Research Article
Hartmann’s Procedure versus Primary Anastomosis for Emergency Left Colectomy: A Retrospective Cohort Study
Hugo Teixeira Farinha, Fabian Grass, Emmanuel Melloul, Dieter Hahnloser, Nicolas Demartines and Martin Hübner*
Department of Visceral Surgery, University Hospital of Lausanne (CHUV), Switzerland
*Corresponding author: Martin Hübner, Department of Visceral Surgery, University Hospital CHUV, 1011 Lausanne, Switzerland
Published: 13 Dec, 2016
Cite this article as: Farinha HT, Grass F, Melloul E,
Hahnloser D, Demartines N, Hübner
M. Hartmann’s Procedure versus
Primary Anastomosis for Emergency
Left Colectomy: A Retrospective Cohort
Study. Clin Surg. 2016; 1: 1247.
Abstract
Background: The ideal surgical strategy for left emergency colectomy remains controversial and is decided on a case-by-case basis. The aim of this study was to analyze our current practice and
outcomes to define a standardized approach.
Method: This retrospective review included all consecutive patients who underwent left emergency
colectomy between July 2006 and June 2013. Demographics, surgical data, and postoperative
outcomes were compared between patients with primary anastomosis (PA group) and those having
Hartmann’s procedure (HP group).
Results: Final analysis included 148 patients (89 men, median age 76 (range 22-95) years). Patients
with HP (n=73) were older and had higher ASA score (p< 0.001) and Charlson index (p< 0.001)
than patients with PA (n=75). Indications were similar between the comparative groups, but
noradrenalin requirements and Hinchey III/IV status were more frequent in the HP group. Patients
with HP had higher in-hospital mortality (15 vs. 4%, p=0.020), overall (78 vs. 56%, p=0.005) and
severe complications (29 vs. 17%, p=0.033). Anastomotic leak rate was 8% (n=6) after PA. Only
18 patients (25%) of the HP group proceeded to stoma take-down, while all patients with PA were
stoma-free 12 months after hospital discharge.
Conclusion: Emergent left-sided colectomy remains associated with high postoperative morbimortality.
Patient’s condition and intraoperative findings appear to guide surgical decision-making.
Hartmann’s procedure does not prevent adverse outcomes and entails a high permanent ostomy
rate.
Abbreviations
ASA: American Society of Anaesthesiologists; ICU: Intensive Care Unit; PA: Primary Anastomosis; HP: Hartmann Procedure; SSI: Surgical Site Infection
Introduction
Minimal-invasive surgery and optimized perioperative care by standardized multimodal
pathways have lead to significant improvements of outcomes after elective colonic resections that
are mostly performed by colorectal specialist surgeons [1,2]. In the emergency setting, multimodal
pathways are challenging and surgery is often performed by general surgeons or junior staff. In
addition, patients’ condition and intraoperative findings are worse when compared to the elective
situation. Therefore, reported outcomes are considerably worse with reported morbidity and
mortality rates up to 40 and 20%, respectively [3-6]. All of the above explains why Hartmann’s
procedure is often the preferred approach for emergent left-sided resections in order to avoid
anastomosis and shorten the operative time [7]. The reported permanent ostomy rate is high
(75% for ASA III) after Hartmann’s procedure and more recent reports have suggested interesting
alternatives to this strategy [8-11].
The aim of the present study was to assess our institutional practice and outcomes for emergency
left colectomy comparing Hartmann’s procedure and resections with primary anastomosis.
Figure 1
Methods
Patients
This retrospective analysis included all consecutive patients who underwent emergency leftsided
colonic resection from July 2006 to June 2013 in the department of visceral surgery, in
Lausanne University Hospital. Left-sided resections included formal
and extended left colectomy as well as sigmoidectomy, while rectal
resections and total colectomies were excluded. In addition, patients
with two-stage procedure and planned second look and deferred
anastomosis or HP were not included. Emergency intervention
was defined as being performed during an unplanned admission.
Surgeries were performed by the general surgeon on call (all board
certified). The study was approved by the local Ethics Committee.
STROBE statement was followed for analysis (www.strobe-statement.
org) and reporting, and the study were online-registered (www.
researchregistry.com; UIN: 1750).
Hartmann’s procedure (HP group) was compared with resections
and synchronous primary anastomosis with or without protective
ileostomy (PA group).
Data collection
Demographics and risk factors as well as outcome measures were
defined a priori and entered in a computerized database. Documented
co-morbidities included diabetes (insulin-dependent and no insulindependent),
overweight (>25 (Kg/m2)), chronic renal failure,
cirrhosis, cardiomyopathy, tobacco smoking or immunosuppressive
treatment including corticoids, anti-TNF and chemotherapy [12-14].
Patient preoperative co-morbidities were prospectively graded using
the American Society of Anesthesiology (ASA) score and Charlson
co-morbidity Index [15,16]. Surgical data included operative
time, surgeon’s expertise (junior or senior consultant), estimated
blood loss (ml), as well as intraoperative vasopressor requirements
(Noradrenalin >10ug/min intravenously) [13,17].
Outcomes
Mortality and postoperative complications were recorded until
30 days after surgery according to the Clavien classification. Grade III
and IV were defined as severe complications [18]. Ostomy rate was
assessed at one year after index surgery.
Other outcomes included length of hospital stay (days), length
of intensive care unit (ICU) stay (days), destination after discharge
(home or rehabilitation) and time to stoma reversal (months).
Reasons not to close the stoma were entered in the database.
Statistical analysis
Descriptive statistics for continuous variables were reported as
median (interquartile range: IQR and range), categorical variables
were reported as frequency (%). Chi-square was used for comparison
of categorical variables and the Wilcoxon test for continuous data. All
statistical tests were two-sided and a level of 0.05 was used to indicate
statistical significance. Data analyses were performed using SPSS Inc.
released 2012. For Mac (Version 21.0. Chicago, USA).
Table 1
Table 2
Table 3
Results
Patients
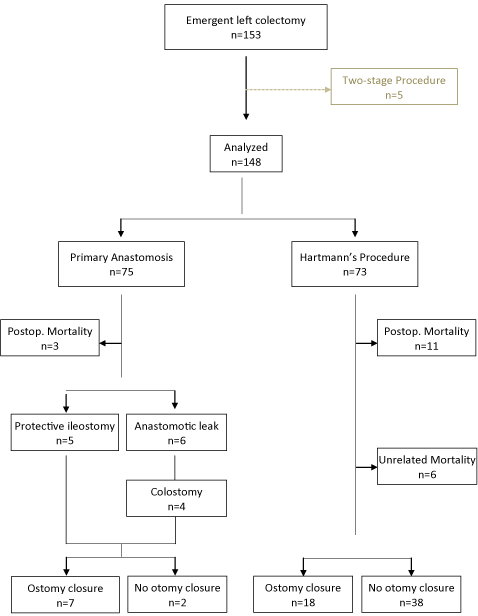
One hundred and fifty-three patients underwent emergency
left colectomy during the study period. Five patients underwent
a two-stage procedure with planned second look; these patients
were excluded from analysis according to the study protocol. The
remaining 148 patients were eligible for analysis (Figure 1). Seventythree
patients (49%) underwent a primary HP, while primary
anastomosis was performed in 75 (51%) patients. Protective ileostomy
was constructed in 5 out of these 75 patients (6.7%).
Demographic information for the two comparative groups
is displayed in Table 1. Patients having a HP were older and had
significantly more co-morbidities (higher ASA score, higher Charlson
index, higher prevalence of smoking) as their counterparts in the PA
group.
Surgical data
Indications for emergency left colectomy were similar in both
groups. Perforation was the most frequent indication (overall 62%),
followed by mechanical obstruction (overall 24%) and haemorrhage
(overall 9%); other causes for emergent left colectomy were volvulus
(n=3), Crohn’s disease (n=2) and ileus due to adhesions (n=2) (Table
2).
Causes for perforation were diverticulitis 66% (n=61), obstruction
due to malignancy 19% (n=17), iatrogenic perforation post
colonoscopy (n=6), ischemia (n=5), abdominal trauma (n=2) and
faecaloma (n=1). Mechanical obstruction without perforation was
caused by malignancy in 70% (n=25) of patients and by inflammatory
stenosis due to diverticulitis in 30% (n=11). Haemorrhage was due
to inflammatory colitis in 8 patients and due to diverticulosis in=5
patients.
The median operation time was 180 min (62–466 min) in PA
group versus 174 min (52-350min) for no-PA group (p=0.228).
Five procedures with primary anastomosis were performed
laparoscopically, 2/73 in the HP group (p=0.442). 36/75 anastomoses
in the PA group were stapled. Hinchey classification was applicable
in 31 and 30 patients in PA vs. HP group, respectively. Five and
fifteen patients in the HP group had Hinchey class III and IV, as
compared with 8 and 3 patients in the PA group, respectively. More
patients in the HP group required elevated doses of Noradrenalin
intraoperatively (56 vs. 35%, p=0.013). No significant difference
was found with regards to qualification of the operating surgeon
(junior vs. senior) or the timing of the procedure (working hours vs.
nightshift (Table 2).
Clinical outcomes
Eleven patients (15%) died after HP, and 3 (4%) patients after
resection with primary anastomosis 4% in patients with primary
anastomosis (p=0.020), giving an overall mortality of 9%. In the PA
group, 2 patients died of multiple organ failure (MOF) following
septic shock of abdominal origin and one of pulmonary embolism.
Causes of death in the HP group were abdominal sepsis with
consecutive MOF in 8 patients, ruptured abdominal aortic aneurysm
(causing colonic ischemia) (n=2), and bronchoaspiration and acute
myocardial infarction in one patient each (Table 3)
Overall complication rate after emergent left-sided colectomy
was 66%. Patients having HP procedure had significantly more
overall and severe complications. Type and incidence of individual
complications are detailed in Table 3. Anastomotic leak occurred
in 6 patients of the PA group (8%). Two leaks were managed by reanastomosis
and 4 with re-exploration and terminal colostomy. Two
had HP reversal the remaining two had definitive colostomy due to
medical contraindication.
Mean ICU stay and hospital stay was significantly longer in the
HP group. Patients with PA were more likely to be discharged home
as compared to more transfers to rehabilitation units for patients after
HP (Table 3).
Ostomy closure
Six patients of the HP group died after discharge due to unrelated
causes. Accounting for the post-operative mortalities (n=11), 57
patients were eligible for HP reversal. Eighteen of those patients
(32%) actually proceeded to ostomy closure. Median time to ostomy
take-down was 6 (3-31) months. No anastomotic leak occurred
after ostomy closure. Causes for no ostomy closure were medical
contraindication (n=28), and patients’ preference or unknown in 29
patients.
Five primary anastomoses were protected with a loop ileostomy;
all protective’s ileostomy was closed. Four colostomies were created
for anastomotic leaks; two were closed.
Discussion
In this series, morbi-mortality remains high after emergent
left-sided colectomy irrespective of the surgical strategy. Patient’s
condition and intraoperative findings appear to guide intraoperative
decision-making. HP does not prevent adverse outcomes and entails
a high permanent ostomy rate.
The present study was not designed to compare primary
anastomosis vs. HP for left-sided resections. Not surprisingly,
patients in the HP group were older, sicker and had worse
intraoperative findings (Hinchey class, noradrenalin requirements).
Most surgeons would have decided probably for the “safer” HP in
this context. However, permanent ostomy rates after HP are between
50 and 75% [8]. Furthermore, it has never been convincingly
shown that HP is really the better option in terms of postoperative
morbidity. A prospective study with comparative groups wellmatched
by randomization could answer this question but is difficult
to conduct. In a multicenter randomized trial comparing HP vs. PA
and protective loop ileostomy in patients with left-sided colonic
perforation (Hinchey III/IV), the study was discontinued after an
interim analysis of 30 patients in the HP group and 32 patients with
primary anastomosis [11]. Morbi-mortality was equally high (67 vs.
57%, 9 vs. 13%) in both groups after the initial surgery. However,
PA group compared favorably with regards to higher stoma reversal
rate (90 vs. 57%) and significantly better outcomes after stoma takedown
in terms of operation time, complications and costs [11]. The
main limitation of this study was the non-inclusion of a considerable
number of eligible patients, arguably those in worse shape. Our data
confirmed that anastomotic leak rate and outcomes in patients with
PA and without protective ileostomy compared favorably to the
outcomes of patients with PA with ileostomy. However, it remains
unclear whether protective ileostomy is necessary in this setting. This
is paramount since stoma-associated morbidity remains high [11].
For the case of diverticular perforation, organ-sparing
approaches such as laparoscopic lavage have been suggested to lower
complications and secondary resection rates [9]. On the other hand,
organ-preserving lavage is not an option for the other frequent
indications such as obstruction, ischemia and bleeding [19-22].
Several attempts have been made to target risk factors for worse
outcomes after colonic resection. In addition to the emergency, which
is a risk factor for postoperative complications patient-related risk
factors are age (>70 years), gender (male), ASA score >3, diabetes,
smoking or immunosuppression. Procedure-related risk factors
include intra-operative blood transfusion, hemodynamic instability,
surgeon expertise or interventions performed during night-shift [12-
14]. Some of those risk factors were significantly more prevalent in
patients having HP and have probably influenced surgeons’ decision
to abstain from primary anastomosis. However, it remains unknown
whether primary anastomosis with our without protective ileostomy
would have been worse in this context. Interestingly, surgeon’s
expertise and timing of surgery were not significantly different for
both comparative groups. Other studies showed a clear tendency for
less open resection and less HP, when emergency resections were
performed during daylight and by colorectal specialists [1,2].
For the reasons mentioned above, 2-stage procedures have been
suggested with 2nd look and definitive surgery 24-48h after initial
resection [23]. In line with principles of trauma surgery, only sepsis
control is performed by initial resection of the diseased bowel part
limiting the additional surgical trauma and allowing immediate
resuscitation in the intensive care unit. Early postoperative course
and operative status during 2nd look surgery help to define better
whether primary anastomosis can be performed.
One of the major limitations of the study includes a single-center
experience involving a relatively small number of patients. The
described results can therefore not be generalized. Surgical strategy
was at the discretion of the surgeon on call and no standardized
algorithm was available in our institution at that time. This is likely
to be similar in many institutions, as no widely accepted algorithm
exists so far for the optimal strategy of emergent left-sided resections.
Conclusion
High morbi-mortality and high permanent ostomy rates remain the challenge after emergent left-sided colectomy and the ideal strategy remains yet to be defined. Primary HP does not prevent severe surgical complications, but primary anastomosis appears to be safe in selected patients in good condition and with favorable intraoperative findings. In the remaining patients, two-stage procedure with planned 2nd look and deferred anastomosis could help to improve early outcomes and to reduce permanent ostomy rates. A prospective study is underway to evaluate this strategy.
References
- Zorcolo L, Covotta L, Carlomagno N, Bartolo DCC, O’Connell PR. Toward Lowering Morbidity, Mortality, and Stoma Formation in Emergency Colorectal Surgery: The Role of Specialization. Dis Colon Rectum. 2003; 46: 1461-1468.
- Biondo S, Kreisler E, Millan M, Fraccalvieri D, Golda T, Frago R, et al. Impact of surgical specialization on emergency colorectal surgery outcomes. Arch Surg. 2010; 145: 79-86.
- van der Pas MH, Haglind E, Cuesta M a, Fürst A, Lacy AM, Hop WC, et al. Laparoscopic versus open surgery for rectal cancer (COLOR II): shortterm outcomes of a randomised, phase 3 trial. Lancet Oncol. 2013; 14: 210- 218.
- Gibbons G, Tan CJ, Bartolo DCC, Filgate R, Makin G, Barwood N, et al. Emergency left colonic resections on an acute surgical unit: Does subspecialization improve outcomes? ANZ J Surg. 2015; 85: 739-743.
- Zorcolo L, Covotta L, Carlomango N, Bartolo DCC. Safety of primary anastomosis in emergency colo-rectal surgery. Color Dis. 2003; 5: 262-269.
- Tabbara M, Velmahos GC, Butt MU, Chang Y, Spaniolas K, Demoya M, et al. Missed opportunities for primary repair in complicated acute diverticulitis. Surgery. 2010; 148: 919-924.
- Greco M, Capretti G, Beretta L, Gemma M, Pecorelli N, Braga M. Enhanced recovery program in colorectal surgery: A meta-analysis of randomized controlled trials. World J Surg. 2014; 38: 1531-1541.
- Banerjee S, Leather AJM, Rennie JA, Samano N, Gonzalez JG, Papagrigoriadis S. Feasibility and morbidity of reversal of Hartmann’s. Color Dis. 2005; 7: 454-459.
- Myers E, Hurley M, O’Sullivan GC, Kavanagh D, Wilson I, Winter DC. Laparoscopic peritoneal lavage for generalized peritonitis due to perforated diverticulitis. Br J Surg. 2008; 95: 97-101.
- Breitenstein S, Kraus A, Hahnloser D, Decurtins M, Clavien PA, Demartines N. Emergency left colon resection for acute perforation. Primary anastomosis or hartmann’s procedure? A case-matched control study. World J Surg. 2007; 31: 2117-2124.
- Oberkofler CE, Rickenbacher A, Raptis DA, Lehmann K, Villiger P, Buchli C, et al. A multicenter randomized clinical trial of primary anastomosis or Hartmann’s procedure for perforated left colonic diverticulitis with purulent or fecal peritonitis. Ann Surg. 2012; 256: 819-826.
- Kingham TP, Pachter HL. Colonic Anastomotic Leak: Risk Factors, Diagnosis, and Treatment. J Am Coll Surg. 2009; 208: 269-278.
- Leichtle SW, Mouawad NJ, Welch KB, Lampman RM, Cleary RK. Risk factors for anastomotic leakage after colectomy. Dis Colon Rectum. 2012; 55: 569-575.
- Ziegler MA, Catto JA, Riggs TW, Gates ER, Grodsky MB, Wasvary HJ. Risk factors for anastomotic leak and mortality in diabetic patients undergoing colectomy: analysis from a statewide surgical quality collaborative. Arch Surg. 2012; 147: 600-605.
- Saklad M. Grading of patients for surgical procedures. Anesthesiol. 1941; 2: 281-284.
- Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987; 40: 373-383.
- Adanir T, Nazli O, Kara C, Aksun M, Sozutek A, Sencan A, et al. The relationship between vasopressor dose and anastomotic leak in colon surgery: An experimental trial. Int J Surg. 2010; 8: 221-224.
- Dindo D, Demartines N, Clavien P-A. Classification of Surgical Complications. Ann Surg. 2004; 240: 205-213.
- Schultz JK, Yaqub S, Wallon C, Blecic L, Forsmo HM, Folkesson J, et al. Laparoscopic Lavage vs Primary Resection for Acute Perforated Diverticulitis: The SCANDIV Randomized Clinical Trial. JAMA. 2015; 314: 1364-1375.
- Kaushik M, Bhullar JS, Bindroo S, Singh H, Mittal VK. Minimally Invasive Management of Complicated Diverticular Disease: Current Status and Review of Literature. Dig Dis Sci. 2016; 61: 663-672.
- Swank HA, Vermeulen J, Lange JF, Mulder IM, van der Hoeven JAB, Stassen LPS, et al. The ladies trial: laparoscopic peritoneal lavage or resection for purulent peritonitis and Hartmann’s procedure or resection with primary anastomosis for purulent or faecal peritonitis in perforated diverticulitis (NTR2037). BMC Surg. 2010; 10: 29.
- McDermott FD, Collins D, Heeney A, Winter DC. Minimally invasive and surgical management strategies tailored to the severity of acute diverticulitis. Br J Surg. 2014; 101: e90-99.
- Teixeira Farinha H, Melloul E, Hahnloser D, Demartines N, Hübner M. Emergency right colectomy: which strategy when primary anastomosis is not feasible? World J Emerg Surg. 2016; 11: 19.