are positioned with the right side slightly
elevated. A bean bag is used to position the patient, and the right arm
is extended such that the right elbow is posterior to the patient’s back.
Care is taken to protect the lunar nerve at the elbow, and to support
the arm in order to minimize retraction on the brachial plexus. Using
this technique, there have been no incidents of brachial plexus or
ulnar nerve injuries.
A 5-6 cm right mini-thoracotomy incision is made just lateral and
inferior to the right nipple. In the case of women, the skin incision is
usually made ~ 1cm superior to the right breast crease. The lateral
edge of the pectoralis major muscle is identified, and the muscle is
retracted medially. The 3rd or 4th intercostal space is then identified,
and with the right lung deflated, the right pleural space is entered.
In the ideal circumstance, the right hilum should be visible in the
middle of the incision. This usually allows for access to both the mitral
valve and the ascending aorta, which is important for placement of
the cardioplegia and aortic root vent catheter. After the intercostal
incision is made, an Alexi’s soft tissue retractor (Applied medical) is
placed, as well as a rib-spreading retractor.
After opening the pericardium from the SVC to the IVC,
three pledgeted 2-0 Tevdek (Teleflex) sutures are placed along the
posterior edge of the pericardium. These are then brought through
the skin through separate stab incisions using a skewer to pull the
sutures through the intercostals space and skin. If the diaphragm is
in the surgical field, it can be retracted inferiorly by placing a similar
suture through the fibrous/tendinous portion of the diaphragm. Care
must be taken to avoid passing the needle too deep in the tissue, and
damaging the liver.
After developing the inter-atrial groove, the left atrium is entered,
and the mitral valve is exposed using the Edwards ergonic left
atrial retractor system with articulating blades (Edwards). Prior to
placing the left atrial retractor blade, a 3-0 gore-tex suture is placed
in the middle of the interatrial septum, in order to facilitate anterior
retraction. The Edwards retractor system has a very adjustable system
for stabilizing the retractor handle to the anterior chest wall (Figure
2). In addition to being able to be adjusted left or right, along the
interatrial septum, it also has the ability to provide more anterior
retraction by pivoting the blade perpendicular to the plane of the
interatrial septum.
Visualization of the valve is vastly improved by placement of the
annuloplasty sutures first, and securing them under traction outside
of the chest incision. Regarding visualization, several surgeons
advocate a completely videoscopic approach, as this allows for
smaller incisions, and less retraction on the interspace, both of which
may result in less post-operative pain. However, I have found it much
easier to operate using direct vision through my surgical loupes. It
is my feeling that 3 dimensional visualization of the valve is a very
important factor in performing complex mitral valve repair surgery.
By limiting the length of the incision, the amount of opening of the
intercostal space is also limited. A 10mm Endocameleon (Karl Storz)
scope is used to provide extra light in the field, and allow the assistant
and surgical nurse to see what the surgeon is doing, thereby allowing
them to be more effective and helpful to the surgeon. Because the
Endocameleon’s scope position can be rotated over 120 degrees, it
can enter the chest through a separate stab incision inferior to the
mini-thoracotomy incision. This keeps the scope well away from the
surgical field, and prevents it from interfering with the surgeon’s
movements (Figure 3).
Manipulation of the cardiac tissues through the mini-thoracotomy
incision requires long, shafted instruments. There are now several
companies who are manufacturing these instruments. The basic
minimum set should include: 2 needle drivers, a DeBakey type of
tissue forcep, a knot pusher, a suture cutter, a Chitwood cross clamp
(Sorin), hooks, and scissors. Until recently, tying the annuloplasty or
valve sutures has been one of the more time-consuming parts of the
operation, as each pair of sutures has to be tied using a knot pusher.
This usually requires coordination between the surgeon and his or her
assistant, and if not done correctly, can result in air knots or broken
sutures. Devices which eliminate the need to tie annuloplasty sutures
have been shown to significantly reduce operative times [9,10]. A new
and recent technology (Cor-Knot. LSI Solutions) eliminates the need
to tie these sutures. Instead, a titanium clip is used to crimp the two
ends of each suture, and the device cuts the suture ends at the same
time. Use of this device can save a substantial amount of operating
room time, while at the same time, reducing surgeon stress and
fatigue. It also reduces the difficulty of the operation, and may allow
for more broad application of minimally-invasive surgery.
Cannulation
A 2.5-3 cm incision is made just superior to the right groin in order
to expose only the anterior surface of the femoral artery and vein.
Purse-strings are placed on the anterior surface of each vessel using
4-0 prolene sutures. Appropriate sized heparin-bonded cannulas are
chosen according to the size of the patient and vessel. In most cases, a
21fr DLP femoral arterial (Medtronic) arterial cannula is placed, and
a Sorin 23/25fr venous cannula is placed. For placement of the venous
cannula, the bi-caval view on TEE is used (DESCRIBE). When using
the Sorin cannula, it is important to watch the wire, and the cannula,
entering the SVC. The arterial cannula is placed using TEE to confirm
placement of the guidewire in the descending thoracic aorta. In
patients who are undergoing atrial septal defect closure or tricuspid
valve surgery, or who are > 90 – 100kg, a separate cannula is placed
percutaneously into the SVC via the right internal jugular vein. This
is done at the beginning of the case with TEE guidance, as described
in the section on “Anesthesiologists”.
Myocardial protection
Since February 2010, all cases have been performed using a
combination of antegrade and retrograde cardioplegia. The retrograde
cardioplegia cannula is placed using TEE guidance (Figure 1) before
initiating cardiopulmonary bypass (CPB), and before placement of
the venous cannula.
A Chitwood cross clamp (Sorin) is placed through a separate
stab incision, usually in the mid-axillary line, as close to the axilla as
possible, in order to the keep the handles of the clamp away from the
surgical field.
After arresting the heart with antegrade cold blood cardioplegia,
500cc of cold blood retrograde cardioplegia is administered every
15 – 20 minutes, or whenever it can be administered without
excessively obstructing the surgical field. The use of cold blood
retrograde cardioplegia has been an important development in our
program as it has allowed for longer cross clamp times without risk
of myocardial injury. Unfortunately, retrograde cardioplegia cannot
always be delivered with complete confidence. In situations where
the coronary sinus pressure does not react appropriately to increases
in cardioplegia flows, we have assumed that retrograde cardioplegia
is not being delivered, and give intermittent antegrade cardioplegia
instead.
Alternative strategies for delivery of cardioplegia include the
percutaneous heart port coronary sinus catheter, which is not used
at VGH because of the expense of the catheter (~$2500/case), and
so-called single-shot cardioplegia solutions, like Custodiol HTK
(Essential Pharmaceuticals) or Del Nido solution [11,12]. Placement
of the ascending aortic cardioplegia/vent catheter is done after
initiation of CPB as the right atrial appendage usually obscures
access to the aorta proximal to the transverse sinus when the heart
is full. Two purse strings are placed: a pledgeted3-0 prolene pursestring,
and a non-pledgeted 4-0 prolene purse-string. The catheter is
placed proximal to the transverse sinus, as the posterior cross clamp
blade passes under the aorta at this level. At the end of the case, CPB
flows are minimized, and the valves are evaluated with TEE. After
the valves have been evaluated, and any left-sided intra-cardiac air
has been evacuated, the patient is placed back onto full CPB and the
aortic catheter is removed with the right atrial appendage collapsed
and out of the way.
De-Airing
In order to minimize the risk of air embolism, carbon dioxide
(CO2) is flooded into the right pleural space using a 6fr angiocath
catheter at a rate of 2 liters/minute. A weighted vent is placed into the
right superior pulmonary vein through a 4-0 prolene purse-string.
Care is taken to avoid pushing the tip of the vent beyond the orifice
of the left superior pulmonary vein, in order to avoid entraining air
into the pulmonary venous system. The rate of the vent is adjusted so
that it is just barely keeping the field clear of blood. After completion
of the repair, the weighted vent is removed, and a dedicated left
ventricular vent is placed through the mitral valve, with the vent rate
reduced to ~ 50cc/minute in order to prevent accumulation of air
in the left ventricle (LV). Before completing closure of the left atrial
incision, the heart is de-aired by turning off the LV vent, leaving
volume in the heart, and inflating the lungs. The patient is then placed
in trendelenburg position with both the aortic root and LV vents at
high flow (~500cc/minute each) before removing the cross clamp.
Using this sequence of steps, there is rarely any substantial amount
of air seen in the left heart, and no incidents of cerebral air embolism
in the VGH program.
Patients with prior sternotomy-based cardiac surgery
The mini-thoracotomy approach is a particularly good approach
for patients who require mitral valve surgery, but have had a prior
sternotomy-based procedure. In this situation, it is desirable to
minimize dissection around the heart, and an approach of ventricular
fibrillatory arrest can be used. When this strategy is used, there is
no need for cardioplegia or an aortic cross clamp, and therefore the
aortic root is not manipulated. This is important in patients who have
live coronary bypass grafts coming off of the ascending aorta. There
are 2 main methods for achieving fibrillatory arrest. The first is to use
a fibrillator. The method used at VGH is to cool the patient until the
heart fibrillates, at which point cooling is stopped, and the patient is
maintained at a temperature of < 25C. Substantial aortic insufficiency
is a contraindication to this technique. It is important to keep the
patient in the trendelenburg position while the left atrium is open,
and to keep the arterial perfusion pressures high (> 70 – 80), in order
to keep the aortic valve closed. Meticulous de-airing is also critical
with this technique. With this technique, there tends to be more
blood in the field, as there is inevitable some regurgitation of blood
through the aortic valve. Also, exposure of the anterolateral portion
of the valve, especially the anterolateral commissure area can be more
challenging because the aortic root is pressurized.
Figure 2
Figure 3
Table 1
Table 2
Results
Between March 2006 and October 2016, 123 consecutive patients underwent cardiac surgical procedures through a right minithoracotomy approach. A further 2 patients were scheduled to have right mini-thoracotomy cardiac surgery, however, their operations were abandoned due to presumed SVC injury during attempted percutaneous cannulation of the SVC via the right internal jugular vein. The mean age was 60.7 years, and 38 of the patients were female. The mean ejection fraction was 58.5%. All patients undergoing mitral valve surgery had severe mitral regurgitation or stenosis. The patients undergoing tricuspid surgery had severe tricuspid regurgitation. The patients undergoing atrial septal defect closure were not candidates for percutaneous closure, and had a significant left to right shunt. The mean operative time was 5hours, 56 minutes. The mean operative time decreased with experience, from a mean of 6hours, 31 minutes for the first 10 cases, to a mean of 5hours, 28 minutes for the last 10 cases. The mean post-operative length of stay was 8.3 days, with a median length of stay of 6 days. The operative times differed, depending on the type of procedure Table 1. No patients had a CVA, and there was only one peri-operative mortality. This was in a 77 year old woman with multiple morbidities and severe tricuspid regurgitation. She weighed 48.1 kg, and was using home oxygen at night. Although she appeared to be quite frail, she insisted that she was working with a personal trainer twice a week. Unfortunately, she had dense pericardial adhesions which prevented safe dissection around her SVC or IVC. She had been turned down for a sternotomy approach by another surgeon, and she herself did not want to proceed to a sternotomy. The operation was therefore abandoned. Although she was extubated on POD 1, she remained very weak postoperatively, and on POD 4 refused reintubation or deep suctioning, and requested that she be converted to comfort care. She passed away shortly afterwards. The majority of the cases have been mitral valve surgery cases. The type of valvular pathology is summarized in Table 2. One of the main priorities for the program at VGH has been ensuring that patients with mitral valve disease have the same rate of durable repairs as those patients having mitral valve surgery through a midline sternotomy. Therefore, all of the same repair techniques have been used, including resections, sliding plasties, and placement of 3-0 gore-tex neo chordae. When resections are performed, the amount of tissue resected is minimized. All patients at VGH received a Carpentier Edwards Physio or Physio II annuloplasty ring (Edwards). Of the patients who were taken to the operating room for a planned mitral valve repair, 87/88 had a successful repair (98.9% planned repair rate). The patient who required a mitral valve replacement was the 16th patient in the series, and the 11th planned mitral repair patient. This patient was also the only patient who required intra-operative conversion to a sternotomy approach. There were an additional two patients who had had previous cardiac surgery, and were presenting for mitral valve surgery. One had a perforated anterior leaflet secondary to a complex aortic root replacement, and the other had an anterior leaflet cleft and calcification from a prior septum premium atrial septal defect repair. Both had successful repairs, although the pre-operative expected likelihood of repair was low. Fifteen patients had a planned mitral valve replacement, 11 patients had tricuspid valve repair or replacement, and 9 patients had an atrial septal defect closed. Fifteen of the patients had had a prior sternotomy, and all had their operations with systemic hypothermia and fibrillatory arrest.
Conclusion
Patients are increasingly demanding less invasive approaches for surgical management of cardiac pathology. Unfortunately, some of the early experience with minimally-invasive cardiac surgery was suboptimal, and patient outcomes were not as good as those obtained through sternotomy-based operations. Fortunately, over the past 10 years, there have been substantial improvements in the tools available to the surgeon practicing minimally-invasive surgery. By establishing a strong team of health care professionals trained in mini-thoracotomy cardiac surgery, and by taking the time to acquire the necessary skills, excellent results can be achieved. As technology and experience continue to improve, mini-thoracotomy cardiac surgery should continue to expand, and eventually become the standard approach to many cardiac surgical procedures.
References
- Atluri P, Stetson RL, Hung G, Gaffey AC, Szeto WY, Acker MA, et al. Minimally invasive mitral valve surgery is associated with equivalent cost and shorter hospital stay when compared with traditional sternotomy. J Thorac Cardiovasc Surg. 2016; 151: 385-388.
- Cohn LH, Adams DH, Couper GS, Bichell DP, Rosborough DM, Sears SP, et al. Minimally Invasive Cardiac Valve Surgery Improves Patient Satisfaction While Reducing Costs of Cardiac Valve Replacement and Repair. Ann Surg. 1997; 226: 421-426.
- Woo YJ, Nacke EA. Robotic minimally invasive mitral valve reconstruction yields less blood product transfusion and shorter length of stay. Surgery. 2006; 140: 263-267.
- Ad N, Holmes SD, Shuman DJ, Pritchard G, Massimiano PS. Minimally invasive mitral valve surgery without aortic cross-clamping and with femoral cannulation is not associated with increased risk of stroke compared with traditional mitral valve surgery: a propensity scorematched analysis. Eur J Cardiothorac Surg. 2015; 48: 868-872.
- Bedeir K, Reardon M, Ramchandani M, Singh K, Ramlawi B. Elevated Stroke Risk Associated With Femoral Artery Cannulation During Mitral Valve Surgery. Semin Thorac Cardiovasc Surg. 2015; 27: 97-103.
- Massimiano PS, Yanagawa B, Henry L, Holmes SD, Pritchard G, Ad N. Minimally invasive fibrillating heart surgery: a safe and effective approach for mitral valve and surgical ablation for atrial fibrillation. Ann Thorac urg. 2013; 96: 520-527.
- Edmondson A, Bohmer RMJ, Pisano G. Speeding up team learning. Harvard Business Review. 2001; 125-132.
- Bainbridge DT, Chu MW, Kiaii B, Cleland A, Murkin J. Percutaneous superior vena cava drainage during minimally invasive mitral valve surgery: a randomized, crossover study. J Cardiothorac Vasc Anesth. 2015; 29: 101-106.
- Grapow MT, Mytsyk M, Fassl J, Etter P, Matt P, Eckstein FS, et al. Automated fastener versus manually tied knots in minimally invasive mitral valve repair: impact on operation time and short- term results. J Cardiothorac Surg. 2015; 10: 146.
- Cook RC, Nifong LW, Enterkin JE, Charland PJ, Reade CC, Kypson AP, et al. Significant reduction in annuloplasty operative time with the use of nitinol clips in robotically assisted mitral valve repair. J Thorac Cardiovasc Surg. 2006; 133: 1264-1267.
- Matzelle SJ, Murphy MJ, Weightman WM, Gibbs NM, Edelman JJ, Passage J. Minimally invasive mitral valve surgery using single dose antegrade Custodiol cardioplegia. Heart Lung Circ. 2014; 23: 863-868.
- Misfeld M, Davierwala P. Crystalloid-based cardioplegia for minimally invasive cardiac surgery. Semin Thorac Cardiovasc Surg. 2012; 24: 305- 307.