Editorial
Endoscopic-Vacuum-Therapy in Esophgageous Perforations and Anastomotic Leakages after Esophagectomy. The Best Treatment Option to Solve a Major Complication in Upper GI-Surgery?
Heits N1*,Egberts JH1, Schniewind B2, Hampe J3, Becker T1 and Schafmayer C1
1Department of General, Visceral-, Thoracic-, Transplantation- and Pediatric Surgery, University Hospital Schleswig-Holstein, Germany
2Department of General Surgery and Thoracic Surgery, Hospital of Lueneburg, Germany
3University Hospital TU Dresden, Germany
*Corresponding author: Nils Gerrit Heits, Department of General, Visceral-, Thoracic-, Transplantation- and Pediatric Surgery, University Hospital Schleswig-Holstein, Germany
Published: 30 Dec, 2016
Cite this article as: Heits N, Egberts JH, Schniewind
B, Hampe J, Becker T, Schafmayer
C. Endoscopic-Vacuum-Therapy
in Esophgageous Perforations
and Anastomotic Leakages after
Esophagectomy. The Best Treatment
Option to Solve a Major Complication
in Upper GI-Surgery?. Clin Surg. 2016;
1: 1281.
Editorial
Anastomotic leakages and esophageous perforations remain to be severe complications of the
upper gastrointestinal (GI) tract. Reported leak rates after esophagectomy vary widely from 1% to
30% [1]. According to the severity of the anastomotic leakage, the time interval after primary surgery,
the general condition of the patient and/or the anastomotic location several treatment options are
available, such as conservative, endoscopic or surgical treatment [2-4]. Nevertheless, the mortality
of anastomotic leakage can exceed 50% [5]. Also, in patients with esophageal perforations high
mortality rates close to 20% [6-8], despite recent progress in surgical, endoscopic, intensive care, and
infectious treatment options have been reported. The mortality rate of patients requiring surgical
revision due to an anastomotic leakage after esophageal resection are very divergent in the literature
and reach from 3.3% up to 67% [3,9-11]. Therefore, leakage from an esophageal anastomosis is a
life-threatening complication and optimal management is of tremendous importance. The surgical
treatment options reach from the redo of the anastomosis with augmentation of the anastomotic
area by vital tissue (e.g. muscle flap, omental flap) to complete surgical deviation by taking down the
esophageal conduit and creation of a cervical stoma [3,9-11].
Next to surgical revision various strategies have been employed in the management of these
complications. Several endoscopic approaches have been suggested. The by far most prevalent
approach at present is the application of covered self-expanding metallic or plastic stents. Since
the millennium more than 20 studies had referred to this topic with reported mortality rates of 0%
to 50% [4,12-21]. Next to endoscopic stenting several other techniques like clipping, application
of fibrin glue, endoscopic suturing and the application biodegradable fistula plugs have also been
suggested [22]. In the case of small, clinically silent leaks, conservative management with nasogastric
decompression and tube feeding has been suggested [23]. The difficulty of managing the septic
condition in major anastomotic leakages remains a major problem with all of these strategies. In
this context the endoscopic endoluminal vacuum therapy (EVT), an endoscopic vacuum-assisted
wound closure system, might add to the currently available therapeutic strategies by addressing the
septic focus by using constant vacuum drainage of infected wound fluid from the wound cavity.
This treatment option has been introduced and successfully used in the last decade [3,4]. With this
technique the endoscope is used to appraise the size of the wound cavity, and apolyurethane sponge
is trimmed to the cavity size. In cases of a large cavity a second sponge can be placed to fully seal
and better drain the cavity. An over tube is placed over the endoscope and brought into the cavity
under vision using a pusher device. The placed sponge is connected to the drainage tube, which
was diverted through the nose and connected to a vacuum pump orredon bottle that provided a
continuous negative pressure of 70 to 125 mmHg [2]. The replacement intervals are every two to
3 days depending on the condition of the patient. Longer intervals have shown to be a risk for
in growth and device integration of the sponge [24]. Advantages of the EVT-procedure are the
possibility of an endoscopic jet lavage, active closure and drainage of the septic wound cavity and
the inspection of the wound cavity during the change of the sponge at least every three days. The
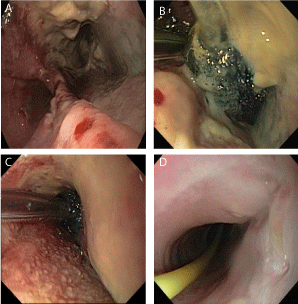
use of EVT is terminated when the remaining wound cavity is closed and granulated (Figure 1a-1d).
Several studies showed, that under EVT the inflammatory
septic response can be controlled and stabilized by drainage of the
mediastinal leakage cavity and a healing of the wound cavity can be
achieved [12,13,25]. Different studies reported good results in short
term survival of 80-96% [4,12-18] in EVT for anastomotic leakages
and perforations. A successful healing of the leakage or perforation
cavity is reported in 84-100% [26-34]. Recently the first groups from
the USA and South Korea were able to confirm these good results of
wound closure and survival [30,31]. Furthermore, three studies from
Germany and South Korea showed a higher success rate in healed
and closed anastomotic leakages comparing EVT and stent therapy
[31,34,35] (Table 1). The healing rates were reported to be successful
in 84-100% after EVT and 54-64% after stent placement. EVT-related
complications. Anastomotic strictures after EVT are a known longterm
complication with an incidence 4.2-16% [15-18,31], which
can be successfully cured by endoscopic balloon dilation in most
of the cases. A reason for the higher incidence is mostly due to the
inflammatory response in the leakage cavity and following enhanced
cicatrization.
Due to these good results in esophageous perforations and
leakages of the upper GI-tract different authors recently started to
use EVT to drain leakages after bariatric sleeve gastrectomy or gastric
bypass surgery. Moreover, some case reports reported of a successful
use of EVT to drain infectious pancreatic pseudocysts or leakage of
a pancreaticogastrostomy [36-40]. However, all studies are single
center studies and case reports with small numbers of treated patients
without reporting on long-term survival and Quality of Life. To
prove the efficacy of the endoscopic vacuum therapy at the upper GItract
and assess clinical data in an international multicenter study a
prospective open online registry was set up in collaboration between
the University Hospital Schleswig-Holstein, Campus Kiel, and the
department of Medical Scientific Affairs of the company B.Braun©,
Aesculap AG [41].
Figure 1
Figure 1
EVT in a patient after an iatrogenic esophageous perforation before start of EVT (a), after initial sponge placement (b), during therapy 9 days after start of the EVT (c) and the follow up endoscopy with the healed perforation cavity 7 days after successful EVT (d).
Table 1
Table 1
Published data of outcome and treatment characteristics for patients treat by EVT for anastomotic leakages after esophagectomy or esophageous perforations in 2009-2016 (EVT: Endoscopic Vacuum Therapy, d: days, n.s.: not shown).
Conclusion
EVT is an effective procedure for the management of major leakage from esophageal anastomoses and other conditions in the upper gastrointestinal tract and might be superior especially in septic patients compared to surgical revision and stent placement. In addition, in selected cases EVT might also be a good treatment option to drain septic cavities in the upper GI-tract and abdominal cavity after bariatric and pancreatic surgery. Decisive for the rate of success using EVT therapy is an interdisciplinary work between thoracic surgery, radiology, endoscopy, and intensive care medicine departments in an experienced center.
References
- Wu PC, Posner MC. The role of surgery in the management of oesophageal cancer. The lancet oncology. 2003; 4: 481-488.
- Ahrens M, Schulte T, Egberts J, Schafmayer C, Hampe J. Drainage of esophageal leakage using endoscopic vacuum therapy: a prospective pilot study. Endoscopy. 2010; 42: 693-698.
- Crestanello JA, Deschamps C, Cassivi SD, Nichols FC, Allen MS. Selective management of intrathoracic anastomotic leak after esophagectomy. J Thoracic Cardiovasc Surg. 2005; 129: 254-260.
- Feith M, Gillen S, Schuster T, Theisen J, Friess H. Healing occurs in most patients that receive endoscopic stents for anastomotic leakage; dislocation remains a problem. Clin gastroenterol hepatol. 2011; 9: 202-210.
- Pennathur A, Luketich JD. Resection for esophageal cancer: strategies for optimal management. The Ann Thorac Surg. 2008; 85: S751-S756.
- Chirica M, Champault A, Dray X, Sulpice L, Munoz-Bongrand N. Esophageal perforations. J Visc Surg. 2010; 147: e117-e128.
- Brinster CJ, Singhal S, Lee L, Marshall MB, Kaiser LR. Evolving options in the management of esophageal perforation. Ann Thorac Surg. 2004; 77: 1475–483.
- Abbas G, Schuchert MJ, Pettiford BL, Pennathur A, Landreneau J. Contemporaneous management of esophageal perforation. Surgery. 2009; 146: 749–756.
- Griffin SM, Lamb PJ, Dresner SM, Richardson DL, Hayes N. Diagnosis and management of a mediastinal leak following radical oesophagectomy. Br J Surg. 2001; 88: 1346-1351.
- Martin LW, Swisher SG, Hofstetter W, Correa AM, Mehran RJ. Intrathoracic leaks following esophagectomy are no longer associated with increased mortality. Ann Surg. 2005; 242: 392-399.
- Page RD, Shackcloth MJ, Russell GN, Pennefather SH. Surgical treatment of anastomotic leaks after oesophagectomy. Eur J Cardiothoracic Surg. 2005; 27: 337-343.
- D'Cunha J, Rueth NM, Groth SS, Maddaus MA, Andrade RS. Esophageal stents for anastomotic leaks and perforations. J Thorac Cardiovasc Surg. 2011; 142: 39-46.
- Nguyen NT, Rudersdorf PD, Smith BR, Reavis K, Nguyen XM. Management of gastrointestinal leaks after minimally invasive esophagectomy: conventional treatments vs. endoscopic stenting. J Gastrointest Surg. 2011; 15: 1952-1960.
- Schweigert M, Dubecz A, Stadlhuber RJ, Muschweck H, Stein HJ. Treatment of intrathoracic esophageal anastomotic leaks by means of endoscopic stent implantation. Interact Cardiovasc Thorac Surg. 2011; 12: 147-151.
- Laukoetter MG, Mennigen R, Neumann PA, Dhayat S, Horst G. Successful closure of defects in the upper gastrointestinal tract by endoscopic vacuum therapy (EVT): a prospective cohort study. Surg Endosc. 2016.
- Möschler O, Nies C, Mueller MK. Endoscopic vacuum therapy for esophageal perforations and leakages. Endosc Int Open. 2015; 3: E554-E558.
- Bludau M, Hölscher AH, Herbold T, Leers JM, Gutschow C. Management of upper intestinal leaks using an endoscopic vacuum-assisted closure system (E-VAC). Surg Endosc. 2014; 28: 896-901.
- Schorsch T, Müller C, Loske G. Endoscopic vacuum therapy of anastomotic leakage and iatrogenic perforation in the esophagus. Surg Endosc. 2013; 27: 2040-2045.
- Dai YY, Gretschel S, Dudeck O, Rau B, Schlag PM. Treatment of oesophageal anastomotic leaks by temporary stenting with self-expanding plastic stents. Br J Surg. 2009; 96: 887-891.
- Leers JM, Vivaldi C, Schafer H, Bludau M, Brabender J. Endoscopic therapy for esophageal perforation or anastomotic leak with a self-expandable metallic stent. Surg endosc. 2009; 23: 2258-2262.
- Salminen P, Gullichsen R, Laine S. Use of self-expandable metal stents for the treatment of esophageal perforations and anastomotic leaks. Surg eEdosc. 2009; 23: 1526-5230.
- Hampe J, Schniewind B, Both M, Fritscher-Ravens A. Use of a NOTES closure device for full-thickness suturing of a postoperative anastomotic esophageal leakage. Endoscopy. 2010; 42: 595-598.
- Matory YL, Burt M. Esophagogastrectomy: reoperation for complications. J Surg Oncol. 1993; 54: 29-33.
- Schniewind B, Schafmayer C, Both M, Arlt A, Fritscher-Ravens A. Ingrowth and device disintegration in an intralobar abscess cavity during endosponge therapy for esophageal anastomotic leakage. Endoscopy. 2011; 43: E64–E65.
- Pennathur A, Luketich JD. Resection for esophageal cancer: strategies for optimal management. The Ann Thorac Surg. 2008; 85: S751-S756.
- Weidenhagen R, Hartl WH, Gruetzner KU, Eichhorn ME, Spelsberg F. Anastomotic leakage after esophageal resection: new treatment options by endoluminal vacuum therapy. Ann Thorac Surg. 2010; 90: 1674-1681.
- Wedemeyer J, Brangewitz M, Kubicka S, Jackobs S, Winkler M. Management of major postsurgical gastroesophageal intrathoracic leaks with an endoscopic vacuum-assisted closure system. Gastrointest endosc. 2010; 71: 382-386.
- Loske G, Schorsch T, Muller C. Endoscopic vacuum sponge therapy for esophageal defects. Surg endosc. 2010; 24: 2531-2535.
- Heits N, Stapel L, Reichert B, Schafmayer C, Schniewind B. Endoscopic endoluminal vacuum therapy in esophageal perforation. Ann Thorac Surg. 2014; 97: 1029-1035.
- Schniewind B, Schafmayer C, Voehrs G, Egberts J, von Schoenfels W. Endoscopic endoluminal vacuum therapy is superior to other regimens in managing anastomotic leakage after esophagectomy: a comparative retrospective study. Surg Endosc. 2013; 27: 3883-3890.
- Brangewitz M, Voigtlander T, Helfritz FA, Lankisch TO, Winkler M. Endoscopic closure of esophageal intrathoracic leaks: stent versus endoscopic vacuum-assisted closure, a retrospective analysis. Endoscopy. 2013; 45: 433-438.
- Kuehn F, Schiffmann L, Rau BM, Klar E. Surgical endoscopic vacuum therapy for anastomotic leakage and perforation of the upper gastrointestinal tract. J Gastrointest Surg. 2012; 16: 2145-2150.
- Smallwood NR, Fleshman JW, Leeds SG, Burdick JS. The use of endoluminal vacuum (E-Vac) therapy in the management of upper gastrointestinal leaks and perforations. Surg Endosc. 2016; 30: 2473-2480.
- Hwang JJ, Jeong YS, Park YS, Yoon H, Shin CM. Comparison of Endoscopic Vacuum Therapy and Endoscopic Stent Implantation With Self-Expandable Metal Stent in Treating Postsurgical Gastroesophageal Leakage. Medicine (Baltimore). 2016; 95: e3416.
- Mennigen R, Harting C, Lindner K, Vowinkel T, Rijcken E. Comparison of Endoscopic Vacuum Therapy Versus Stent for Anastomotic Leak After Esophagectomy. J Gastrointest Surg. 2015; 19: 1229-1235.
- Wedemeyer J, Kubicka S, Lankisch TO, Wirth T, Patecki M. Transgastrically placed endoscopic vacuum-assisted closure system as an addition to transgastric necrosectomy in necrotizing pancreatitis (with video). Gastrointest Endosc. 2012; 76: 1238-1241.
- Wallstabe I, Tiedemann A, Schiefke I. Endoscopic vacuum-assisted therapy of infected pancreatic pseudocyst using a coated sponge. Endoscopy. 2012; 44: E49-E50.
- Seyfried F, Reimer S, Miras AD, Kenn W, Germer CT. Successful treatment of a gastric leak after bariatric surgery using endoluminal vacuum therapy. Endoscopy. 2013; 45: E267-E268.
- Schorsch T, Muller C, Loske G. Pancreatico-gastric anastomotic insufficiency successfully treated with endoscopic vacuum therapy. Endoscopy. 2013; 45: E141-E142.
- Fischer A, Richter-Schrag HJ, Hoeppner J, Braun A, Utzolino S. Endoscopic intracavitary pull-through vacuum treatment of an insufficient pancreaticogastrostomy. Endoscopy. 2014; 46: E218-E219.
- Eso-Sponge Registry, B.Braun©, Aesculap AG.