Case Report
A Case of Lymphangioma Successfully Treated with Lymphaticovenular Anastomosis
Takagi Katsunori*, Yamashita Shuji, Moriwaki Yuta, Morishita Yuya and Danno Kanako
Department of Plastic and Reconstructive Surgery, University of Tokyo Hospital, Japan
*Corresponding author: Takagi Katsunori, Department of Plastic and Reconstructive Surgery, University of Tokyo Hospital, 7-3-1 Hongo, Bunkyo-ku, Tokyo 113-8655, Japan
Published: 30 Nov, 2016
Cite this article as: Katsunori T, Shuji Y, Yuta M, Yuya M,
Kanako D. A Case of Lymphangioma
Successfully Treated with
Lymphaticovenular Anastomosis. Clin
Surg. 2016; 1: 1181.
Abstract
We present a case of a 40-year-old female with cystic lymphangioma in the left upper arm. The patient presented at our outpatient clinic with pain in the left upper arm. Ultrasonographic examination revealed a cystic mass in the left upper arm. Under ultrasonic guidance, the cyst was punctured using an 18G needle attached to a syringe; 30 ml of yellowish cloudy liquid was obtained. The patient was then administered antibiotic therapy (500 mg/day of oral LVFX for 5 days). There was no history of recent trauma or diabetes mellitus, thus the cause of lymphangitis remained unknown. Complete recovery from lymphangitis took 1 week. After recovery from lymphangitis, we performed magnetic resonance imaging (MRI) investigation of the left upper arm. MRI revealed a clustered cystic mass that was diagnosed as cystic lymphangioma. We performed triple lymphaticovenular anastomosis around the cubital fossa, and performed low pressure compression therapy for 6 months postoperatively. One week after discontinuation of compression therapy, MRI revealed that the lymphangioma had not re-expanded. Lymphangiomais commonly treated with surgical resection alone. This case revealed that lymphaticovenular anastomosis can be an effective treatment for cystic lymphangioma. Lymphaticovenular anastomosis may be preferable to surgical resection due to its relatively shorter incision length and therefore superior esthetic outcome.
Introduction
Surgical resection is the definitive treatment for lymphangioma. However, lymphedema can be effectively treated by lymphaticovenular anastomosis (LVA) [1]. LVA for lymphedema is done to allow the accumulated lymphatic fluid to bypass the area and drain via the venous system before the lymphatic fluid flows into the obstructive lesion. For cases involving large cystic lymphangioma, the incision line of radical resection will be extensive, resulting in an unfavorable esthetic outcome. Therefore, in the present case we performed the less invasive LVA procedure, which successfully treated lymphangioma.
Case Presentation
A 40-year-old female found a cystic mass in her left upper arm in her early twenties and did not
undergo any treatment. However, at 40 years of age, she developed lymphangitis and presented at
our hospital. Under ultrasonic guidance, an orthopedist punctured the cystic mass to drain the fluid.
The pain resolved immediately after drainage of the fluid. Antibiotic therapy (500 mg/day) of LVFX)
was administered orally for 5 days. After recovering from lymphangitis, the patient underwent
magnetic resonance imaging (MRI) investigation. The patient then presented at our department for
radical lymphangitis treatment.
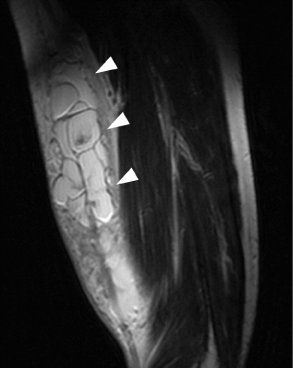
Preoperative T2-weighted MRI showed a cystic mass with a beaded appearance in the left upper
arm; the greatest width of the mass was more than 10 cm (Figure 1).
We performed LVA to drain the lymphatic fluid before it could accumulate in the cystic
lymphangioma. We created three LVAs around the left cubital fossa under local anesthesia
(Figure 2). The skin incisions were each 1.5–2.0 cm. The LVAs were created with 12-0 nylon under
microscopic view, and each involved four to six stitches. Operative time was 2 hours and 15 minutes.
The diameters of the lymphatic channels were 0.3–0.7 mm and lymphatic conditions were
almost normal (Figure 3); the lymphatics were transparent and there was no wall thickening or
fibrosis.
Postoperatively, the patient was discharged to her home with oral antibiotics (300 mg/day of
CFPN for 3 days) and one dose of NSAIDs (60 mg). The postoperative course was uncomplicated.
The patient wore low-pressure compression stockings around the left
upper arm for 6 months postoperatively; compression pressure was
less than 20 mmHg. One week after discontinuation of compression
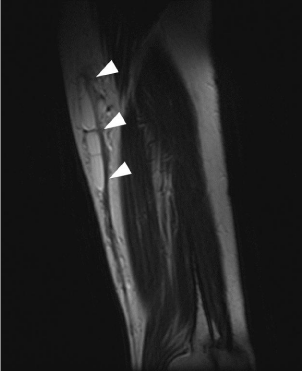
therapy, a follow-up MRI revealed no re-expansion of the cystic
lymphangioma (Figure 4).
Figure 1
Figure 1
Preoperative T2-weightedmagnetic resonance imaging showing a beaded cystic mass in the left upper arm. The white arrows indicate congregated cystic masses of 2–3 cm in the subcutaneous tissue of the upper arm.
Figure 2
Figure 2
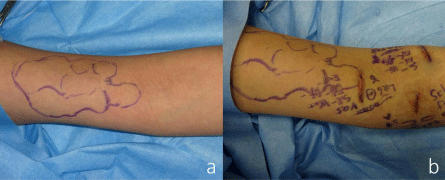
a. Preoperative view of the projected figure of the cystic lymphangioma outlined in crystal violet.
b. Postoperative photograph of the left upper arm after three lymphaticovenular anastomoses were performed.
Figure 3
Figure 3
Intraoperative photographs of the three lymphaticovenular nastomoses. Lymphatic diameter was 0.3–0.7 mm, and venous diameter was 0.5–0.6 mm. The white arrows indicate the lymphatic channels, and the black arrows indicate the veins
Figure 4
Figure 4
Postoperative magnetic resonance image taken 1 week after discontinuation of 6 months of compression therapy showing no re-expansion of cystic lymphangioma.
Discussion
Lymphangioma is a congenital malformation of the lymphatic
system that usually develops as a neck and/or head lesion in children
[2]. There are many reports on lymphangioma in children [3].
Although lymphangioma can reportedly be effectively treated by nonsurgical
methods such as sclerotherapy, radiofrequency ablation, and
laser therapy [4], only sclerotherapy is recommended for macrocystic
lymphangioma. However, sclerotherapy is associated with adverse
effects including soft tissue edema and skin necrosis.
There are only few reports on adult lymphangioma [5,6]. Adult
lymphangioma can be triggered by physical injury in rare cases
[7]. In the present case, the patient’s clinical history suggests that
asymptomatic cystic lymphangioma had probably been present since
childhood.
The patient developed lymphangitis of lymphangioma in her left
upper arm. Differential diagnoses for the cause of infection include
recent trauma such as an insect bite or an inveterate disease such as
diabetes mellitus. However, the patient had no history of insect bite
or diabetes mellitus, and there were no abnormal findings on physical
examination. The cause of lymphangitis remains unknown.
Nguyen et al. [8] reported on lymphangioma treated with
surgical resection in an elderly patient. The tumor in the present
case was more than 10 cm long, and so the skin incision required
for complete resection would have been very long; thus, we chose
LVA as a treatment considering her age and the esthetic outcome.
We conducted LVA in the cubital fossa just before the lymphatic
fluid flowed into the cystic lymphangioma. This aimed to allow the
lymphatic fluid to bypass the lymphatic channel before it flowed into
lymphangioma, thereby reducing lymphatic fluid flow into the cystic
lymphangioma. Although radical resection of cystic lymphangioma
requires general anesthesia, LVA can be performed under local
anesthesia as a day surgery, and the incision length is much shorter
than in radical resection. LVA is therefore a less invasive procedure
that also causes less burden to the medical economy. Sclerotherapy
with OK-432 injection would have been another option in the present
case, but we avoided sclerotherapy due to concerns about previous
lymphangitis [9].
We performed three LVAs in the present case. Lymphatic flow
was maintained, and it appeared that lymphatic conditions were
almost normal. Accordingly, normal functions of lymphatic channels
are expected postoperatively. The anastomoses all achieved excellent
performance outcome.
We continued compression therapy of the upper arm for 6
months postoperatively to maximize the drainage effect. Follow-up
MRI showed no re-expansion of the cystic lymphangioma, which
implies that the LVAs were functioning well in allowing the lymphatic
fluid to bypass the lymphangioma.
Long-term follow-up of this case is needed to confirm that the
LVAs remain effective without compression therapy; we plan to
continue careful monitoring with clinical examination every 3 weeks
in our outpatient clinic.
Conclusion
We used LVA to successfully treat a case of cystic lymphangioma. LVA was an effective treatment for lymphangioma.
References
- Koshima I, Inagawa K, Urushibara K, Moriguchi T. Supermicrosurgical lymphaticovenular anastomosis for the treatment of lymphedema in the upper extremities. J Reconstr Microsurg. 2000; 16: 437-442.
- Guarisco JL. Congenital head and neck masses in infants and children. Part II. Ear Nose Throat J. 1991; 70: 75-82.
- Balakrishnan K, Bauman N, Chun RH, Darrow DH, Grimmer JF, Perkins JA, et al. Standardized outcome and reporting measures in pediatric head and neck lymphatic malformations. Otolaryngol Head Neck Surg. 2015; 152: 948-953.
- Bagrodia N, Defnet AM, Kandel JJ. Management of lymphatic malformations in children. Curr Opin Pediatr. 2015; 27: 356-363.
- Yabe T, Takahashi M. Treatment of microcystic lymphangioma with OK- 432 after intralesional excision. J Dermatol. 2009; 36: 60-62.
- Bains SK, London NJ. Unilateral lower limb swelling secondary to cavernous lymphangioma. Eur J Vasc Endovasc Surg. 2008; 35: 373-374.
- Aneeshkumar MK, Kale S, Kabbani M, David VC. Cystic lymphangioma in adults: can trauma be the trigger? Eur Arch Otorhinolaryngol. 2005; 262: 335-337.
- Nguyen K, Karsif K, Lee S, Chorny K, Chen M. Lymphangioma in an elderly patient: an unusual cause of axillary mass. Breast. 2011; 17: 416- 417.
- Golinelli G, Toso A, Borello G, Aluffi P, Pia F. Percutaneous Sclerotherapy with OK-432 of a Cervicomediastinal Lymphangioma. Ann Thorac Surg. 2015; 100: 1879-1881.