Research Article
The Predictive Value of the Modified Glasgow Prognostic Score (Mgps) in Determining Outcome Following Elective Colorectal Cancer Surgery
Thomas IM, Amin A, Szczachor J, Rayner S, Wood K and Lim M*
Department of General Surgery, Raigmore Hospital, UK
*Corresponding author: Michael Lim, Department of General Surgery, Raigmore Hospital, 4th Floor, Zone 7, Inverness, IV2 3UJ, Scotland, UK
Published: 30 Dec, 2016
Cite this article as: Thomas IM, Amin A, Szczachor
J, Rayner S, Wood K, Lim M. The
Predictive Value of the Modified
Glasgow Prognostic Score (Mgps)
in Determining Outcome Following
Elective Colorectal Cancer Surgery. Clin
Surg. 2016; 1: 1279.
Abstract
Background: The mGPS (modified Glasgow Prognostic Score) provides a measure of systemic
inflammation in patients with cancer, based on C-reactive protein (CRP) and Albumin. The aim
of this study was to determine the role of mGPS in predicting outcome following elective curative
colorectal cancer resections; in terms of survival, stage of disease, likelihood and interval of cancer
recurrence.
Methods: We in Raigmore Hospital, Inverness have a prospective database of patients undergoing
colorectal cancer resections. This database was reviewed between January 2008 and January 2012,
yielding 986 patients. Data on pre-operative CRP and Albumin, survival, pathology, and recurrence
was collected retrospectively, with patients followed up to 1st February 2015. Patients were classified
according to their mGPS score. The mGPS was calculated as follows: CRP< 10mg/L=mGPS 0; CRP
>10mg/L or Albumin≤ 35g/L=mGPS 1; CRP >10mg/L and Albumin≤ 35g/L=mGPS 2. Data was
found to be parametric and comparisons between mGPS groups were done using the ANOVA test.
A p-value of < 0.05 was deemed significant.
Results: 387 patients were eligible for inclusion (having had elective curative resections with a
calculable mGPS). Survival was significantly different at 45.7, 39.9 and 35.9 months for mGPS groups
of 0-2 respectively (p=0.0012). Although there was a trend towards higher TNM staging with rising
mGPS, this was not significant (p=0.054). Higher mGPS predicted an increased chance of disease
recurrence, with a 17.6% vs. 31.6% vs. 37.1% recurrence rate for mGPS groups of 0-2 respectively.
Higher mGPS scores were also associated with a significantly shorter time to recurrence, at 42.3 vs.
34.9 vs. 28.9 months (p=0.0008).
Conclusion: The mGPS has value in predicting survival, disease recurrence and time to recurrence
in colorectal cancer. The score may be helpful as a decision making tool for the multidisciplinary
team.
Introduction
Colorectal cancer (CRC) is the most common cancer of the GI tract and the third most common
cause of cancer death in western countries [1]. There are 41,000 new cases of CRC recorded each
year in the UK with 15900 deaths per year [2]. Despite the improvement and advances in treatment,
approximately half of those undergoing surgery with curative intent subsequently die from the
disease [3].
Currently we rely on accurate staging of the tumour to determine its prognosis. There are other
patient related factors like nutritional status and functional decline that is associated with poorer
outcome independent of tumour stage. These factors are subjective, and therefore not very reliable
[4]. Survival rates vary in CRC patients, even in those with the same tumour node metastasis [5]. It is therefore important to identify a preoperative indicator of poor postoperative outcome to help
guide treatment of such patients.
There is good evidence that the presence of a systemic inflammatory response is a key factor
in the determination of a poor prognosis [4]. Cancer can induce local or systemic inflammation,
the cascade is mediated by the activation of transcription factors and the production of major
inflammatory cytokines which can influence cell proliferation, cell survival, angiogenesis, tumour
cell migration, invasion, metastasis and inhibition of adaptive immunity [6]. CRC has a close
relationship with inflammation – this is typified by inflammatory
bowel diseases which have premalignant lesion that can lead to the
development of CRC [7]. Cyclooxygenase-2 inhibitors and nonsteroidal
anti-inflammatory drugs have been found to decrease the
incidence of colorectal adenoma and CRC [8].
The modified Glasgow Prognostic Score (mGPS) provides a
measure of systemic inflammation in patients with cancer, based on
C-reactive protein (CRP) and albumin measurements. The mGPS
provides a score which ranges between 0 and 2. The mGPS is one of
a group of inflammatory marker-based predictive scores which has
been shown to have prognostic utility in oesophageal, non-small cell
lung and prostate cancers [9-11].
A rise in CRP concentrations is often associated with a
corresponding fall in albumin - this inverse relationship was noted
across different tumour types [4]. Albumin concentrations reflect
both systemic inflammation and the amount of lean tissue, and
it would be important to determine the prognostic value of both
combined [12]. The GPS was recently modified on the basis that
hypoalbuminemia in patients without an elevated CRP concentration
had no significant association with cancer-specific survival [13].
The modified score is simple to measure and can be standardised
worldwide. There have been a few studies to show the prognostic
value of mGPS in colorectal cancer [14-18], hitherto, this is the first
study which examined the role of mGPS in the prediction of more
commonly examined variables after curative surgery. The aim of this
study was to determine the role of mGPS in predicting outcome after
elective curative colorectal surgery with attention to survival, disease
stage, and likelihood of recurrence.
Table 1
Methods
Raigmore Hospital in Inverness is a large district general
hospital. Patients who met the inclusion criteria, namely those with
non metastatic colorectal cancer who underwent an elective colonic
resection as part of the treatment programme were included in the
study. All patients with colonic cancers would have had staging CT
scans of their chest, abdomen and pelvis. Those with rectal cancers
would have had an MRI scan for local staging in addition to the
above mentioned CT scans. Patient would have had their diagnosis
confirmed with preoperative biopsies at time of colonoscopy. All
patients were discussed routinely in the MDT meeting. A prospective
database of all patients undergoing colorectal cancer resections is
maintained. In order to capture data for this study, the database was
reviewed to capture patients with a diagnosis of primary colorectal
cancer over a 5 year period between January 2008 and January 2012.
This review identified a total of 986 patients.
After identification of this cohort of patients, all demographic
data and pre-operative CRP and Albumin data were collected
retrospectively. This involved examination of casenotes electronically
and when necessary, hard copies of medical records. CRP and
albumin levels were usually (but not always) measured as part of
anaesthetic work-up prior to surgery. This enabled a calculation of a
mGPS score for the majority of patients. Patients were grouped into
one of three mGPS categories depending on their CRP and albumin
measurement. Table 1 details the calculation of the mGPS score.
Unfortunately patient who did not have a CRP or albumin measured
preoperatively had to be excluded from the study. Likewise, patients
who presented as emergencies and/or had other obvious adverse
conditions at the time of blood collection were excluded from the
study as their CRP and/or albumin measurement would have been
predictably abnormal. Patients unfit for surgery and patients who
had non-curative resections, largely for purposes of palliation were
excluded from the study. As this was a longitudinal study to examine
the role of the mGPS, patients who died prior to their surgery or
within 30 days of their operation were also excluded.
Surgery was performed by one of 4 fully accredited colorectal
surgeons within the department of general surgery. All postoperative
histopathology was recorded using a standard proforma. Followup
data for the above cohort of patients was collected until 1st
February 2015. Patients who had potentially curative surgery were
then followed up in our nurse led clinic. Our follow up programme
consists of regularly consultations at 3 monthly intervals for the first
year. Thereafter consultations are conducted at 6 monthly intervals
for another 4 years. Cross sectional imaging in the form of CT scans
(chest, abdomen and pelvis) are routinely performed 6 monthly for
the first 3 years. Endoluminal surveillance in the form of colonoscopy
to exclude metachronous lesions and disease recurrence are done at
one and 5 years after index surgery.
All recorded data was found to be parametric. Chi-Square analysis
and ANOVA analysis was performed for categorical and continuous
data respectively, A p-value of < 0.05 was deemed significant.
Table 2
Table 3
Table 4
Table 5
Figure 1
Figure 2
Figure 3
Results
There were three hundred and eighty seven patients that were
appropriate for analysis. These patients had complete data (with a
calculable mGPS) and were therefore included in the final analysis.
The remaining 587 were excluded. Table 2 provides further details
and reasons for their exclusion.
There were 214 (55%) males. Mean age at time of surgery was
69.4 years. The most common site for cancer in our study population
was within the rectum. Further details for our study population are
provided in Table 3. More than 40 percent of our patients had lymph
node positive disease on postoperative histology. Despite excluding
patients with known metastatic disease (after preoperative staging)
from our study, a further 1.8% of patients were found to have features
of metastatic disease within their postoperative histology. Further
details of postoperative histopathology are provided in Table 4.
Neoadjuvant therapy was utilised in approximately 10.6% of
patients, this was utilised predominantly for patients with margin
threatened rectal cancers. Patients with lymph node positive disease
were offered adjuvant chemotherapy if fit. This was offered to 27.4%
of patients in our study population.
The vast majority of patients in the study had a mGPS score of 0
(57.4%). Less than a quarter had a mGPS of 1 (24.5%) and finally only
18.1% had an mGPS of 2. Survival was significantly poorer for patients
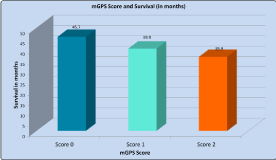
with a higher mGPS score. As shown in Figure 1, mean survival was
45.7, 39.9 and 35.9 months for mGPS groups of 0-2 respectively,
with standard deviations of 18.7, 22 and 24.5 months. The difference
between groups was significant on ANOVA testing (p=0.0012).
Disease stage was more advanced in patient with a higher mGPS
score although this did not reach statistical significance. (p=0.054).
The stage of disease according to mGPS score is detailed in Table 5.
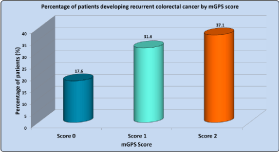
On long term follow-up, 95 out of 387 patients (24.5%) developed
recurrent cancer. As shown in Figure 2, a higher mGPS was associated
with a greater likelihood of disease recurrence. Disease recurrence
had developed in 17.6% vs. 31.6% vs. 37.1% of patients with mGPS
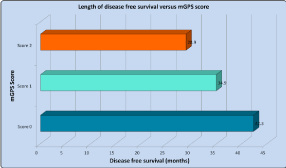
scores of 0-2 respectively (p=0.0007). Figure 3 illustrates that a higher
mGPS was also associated with a significantly shorter interval to
disease recurrence, at 42.3 vs. 34.9 vs. 28.9 months for mGPS groups
of 0-2 respectively (p=0.0008), with standard deviations of 21.1, 22.6
and 26.4 months.
Discussion
The purpose of this study was to assess the prognostic utility of
the mGPS score in a cohort of patient with colorectal cancer who
underwent potentially curative surgery. We have demonstrated that
patients with higher mGPS scores have a greater likelihood of disease
recurrence and poorer survival on long term follow-up. Currently,
postoperative pathological examination is the gold standard for
predicting postoperative outcome; of course this is only possible after
surgical resection. Even with such information available to clinicians,
it has been shown that patients can have very different outcomes
despite having very similar diseases stages [17]. Thus, there remains a
need for a preoperative predictive marker.
Studies have shown that elevated CRP may be associated with
tumour size, distant metastasis, vascular invasion, lymph node
metastasis, and tumour recurrence, resulting in poor prognosis [19].
The GPS, an inflammation based prognostic score involving CRP and
albumin was designed by Forrest et al. [20]. CRP and albumin are
acute phase proteins, which are produced in the liver in response to
inflammatory cytokines, mainly IL-6 and IL-1β [21]. CRP elevates
in response to an inflammatory stimulus, but albumin is known
to decrease in cancer patients due to malnutrition and systemic
inflammation [12].
The utility of simple markers such as CRP and albumin is
interesting. Links between cancer and inflammation have been
postulated since the 19th century – these have been based on
observations that tumours often arise at sites of chronic inflammation
and that inflammatory cells are often present in biopsy specimens
collected from tumour tissue [7 and 14]. In colitis associated colon cancer,
chronic inflammation causes oxidative damage to the DNA, leading
to p53 mutations in tumour cells, and the inflamed epithelium
and the inflammatory microenvironment at the tumour border
can influence several key stages of invasion and metastasis [22]. In
general, inflammation is known to affect all phases of carcinogenesis
and may be responsible for the initiation of genetic mutation.
Inflammation may modify the tissue microenvironment directly,
which permits cancer cells to progress and metastasize. Alternatively,
inflammation may allow tumour progression by indirect suppression
of the immune response to tumour cells [23].
We are conscious that this paper has several weaknesses. First,
this study has been done retrospectively, so it was not possible to
ensure CRP and albumin measurements in all suitable patients. There
was also a subset of patients that had to be excluded due to missing
data. We limited our dataset because we were strict with our exclusion
criteria. In order to exclude potential confounders, we were careful
not to include patients who may have had artificially raised CRPs
from concomitant pathology or obstruction at time of emergency
presentation. However, if such patients (i.e. patients with emergency
presentation and tumour perforations) were to be included in the
overall analysis, we would have detected an even stronger relationship
between high mGPS scores and poorer outcomes. This would have
led to positive bias in the study.
The mGPS should not replace gold standard assessments for
disease staging or follow-up nor should it replace the time honoured
surgical judgement about appropriateness for surgery. However, the
mGPS may be used to help aid decision making. We believe that it is
particularly useful in the aging octogenarian with borderline mental,
physical and social function, where the ‘time to benefit’ from surgery
for their colorectal cancer is severely curtailed because of advance
age and concurrent co-morbidity. If elderly patients have technically
operable disease, but are marginally fit or unsure about an operation,
a poor mGPS score preoperatively may provide reassurance to the
clinician that surgery is less likely to be in the interest of the patient.
Conclusion
The mGPS has prognostic value in patients with colorectal cancer for the prediction of survival, likelihood of disease recurrence and interval to such an occurrence. In frail patients where surgery can be fraught with risk, the use of the mGPS may help the multidisciplinary team to make the right decision.
References
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA cancer j clin. 2012; 62: 10-29.
- Cancerstats Cancer Research UK. 2014.
- Oliphant R, Nicholson GA, Horgan PG, Molloy RG, McMillan DC, Morrison DS, et al. Deprivation and colorectal cancer surgery: longerterm survival inequalities are due to differential postoperative mortality between socioeconomic groups. Ann Surg Oncol. 2013; 20: 2132-2139.
- KMcMillan DC. The systemic inflammation-based Glasgow Prognostic Score: a decade of experience in patients with cancer. Cancer Treat Rev. 2013; 39: 534-540.
- Roxburgh CS, Salmond JM, Horgan PG, Oien KA, McMillan DC. The relationship between the local and systemic inflammatory responses and survival in patients undergoing curative surgery for colon and rectal cancers. J Gastrointest Surg. 2009; 13: 2011-2019.
- Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008; 454: 436-444.
- Triantafillidis JK, Nasioulas G, Kosmidis PA. Colorectal cancer and inflammatory bowel disease: epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategies. Anticancer Res. 2009; 29: 2727- 2737.
- Rostom A, Dubé C, Lewin G, Tsertsvadze A, Barrowman N, Code C, et al. Nonsteroidal anti-inflammatory drugs and cyclooxygenase-2 inhibitors for primary prevention of colorectal cancer: a systematic review prepared for the US Preventive Services Task Force. Ann Intern Med. 2007; 146: 376-389.
- Shafique K, Proctor MJ, McMillan DC, Leung H, Smith K, Sloan B, et al. The modified Glasgow prognostic score in prostate cancer: results from a retrospective clinical series of 744 patients. BMC Cancer. 2013; 13: 292.
- Zhang P, Mian Xi, Qiao-Qiao Li, Li-Ru He, Shi-Liang Liu, Lei Zhao, et al. The modified Glasgow Prognostic Score in an independent prognostic factor in patients with inoperable thoracic esophageal squamous cell carcinoma undergoing chemoradiotherapy. J Cancer. 2014; 5: 689-695.
- Pinato DJ, Shiner RJ, Seckl MJ, Stebbing J, Sharma R, Mauri FA. Prognostic performance of inflammation-based prognostic indices in primary operable non-small cell lung cancer. Br J Cancer. 2014; 110: 1930-1935.
- McMillan DC, Watson WS, O'Gorman P, Preston T, Scott HR, McArdle CS. Albumin concentrations are primarily determined by the body cell mass and the systemic inflammatory response in cancer patients with weight loss. Nutr Cancer. 2001; 39: 210-213.
- McMillan DC, Crozier JE, Canna K, Angerson WJ, McArdle CS. Evaluation of an inflammation-based prognostic score (GPS) in patients undergoing resection for colon and rectal cancer. Int J Colorectal Dis. 2007; 22: 881- 886.
- Eren T, Burcu B, Tombalak E, Ozdemir T, Leblebici M, Ozemir IA, et al. Clinical Significance of the Glasgow Prognostic Score for Survival after Colorectal Cancer Surgery. J Gastrointest Surg. 2016; 20: 1231-1238.
- Woo HD, Kim K, Kim J. Association between preoperative C-reactive protein level and colorectal cancer survival: a meta-analysis. Cancer Causes Control. 2015; 26: 1661-1670.
- Guthrie GJ, Roxburgh CS, Farhan-Alanie OM, Horgan PG, McMillan DC. Comparison of the prognostic value of longitudinal measurements of systemic inflammation in patients undergoing curative resection of colorectal cancer. Br J Cancer. 2013; 109: 24-28.
- Lin MS, Huang JX, Yu H. Prognostic significance of Glasgow prognostic score in patients with stage II colorectal cancer. Int J Clin Exp Med. 2015; 8: 19138-19143.
- Nozoe T, Matono R, Ijichi H, Ohga T, Ezaki T. Glasgow Prognostic Score (GPS) can be a useful indicator to determine prognosis of patients with colorectal carcinoma. Int Surg. 2014; 99: 512-517.
- Kinoshita A, Onoda H, Imai N, Iwaku A, Oishi M, Tanaka K, et al. The Glasgow Prognostic Score, an inflammation based prognostic score, predicts survival in patients with hepatocellular carcinoma. BMC cancer. 2013; 13: 52.
- Forrest LM, McMillan DC, McArdle CS, Angerson WJ, Dunlop DJ. Evaluation of cumulative prognostic scores based on the systemic inflammatory response in patients with inoperable non-small-cell lung cancer. Br J Cancer. 2003; 89: 1028-1030.
- Choi KW, Hong SW, Chang YG, Lee WY, Lee B, Paik IW, et al. Inflammation-based score (Glasgow prognostic score) as an independent prognostic factor in colorectal cancer patients. Ann Surg Treat Res. 2014; 86: 309-313.
- Terzić J, Grivennikov S, Karin E, Karin M. Inflammation and colon cancer. Gastroenterology. 2010; 138: 2101-2114.
- Trinchieri G. Cancer and Inflammation: An Old Intuition with Rapidly Evolving New Concepts. Annu Rev Immunol. 2012; 30: 677-706.