Case Report
Primary Adrenal Dedifferentiated Leiomyosarcoma: A Low Grade Leiomyosarcoma with a High-Grade Undifferentiated Component: Case Report
Anatoliy Rudin1, Jorge Torres-Mora2, Neena Natt3 and Melanie L. Richards1*
1Division of Breast, Endocrine, Metabolic and Gastrointestinal Surgery, Mayo School of Graduate Medical Education, USA
2Laboratory Medicine and Pathology, Mayo School of Graduate Medical Education, USA
3Division of Endocrinology, Mayo School of Graduate Medical Education, USA
*Corresponding author: Melanie L. Richards, Division of Breast, Endocrine, Metabolic and Gastrointestinal Surgery, Mayo School of Graduate Medical Education, USA
Published: 02 Dec, 2016
Cite this article as: Rudin A, Torres-Mora J, Natt N,
Richards ML. Primary Adrenal
Dedifferentiated Leiomyosarcoma:
A Low Grade Leiomyosarcoma
with a High-Grade Undifferentiated
Component: Case Report. Clin Surg.
2016; 1: 1193.
Abstract
Primary adrenal leiomyosarcomas are very rare tumors that have been reported in approximately
30 cases in literature. This is a case report of a 72 year old man who was diagnosed with a 5.1
cm left adrenal mass. Patient underwent surgical resection and diagnosed with a primary adrenal
leiomyosarcoma with two distinct histologic components. Patient did not have any adjuvant
treatment and no evidence of recurrence 3 months postoperatively.
Keywords: Adrenal gland; Adrenal tumor; Dedifferentiated; Leiomyosarcoma; Immunohistochemistry
Introduction
Leiomyosarcomas are rare tumors that account for approximately 10% of soft tissue sarcomas and are diagnosed primarily in middle-aged and older adults with occurrence more commonly in the abdomen and retroperitoneum. Of those, primary adrenal leiomyosarcomas are exceedingly rare with approximately 30cases reported in literature to date (1-13,15-16,20-35). Pain may be the initial presenting symptom, and leiomyosarcomas may reach a significant size before diagnosis is made. Histologically, these tumors resemble native smooth muscle but rare low grade cases can show a distinct high grade component that lacks immunoreactivity for smooth muscle markers, a phenomenon known as dedifferentiation. To our knowledge, this is the first case of a primary adrenal leiomyosarcoma with well documented dedifferentiation.
Case Presentation and Management
A 72 year old man with a past medical history of type 2 diabetes mellitus, hypertension,
hyperlipidemia, COPD, hypothyroidism, and morbid obesity (BMI 41.7) presented for evaluation
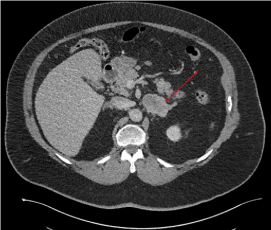
of an enlarging left adrenal mass discovered two years ago. At that time, the patient had a workup
for left sided abdominal pain and CT scan of the abdomen and pelvis revealed a 1.4 cm left
adrenal nodule (Figure 1). The patient had negative biochemical testing for hyperaldosteronism,
hypercortisolism and pheochromocytoma. A recommendation was made to follow up with repeat
imaging in 6 to 12 months.
However, the patient came back for follow up two years later and repeat CT abdomen/pelvis
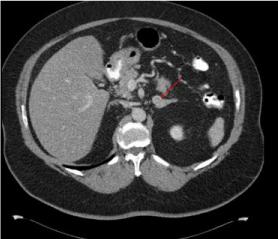
showed progressive enlargement of the adrenal mass to 5.1 cm (Figure 2). Hounsfield units ranged
between 40 and 60 with a contrast washout of 22%. The right adrenal gland appeared normal. No
additional abdominal masses were noted. The liver, kidneys, spleen, pancreas, and bowel appeared
normal.
He denied polyuria, polydipsia, and no changes in energy level. His diabetes progressed and
he now required addition of insulin to his regimen. His adrenal functional studies confirmed a
suppressed cortisol following dexamethasone challenge, normal renin, aldosterone, metanephrines,
and a normal 24-hour urine free cortisol. The patient had a history of a recent fall and adrenal
hemorrhage was considered in the differential given significant enlargement over a short time
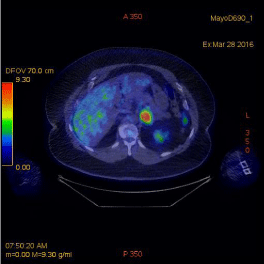
frame. Therefore, PET/CT was obtained and showed uptake in the adrenal gland (Figure 3).
Recommendation was made for surgical resection.
The patient underwent an attempted laparoscopic adrenalectomy.
However, dissection was difficult due to a desmoplastic reaction
around the tumor and procedure was converted to open left
adrenalectomy. The left adrenal gland tumor was resected with
negative margins. The patient was discharged on postoperative day
number 3 with no major complications.
Pathologic evaluation
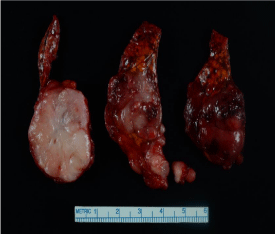
The mass was located towards the lateral aspect of the adrenal
gland and showed two distinct components, including a 4.6 cm
tan-white, lobulated fleshy area that focally grew into the lumen of
the adrenal vein, and an adjacent 3.0 cm red and hemorrhagic soft
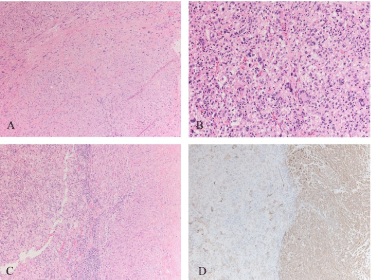
area (Figure 4). Under light microscopy with hematoxylin and eosin
stained slides, the tan-white area of the tumor corresponded to a
low grade leiomyosarcoma and consisted of intersecting fascicles
of spindle cells with atypical cigar shaped nuclei and abundant
eosinophilic cytoplasm. Scattered mitotic figures were present but no
necrosis was seen (Figure 5A). By immunohistochemistry, performed
by conventional methods, the spindle cells were diffusely and strongly
positive for smooth muscle actin (SMA) (Figure 5D) and negative
for desmin, HMB45, MITF, and inhibin (not depicted). The red
and hemorrhagic area corresponded to the dedifferentiated element
and was composed of patternless sheets of polygonal, markedly
pleomorphic and frequently multinucleated cells with abundant
eosinophilic to foamy cytoplasm and a variable number of infiltrating
lymphocytes (Figure 5B). The pleomorphic cells show focal and weak
SMA reactivity in a nonspecific pattern (Figure 5D) but they were
negative for the rest of the markers mentioned above. The transition
between the two components was sharp (Figure 5C). The final
pathology diagnosis was dedifferentiated leiomyosarcoma. Tumor
tissue was sent for cytogenetic study but unfortunately the results
revealed a normal karyotype, consisting with fibroblast overgrowth.
Follow up
The patient was seen in follow up by medical and radiation
oncology and recommendations were made for close follow up with
serial imaging. He had an MRI of abdomen and pelvis three months
after resection with no evidence of recurrent disease.
Figure 1
Figure 1
CT scan abdomen/pelvis 2014 demonstrating 1.4 cm enhancing left adrenal nodule with indeterminate characteristics.
Figure 2
Figure 2
CT scan abdomen/pelvis 2016 demonstrating 5.1 enhancing left adrenal nodule. The mass demonstrated heterogeneous enhancement with indeterminate washout characteristics.
Figure 3
Figure 3
NMPET/CT of abdomen and pelvis. The 5 cm left adrenal mass has an SUV max of 11.6 consistent with malignancy.
Figure 4
Figure 4
Cross-sections of the adrenal mass. The tumor was located towards the lateral aspect of the adrenal gland. The 4.6 cm tan-white, lobulated fleshy area (slice on the left) corresponds to the low grade leiomyosarcoma component. The 3.0 cm red and hemorrhagic soft area (center and right slices), corresponds to the undifferentiated component.
Figure 5
Figure 5
A: Low grade leiomyosarcoma component, Hematoxylin and eosin stain (H&E), 10x magnification: Intersecting fascicles of spindle cells with atypical cigar shaped nuclei and abundant eosinophilic cytoplasm, a very characteristic appearance of leiomyosarcomas.
B: Undifferentiated component, H&E stain, 20x magnifications: patternless sheets of polygonal, markedly pleomorphic and frequently multinucleated cells with abundant eosinophilic to foamy cytoplasm and a variable number of infiltrating lymphocytes.
C: Intersection between the low and high grade components of the tumor, H&E stain, 10x magnification: Notice the sharp transition between the two components and the increased number of infiltrating lymphocytes at the intersection.
D: Smooth muscle actin immunoperoxidase (SMA), 10x magnification: Same area as depicted in figure 2C. Notice the strong and diffuse staining in the low grade leiomyosarcoma (right side) as opposed to the weak and very focal staining seen in the undifferentiated area (right side).
Discussion
The etiology of primary adrenal leiomyosarcoma has not been
clearly elucidated. These tumors are believed to originate from the
smooth muscle wall of the central adrenal vein and its branches [1].
It has been suggested that HIV and EBV could play a role [2,3]. The
presence of a high grade component that lacks evidence of smooth
muscle differentiation, and arising in association with a conventional
leiomyosarcoma is a rare event known as dedifferentiation. This
phenomenon is not limited to smooth muscle tumors and is well
known to occur in soft tissue and bone sarcomas and also in some
carcinomas. It is important to document this finding since its presence
signifies a more aggressive tumor behavior [17]. Our case fulfilled the
histologic criteria for the diagnosis of dedifferentiated leiomyosarcoma
of the adrenal gland. Due to the location and unusual appearance of
the tumor, other diagnostic possibilities were considered, including a
collision tumor composed of PEComa or adrenal cortical carcinoma,
arising in association with a leiomyosarcoma. However, the results of
the immunoperoxidase studies ruled out those possibilities.
The typical age of presentation can range from 30-78 years, mean
age of 56, with similar frequency of occurrence in both males and
females [4]. Identification of the tumor at an early stage is difficult
because symptoms are non- specific and may include abdominal
pain, flank pain, fever, vomiting, or weight loss [5-13]. Our patient
presented with left sided abdominal pain and his adrenal mass was
incidentally noted on imaging. There are no reliable tumor markers
that are useful for early detection; however, neuron-specific enolase
has been reported as a potential marker for tumor progression or
recurrence [9]
Retroperitoneal leiomyosarcomas in general are associated with
a high mortality rate with a 5 year survival rate of approximately
20–30 % [14]. This could be related to the size of tumor at the time
of presentation and difficulty in achieving clear surgical margins. In
clinically reported cases, these tumors range from 3 to 27 cm with an
average size of approximately 10 cm [4], and lungs, liver and bone
are reported to be primary sites of metastasis [11,15,16]. The tumor
can be locally aggressive with local tissue extension and thrombotic
complications, including inferior vena cava and renal vein thrombosis
[4].
The literature on treatment of primary adrenal leiomyosarcomasis
limited and the majority of the data is extrapolated from study of
retroperitoneal sarcomas [18-19]. This topic remains controversial
with no strong treatment recommendations except for surgical
resection. The overall goal of surgery is to achieve complete
macroscopic resection with negative margins. The role of both
chemotherapy and radiation therapy in this setting has not been
clearly determined [20-23].
Our patient had negative resection margins and postoperative
external beam radiation after complete resection was considered to
be of limited value [24-26]. High dose radiation that would be needed
for postoperative treatment could result in significant toxicity to
adjacent organs with short and long term consequences [27-32].
The role of adjuvant chemotherapy has not been established.
However, in the setting of leiomyosarcomas with a dedifferentiation
component greater than 5 cm, there is data to suggest modest
potential for benefit [33-35]. However, analogous data is not present
for dedifferentiated tumors less than 5 cm. The risk of recurrent
disease over time can approach as high as 30%, and is influenced
by tumor histology and degree of surgical resection. However,
reduction of this risk with systemic chemotherapy is questionable.
We recommended close observation and follow up for our patient
with a possibility for radiation and or chemotherapy for recurrence.
Repeat surgical resection of local recurrences can be offered on an
individualized basis and could be considered in patients with low
grade leiomyosarcoma and long disease-free intervals.
Conclusion
This is the first documented case of a primary adrenal dedifferentiated leiomyosarcoma with clear demonstration of the two histologic components, including a low grade leiomyosarcomajuxta posed to a high grade element that lacks evidence of smooth muscle differentiation. Our patient underwent surgical resection with close follow up. Current literature on treatment of primary adrenal leiomyosarcoma is limited with no clear treatment recommendations except for surgical resection and chemo-radiation therapy consideration on a case by case basis.
References
- Lee CW, Tsang YM, Liu KL. Primary adrenal leiomyosarcoma. Abdom Imaging, 2006; 31: 123-124.
- Tomasich FD, Luz Mde A, Kato M, Targa GZ, Dias LA, Zucoloto FJ, et al. Primary adrenal leiomyosarcoma. Arq Bras Endocrinol Metabol. 2008; 52: 1510-1514.
- Zetler PJ, Filipenko JD, Bilbey JH, Schmidt N. Primary adrenal leiomyosarcoma in a man with acquired immunodeficiency syndrome (AIDS). Further evidence for an increase in smooth muscle tumors related to Epstein-Barr infection in AIDS. Arch Pathol Lab Med. 1995; 119: 1164- 1167.
- Jianguo Wei, Aijing Sun, Jian Tao, Cheng Wang, Fang Liu. Primary adrenal leiomyosarcoma: case report and review of the literature. Int J SurgPathol. 2014; 22: 722-726.
- Alam MM, Naser MF, Islam MF, Rahman MA. Primary adrenal leiomyosarcoma in an adult female. Mymensingh Med J. 2014; 23: 380- 383.
- Bhalla A, Sandhu F, Sieber S. Primary adrenal leiomyosarcoma: a case report and review of the literature. Conn Med. 2014; 78: 403-407.
- Candanedo-González FA, Vela Chávez T, Cérbulo-Vázquez A. Pleomorphic leiomyosarcoma of the adrenal gland with osteoclast-like giant cells. Endocr Pathol. 2005; 16: 75-81.
- Deshmukh SD, Babanagare SV, Anand M, Pande DP, Yavalkar P. Primary adrenal leiomyosarcoma: a case report with immunohistochemical study and review of literature. J Cancer Res Ther. 2013; 9: 114-116.
- Goto J. A rare tumor in the adrenal region: neuron-specific enolase (NSE)- producing leiomyosarcoma in an elderly hypertensive patient. Endocr J. 2008; 55: 175-181.
- Hamada S, Ito K, Tobe M, Otsuki H, Hama Y, Kato Y, et al. Bilateral adrenal leiomyosarcoma treated with multiple local therapies. Int J Clin Oncol. 2009; 14: 356-360.
- Lack EE, Graham CW, Azumi N, Bitterman P, Rusnock EJ, O'Brien W, et al. Primary leiomyosarcoma of adrenal gland. Case report with immunohistochemical and ultrastructural study. Am J Surg Pathol. 1991; 15: 899-905.
- Nanpo Y. [Primary adrenal leiomyosarcoma: a case report]. Nihon Hinyokika Gakkai Zasshi. 2009; 100: 640-645.
- Mencoboni M, Bergaglio M, Truini M, Varaldo M. Adrenal leiomyosarcoma: a case report and literature review. Pathology. 2003; 35: 47-49.
- Hashimoto H, Tsuneyoshi M, Enjoji M. Malignant smooth muscle tumors of the retroperitoneum and mesentery: a clinicopathologic analysis of 44 cases. J Surg Oncol. 1985; 28: 177-186.
- Boudewijn van Etten, Marc GA van Ijken, Wolter J Mooi, Matthijs Oudkerk, Albertus N van Geel. Primary leiomyosarcoma of the adrenal gland. Sarcoma. 2001; 5: 95-99.
- Mario GL, Mai PH. Pleomorphic leiomyosarcoma of the adrenal gland. Arch Pathol Lab Med, 2003; 127: 32-35.
- Chen E, O'Connell F, Fletcher CD. Dedifferentiated leiomyosarcoma: clinicopathological analysis of 18 cases. Histopathology. 2011; 59: 1135- 1143.
- ESMO / European Sarcoma Network Working Group. Soft tissue and visceral sarcomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014; 25: 2-12.
- Pisters PW, BO Sullivan. Retroperitoneal sarcomas: combined modality treatment approaches. Curr Opin Oncol. 2002; 14: 400-405.
- C Wong, U O Von Oppell, D Scott-Coombes. Cold feet from adrenal leiomyosarcoma. J R Soc Med. 2005; 98: 418-420.
- Dekou A, N'dah JK, Kouame B, Kohou L, Abroulaye F, Gowe E, et al. [Primary leiomyosarcoma of adrenal gland, in black African woman: diagnosis and therapeutically aspects]. Prog Urol. 2013. 23: 421-424.
- Dugan MC. Primary adrenal leiomyosarcoma in acquired immunodeficiency syndrome. Arch Pathol Lab Med. 1996; 120: 797-798.
- Fernández JM, Huescar AM, Ablanedo P, Rábade CJ, Pérez García FJ, Rodríguez Martínez JJ, et al., [Primary leiomyosarcoma. A rare tumor of the adrenal gland]. Arch Esp Urol. 1998; 51: 1029-1031.
- Murat Tolga Gulpinar, Asif Yildirim, Berrin Gucluer, Ramazan Gokhan Atis, Cengiz Canakci, Cenk Gurbuz, et al. Primary leiomyosarcoma of the adrenal gland: a case report with immunohistochemical study and literature review. Case Rep Urol. 2014; 2014: 489630.
- Kato T, Kato T, Sakamoto S, Kobayashi T, Ikeda R, Nakamura T. Primary adrenal leiomyosarcoma with inferior vena cava thrombosis. Int J ClinOncol. 2004; 9: 189-192.
- Seungkoo Lee, Gail Domecq C Tanawit, Rolando A Lopez, Jaime T Zamuco, Betsy Grace G Cheng, Menandro V Siozon. Primary leiomyosarcoma of adrenal gland with tissue eosinophilic infiltration. Korean J Pathol. 2014; 48: 423-425.
- Mencoboni M, Bergaglio M, Truini M, Varaldo M. Primary adrenal leiomyosarcoma: a case report and literature review. Clin Med Oncol. 2008; 2: 353-356.
- Yoshiyuki Matsui, Keita Fujikawa, Hiroya Oka, Shigeki Fukuzawa, Hideo Takeuchi. Adrenal leiomyosarcoma extending into the right atrium. Int J Urol. 2002; 9: 54-56.
- Mohanty SK, Balani JP, Parwani AV. Pleomorphic leiomyosarcoma of the adrenal gland: case report and review of the literature. Urology. 2007; 70: 591.e5-7.
- Nagaraj V, Mustafa M, Amin E, Ali W, Naji Sarsam S, Darwish A. Primary adrenal leiomyosarcoma in an arab male: a rare case report with immunohistochemistry study. Case Rep Surg. 2015; 2015: 702541.
- Ozturk H. Vena Cava invasion by Adrenal Leiomyosarcoma. Rare Tumors. 2014; 6: 5275.
- Javier Pereira-Beceiro, Andrés Rodríguez-Alonso, José Manuel Janeiro- Pais, Juan Carlos Alvarez-Fernández, Cristina Durana Tonder. Adrenal pleomorphic leiomyosarcoma, an uncommon malignant suprarenal tumour. Cir Esp. 2015; 93: 256-258.
- Quildrian S, Califano I, Carrizo F, Daffinoti A, Calónico N, et al., Primary adrenal leiomyosarcoma treated by laparoscopic adrenalectomy. Endocrinol Nutr. 2015; 62: 472-473.
- Van Laarhoven HW, Vinken M, Mus R, Flucke U, Oyen WJ, Van der Graaf WT. The diagnostic hurdle of an elderly male with bone pain: how 18F-FDG-PET led to diagnosis of a leiomyosarcoma of the adrenal gland. Anticancer Res. 2009; 29: 469-472.
- Zhou Y, Tang Y, Tang J, Deng F, Gong G, Dai Y. Primary adrenal leiomyosarcoma: a case report and review of literature. Int J Clin Exp Pathol. 2015; 8: 4258-4263.