Research Article
Selective Lobar Pulmonary Artery Thrombolysis: A Simple and Effective Technique in the Era of Rapidly Advancing Catheter Technology
Nassiri N1*, Samuel Kogan S1, Truong H1, Huntress L1, Vagaonescu T2, Nagarsheth K1, Shafritz R1 and Rahimi S1
1Department of Surgery, Rutgers Robert Wood Johnson Medical School, USA
2Department of Medicine, Rutgers Robert Wood Johnson Medical School, USA
*Corresponding author: Naiem Nassiri, Department of Surgery, Division of Vascular Surgery, Rutgers- Robert Wood Johnson Medical School, One Robert Wood Johnson Place MEB 541 New Brunswick, New Jersey 08901, USA
Published: 28 Nov, 2016
Cite this article as: Nassiri N, Samuel Kogan S, Truong
H, Huntress L, Vagaonescu T,
Nagarsheth K, et al. Selective Lobar
Pulmonary Artery Thrombolysis: A
Simple and Effective Technique in the
Era of Rapidly Advancing Catheter
Technology. Clin Surg. 2016; 1: 1177.
Abstract
Purpose: In the current era of rapidly advancing catheter technology for treatment of acute
pulmonary embolism, we present a simple yet effective technique for prompt resolution of right
heart strain in a deteriorating patient with a submassive pulmonary embolism (SMPE).
Methods: A 58-year old female with an acute SMPE (saddle and bilateral lobar PE with RV: LV
ratio= 1.44; pulmonary artery systolic pressure 70 mmHg; BNP 10,624 pg/mL; peak troponin t level
0.33 ng/mL) was recommended to undergo prolonged catheter-directed thrombolytic infusion
(EKOS Corp. Bothell, WA). Intraoperatively, she deteriorated hemodynamically with development
of massive PE (MPE). A decision was made to forego prolonged thrombolytic infusion. Selective
catheterization of main branch and bilateral lobar pulmonary arteries was performed with direct
transcatheter infusion of a total of 20 mg of alteplase over a 15-minute period.
Results: There was prompt improvement in mean PA pressures, right ventricular systolic function,
and pressor requirements. Post-operative CTA and TTE confirmed resolution of saddle embolus
and alleviation of right heart strain. Patient was discharged home on oral anticoagulation without
supplemental oxygen requirements.
Conclusion: Selective transcatheter pulmonary artery thrombolysis is a simple, safe, effective, and
inexpensive alternative to systemic thrombolysis and prolonged thrombolytic infusion particularly
in rapidly deteriorating patients with MPE and SMPE.
Keywords: Pulmonary artery thrombolysis; Pulmonary embolism; Catheter technology
Introduction
Though systemic thrombolysis (ST) remains the recommended first-line treatment for patients
with symptomatic pulmonary embolism (PE), catheter directed therapy (CDT) has evolved as an
attractive alternative that has at some institutions surpassed ST as first line therapeutic modality
[1-3]. Though many different techniques and technologies exist for CDT, the only option currently
with United States Food & Drug Administration (FDA) approval is ultrasound-facilitated
thrombolysis (EKOS Corp. Bothell, WA) – a technique that involves prolonged (typically 12-24
hours) infusion of thrombolytics [4,5]. Single-session CDT options that provide prompt resolution
of central pulmonary embolic burden without the need for prolonged infusion - such as rheolytic
and suction-based techniques – have either been largely abandoned (AngioJet, Boston Scientific.
Marlborough, MA) or remain under investigation and are currently being used off-label (Indigo
System, Penumbra Inc. Alameda, CA).
Here in, we report a simple and effective CDT technique in a rapidly deteriorating patient with
a symptomatic submassive pulmonary embolism (SMPE) that provided immediate intraoperative
hemodynamic improvement and obviated the need for prolonged thrombolytic infusion.
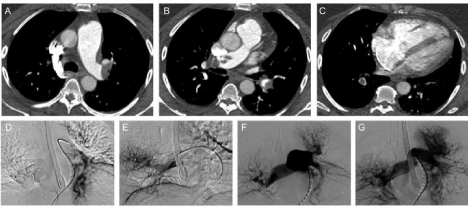
Figure 1
Figure 1
A, B) Pre-intervention CTA demonstrating saddle PE and extensive embolic burden extending into the lobar, segmental and subsegmental branches.
C) Right heart strain demonstrated on CTA by visualization of right ventricular dilatation and flattening of the interventricular septum. Note the segmental embolus adjacent to the arrow.
D, E) Bilateral selective lobar pulmonary artery catheterization with infusion of alteplase. F, G) Pulmonary angiogram immediately following
treatment showing considerable improvement in pulmonary perfusion
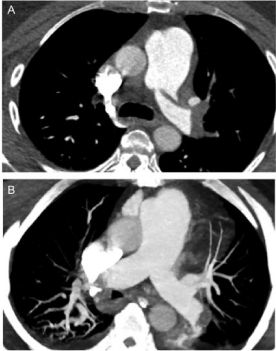
Figure 2
Figure 2
A) Pre-intervention CTA demonstrating saddle PE and extensive embolic burden extending into the lobar, segmental and subsegmental branches.
B) Post-intervention CTA demonstrating resolution of saddle embolus.
Methods
A 58 year-old female patient presented to the emergency department with acute onset of chest
pain and dyspnea on exertion. She was in sinus tachycardia to 120’s and hypoxemic requiring
supplemental oxygen by face mask without hemodynamic instability. Computed tomography
angiography (CTA) revealed a saddle PE with right heart strain (RV:
LV ratio =1.44) (Figure 1A-1C), which was confirmed by transthoracic
echocardiography (TTE) (PA systolic pressure 70 mmHg), serum
troponin levels (peak= 0.33 ng/mL), and BNP levels (10,624 pg.mL).
A SMPE was diagnosed and she was recommended to undergo CDT
per our institution protocol.
Intraoperative technique
At our institution, we routinely perform all PE interventions
in a hybrid operating room under general endotracheal anesthesia
(GETA). This provides a secure airway in case of worsening
hypoxemia caused by distal microembolization and foregoes need
for urgent intraoperative induction and intubation which can further
exacerbate heart strain and compromise hemodynamic status [1].
After successful induction of GETA, selective main branch
pulmonary artery catheterization was achieved via transfemoral
route. Main branch mean pulmonary artery pressure of 67 mmHg
was transduced. Pulmonary angiography confirmed presence of
saddle embolus with bilateral main branch and lobar involvement
(Figure 2A). Following diagnostic angiography and during catheter
exchange for delivery of EKOS infusion catheter, patient continued to
deteriorate clinically with worsening hypoxemia, and development of
cardiogenic shock requiring pressor support. There was no evidence
of perforation, hemothorax, hemopericardium, or tamponade on
fluoroscopy and transesophageal echocardiography (TEE).
Given the deteriorating clinical picture, an intraoperative
decision was made to forego prolonged thrombolytic infusion. Over
a stiff 0.035” platform, a 5-Fr Berenstein catheter (Angiodynamics.
Latham, NY) was introduced and selective catheterization of bilateral
main branch and lobar pulmonary arteries was performed with direct
transcatheter delivery of a total of 20 mg of alteplase over a 15-minute
period (Figure 1D and 1E).
Results
Completion angiogram showed significantly improved bilateral pulmonary artery arborization with reduction in centrally transduced mean PA pressures to 56 mmHg (Figure 1F and 1G) (Table 1). Postintervention CTA showed resolution of the saddle embolus (Figure 2B). TEE confirmed improvement in RV systolic function with reduction in tricuspid regurgitation. There was prompt hemodynamic improvement with gradually decreasing pressor requirements. All wires, catheters and sheaths were removed. Patient remained intubated overnight and observed in the intensive care unit (ICU) on systemic anticoagulation. She was weaned off pressors overnight and extubated successfully the on post-operative day (POD) 1. Postoperative TTE revealed considerable improvement in right heart strain (Table 1). She was discharged home on oral anticoagulation on POD 3 symptom-free without supplemental oxygen. She remained symptom-free with sustained alleviation of right heart strain and no evidence of chronic thromboembolic pulmonary hypertension (CTEPH) or cor pulmonale at 1, 3, and 6-month TTE.
Discussion
As the 3rd leading cause of cardiovascular death in the United States,
acute symptomatic pulmonary embolism remains a common and
lethal clinical entity with an annual incidence of greater than 500,000
and an estimated 30% mortality [6,7]. In the setting of substantial
central pulmonary embolic burden (i.e, saddle, main branch, or
multiple lobar PE), the presence of cardiogenic shock constitutes a
massive PE (MPE), while hemodynamic stability with concomitant
right heart strain (CTA and/or TTE evidence of right ventricular
dilatation [RV: LV> 0.9], McConnell’s sign, severe TR, severe
pulmonary hypertension [> 40 mmHg]) constitutes a submassive PE
(SMPE) [6,8]. Given significantly increased 30-day mortality and the
potential for development of chronic thromboembolic pulmonary
hypertension (CTEPH), MPE and SMPE warrant therapy beyond
anticoagulation alone [2-4].
Despite extensive experience across the literature with various
CDT for treatment of PE, systemic thrombolysis (ST) remains the
recommended first line treatment for treatment of MPE and SMPE
with CDT restricted to those patients with bleeding contraindications
to ST (~30% of all screened PE patients) or those who develop
bleeding complications during ST (~10% with < 5% major). We,
nevertheless, had previously reported our experience with successful
employment of an institution-wide algorithm using CDT as first-line
therapy for acute MPE and SMPE [1]. Given its high technical success
rate and absence of bleeding complications and in-hospital mortality,
CDT has remained our primary therapy of choice for treatment of
acute symptomatic PE with systemic thrombolysis reserved for rare
refractory cases or moribund patients too unstable for transfer to the
hybrid suite.
With the FDA approval of the EKOS ultrasound-facilitated
thrombolytic infusion technology, off-label use of rheolytic (AngioJet,
Boston Scientific) or suction-based (Indigo System, Penumbra Inc.
Alameda, CA) CDT remain limited and under investigation. EKOS
offers selective infusion of thrombolytic agents without mechanical
thromboembolectomy properties [4-5]. In other words, the marketed
ultrasound-facilitated thrombus fragmentation cannot be used
as sole therapy without chemical thrombolysis. Therefore, its use
requires prolonged infusion times with unknown complication
rates in patients who have bleeding contraindications. Given these
limitations, there does exist a clinical need for a CDT that foregoes
prolonged infusion, particularly in rapidly deteriorating patients with
limited cardiopulmonary reserve.
In the current case, the gradual progression to MPE during
pulmonary artery catheterization precluded a prolonged therapeutic
modality. Instead, alteplase was selectively delivered bilaterally
through a multipurpose diagnostic catheter. The 20 mg dose was
chosen arbitrarily given our previous experience with AngioJet Power
Pulse Spray technique, which typically employs 10 mg t-PA on each
side [1]. Prompt resolution of central embolic burden and right heart
strain was achieved and sustained up to 6-months post-operatively.
This was safe, effective, and inexpensive.
Given these outcomes, we have since modified our PE CDT
algorithm in favor of direct intraoperative delivery of t-PA either
selectively via a multi-purpose catheter or centrally through the same
multi-side-hole catheter placed within the right ventricular outflow
tract for diagnostic angiography. Further experience is indeed needed
before any substantial claims can be made regarding this technique.
Nevertheless, the technical and clinical success with sustained benefits
up to 6 months; and the reduction in cost and complexity of technique
are noteworthy and warrant further scrutiny in larger series.
Table 1
References
- Nassiri N, Jain A, McPhee D, Mina B, Rosen RJ, Giangola G, et al. Massive and submassive pulmonary embolism: experience with an algorithm for catheter-directed mechanical thrombectomy. Ann Vasc Surg. 2012; 26: 18-24.
- Jaff MR, McMurtry MS, Archer SL, Cushman M, Goldenberg N, Goldhaber SZ, et al. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011; 123: 1788-1830.
- Kearon C, Akl EA, Ornelas J, Blaivas A, Jimenez D, Bounameaux H, et al. Antithrombotic Therapy for VTE Disease: CHEST Guideline and Expert Panel Report. Chest. 2016; 149: 315-352.
- Piazza G, Hohlfelder B, Jaff MR, Ouriel K, Engelhardt TC, Sterling KM, et al. A Prospective, Single-Arm, Multicenter Trial of Ultrasound- Facilitated, Catheter-Directed, Low-Dose Fibrinolysis for Acute Massive and Submassive Pulmonary Embolism: The SEATTLE II Study. JACC Cardiovasc Interv. 2015; 8: 1382-1392.
- Kucher N, Boekstegers P, Muller OJ, Kupatt C, Beyer-Westendorf J, Heitzer T, et al. Randomized, controlled trial of ultrasound-assisted catheter-directed thrombolysis for acute intermediate-risk pulmonary embolism. Circulation. 2014; 129: 479-486.
- Lin PH, Chen H, Bechara CF, Kougias P. Endovascular interventions for acute pulmonary embolism. Perspect Vasc Surg Endovasc Ther. 2010; 22: 171-182.
- Kahn SR, Houweling AH, Granton J, Rudski L, Dennie C, Hirsch A. Longterm outcomes after pulmonary embolism: current knowledge and future research. Blood Coagul Fibrinolysis. 2014; 25: 407-415.
- Goldhaber SZ, Visani L, De Rosa M. Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet. 1999; 353: 1386-1389.