Case Report
Unravelling the Cocoon: A Case Report, Review of the Literature, and Journey in Rediscovering Congenital Peritoneal Encapsulation
Sabrina Ngaserin Ng Hui Na, Bettina Lieske and Choon-Seng Chong*
Department of Colorectal Surgery, National University Health System, Singapore
*Corresponding author: Choon-Seng Chong, Division of Colorectal Surgery, 1E Kent Ridge Road, National University Health System, #08-00 Tower Block, National University Hospital, Singapore 119228, Singapore
Published: 02 Nov, 2016
Cite this article as: Hui Na SNN, Lieske B, Chong C-S.
Unravelling the Cocoon: A Case Report,
Review of the Literature, and Journey
in Rediscovering Congenital Peritoneal
Encapsulation. Clin Surg. 2016; 1:
1169.
Abstract
Purpose: Congenital peritoneal encapsulation is an uncommon and poorly understood condition. It usually
presents as a diagnostic dilemma with eventual unexpected laparotomy findings and occasional
surgical misadventure. We describe a rare case of an elderly gentleman presenting with acute
intestinal obstruction, with typical radiographic findings raising pre-operative suspicion of an
abdominal cocoon. Surgery was mandatory due to non-resolving mechanical obstruction, and
concerns that there may have been segments of bowel in closed loops. Recognition of the condition
is critical in directing clinical decision-making and successful application of appropriate surgical
techniques, particularly for patients of “idiopathic” or congenital etiology.
Keywords: Abdominal cocoon; Congenital peritoneal encapsulation; Sclerosing encapsulating peritonitis; Intra peritoneal fibro sclerosis; Peritonitis chronica fibrosa incapsulata
Background
Definition itself has raised controversy in the rare condition ‘congenital peritoneal
encapsulation’. More plainly described as ‘congenital abdominal cocoon’, it has been argued
to be both interchangeable and distinct from terms like ‘sclerosing encapsulating peritonitis’,
‘intraperitoneal fibrosclerosis’, and ‘peritonitis chronica fibrosa incapsulata’, all of which encompass
a wider spectrum of conditions broadly classified as primary or secondary encapsulating peritonitis.
The congenital variety is usually asymptomatic and discovered serendipitously at unrelated
surgery or autopsy. However, intestinal obstruction may also result from encasement of variable
lengths of bowel by dense fibrocollagenous membranes, visually resembling a cocoon. The rare and
poorly understood condition usually presents itself as a diagnostic dilemma. More often seen in
younger females [1] and males [2], to the best of our knowledge, this is the first elderly male patient
from Singapore and the 7th reported in international literature.
Case Presentation
A 60 year old Chinese male presented with colicky central abdominal pain, vomiting and lack of
bowel movement for two days. Prior to this he did not have longstanding gastrointestinal symptoms.
He had no significant medical history, specifically autoimmune conditions, tuberculosis, connective
tissue disorders, beta-blocker usage, peritoneal catheters, prior abdominal infection, trauma, nor
surgery. On examination, his abdomen appeared distended and distorted by a mildly tender, diffuse
mass in the right lower quadrant.
Haematological investigations revealed mild leucocytosis. Plain radiographs showed a
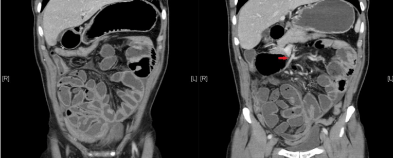
distended gastric bubble and multiple air-fluid levels. Contrast enhanced computed-tomography of
the abdomen and pelvis revealed gross dilatation of the stomach and duodenum, transitioning into
a conglomeration of segmentally dilated loops of jejunum and ileum. A thickened well enhancing
layer surrounded it, likely fibrotic peritoneum. There was subtle crowding of the mesenteric
vessels, but the superior mesenteric vessels remained in a normal anatomic configuration with no
malrotation (Figure 1).
Surgery was mandatory due to non-resolving mechanical obstruction and concerns that there
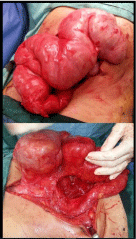
may have been segments of bowel in closed loops. Intra-operatively, the bowel was wrapped in a
near-opaque membranous sac in a concertina-like fashion from the duodenojejunal to ileocaecal
junction (Figure 2a). The entire cocoon was mobile and the sac itself appeared to be free from
the parietal peritoneum. Resolution of mechanical obstruction was
achieved by resection of the encasing membrane and adhesiolysis
with a combination of sharp and blunt dissection. This parted the
small bowel, while preserving its serosa. Apart from mild congestion,
the bowel was otherwise normal. The root of the mesentery was
assessed to be shortened (Figure 2b), contributing to the crowding
of the entire length of small bowel within the membranous sac. The
small bowel was decompressed before closure of the abdomen.
Our patient made an uneventful recovery after a week of postoperative
ileus, and was discharged well nine days later. He remains
healthy and asymptomatic after a year’s follow up.
Figure 1
Figure 1
Contrast enhanced computed-tomography of the abdomen and pelvis displaying the typical small bowel cocoon with crowding of the mesenteric vessels.
Figure 2
Discussion
First described by Cleland [3] and named "peritonitis chronica
fibrosa incapsulata" by Owtschinnikow [4], the idiopathic form of
encapsulating peritonitis was formally identified by Foo et al. [1].
Three decades on, the disease remains too uncommon to quantify its
incidence nor qualify its pathophysiology and natural history. Early
clinical features are non-specific, but established congenital peritoneal
encapsulation may present as either acute or chronic extramural cause
of intestinal obstruction. Patients may also exhibit variable degrees of
weight loss and occasionally there may be a ‘misleading’ abdominal
mass. Complications include malnutrition, ischemia, perforation, or
enterocutaneous fistulae.
To date, there exist questions that possess unsatisfactory answers.
What is the aetiology and pathophysiology?
Abdominal cocoon may classified as primary or secondary.
The development of primary sclerosing encapsulating peritonitis
remains ‘idiopathic’ to date. It was first documented in adolescent
females within 2 years of menarche, originating from warm tropical
and subtropical climates. Hence authors had initially suggested that
this could be related to retrograde menstruation or gynaecological
infection from transvaginal peritonitis 1. Theories behind such a
disease process have little supportive evidence. With more reports of
male patients, some postulate instead that there may be congenital
defective return of the embryonic midgut loop to the intraperitoneal
abdominal cavity [5].
The development of secondary sclerosing encapsulating peritonitis
may be better explained. Risk factors include chronic ambulatory
peritoneal dialysis [6], peritonitis [7], previous abdominal surgery [8],
and prolonged use of beta blockers [9]. There also exist case reports
of disease related to benign gynaecological conditions, ovarian
malignancy, gastrointestinal malignancy, patients presenting along
with abdominal trauma, peritoneal shunts (ventriculoperitoneal and
peritoneal-venous), liver cirrhosis, systemic lupus erythematosus,
tuberculosis, familial Mediterranean fever, ascaris lumbricoides, and
one reporting consumption of cereals infected with fungus containing
biologically active amines. The common underlying disease process
points towards low-grade intra-abdominal sepsis, resulting in sclerosis
and fibrosis. This is consistent with reports of success in treating early
stages of the disease with immunosuppressants, corticosteroids and
tamoxifen.
Many terms have been utilized to describe the same disease.
Perhaps the clinical entity would benefit from realignment of
terminology with clear stratification by proposed aetiology.
What factors will allow us to consider a congenital cause?
The most peculiar anatomical clue is that the prime majority
of ‘idiopathic’ cases only affect the small bowel. Only isolated cases
describe involvement of the small intestine plus other organs (e.g.
appendix, ascending colon, ovaries). This may be consistent with a
developmental defect resulting in the small intestine being covered by
the original dorsal mesentery, which forms the accessory peritoneal
sac.
Are there typical and distinctive radiological features that
can confirm the diagnosis preoperatively?
Anecdotally, the use of radiological imaging modalities has
been the single clearest aid in attaining non-invasive pre-operative
diagnosis. Sonography may reveal small bowel loops arranged in
concertina shape with a narrow posterior base, in a cauliflower
appearance, only if there were concurrent ascites [10]. Typical
features of dilated but congregated bowel loops encased in a thick
membrane of soft tissue density may be evident on computedtomography
scan [10]. Barium follow-through may also show similar
findings along with delayed transit of contrast media. There have
also been recent developments in using dynamic cinematographic
magnetic resonance, coupled with advanced image analysis. Current
studies involve patients with suspected PD-related encapsulating
peritoneal sclerosis, where areas of altered movement might suggest
frank adhesions [11] although diagnosis may be suspected with the
assistance of such radiological findings, true pre-operative diagnosis
still requires a high index of suspicion in the informed clinician.
Surgery is indicated in non-resolving mechanical obstruction,
when there are concerns of bowel viability or complications such as
perforation. Incision or excision of the encasing membrane is the
ideal management option. When indicated, bowel resection or stoma
creation should be considered. More extensive operations, including
unnecessary bowel resections and bypass procedures, were performed
when the condition was not recognised [1]. Only a small number of
cases utilizing laparoscopic approach have been reported [12]. Some
authors suggested prophylactic appendicectomy to avoid difficult
appendicectomy should the patients develop appendicitis later [1].
Histologically, the membrane typically exhibits proliferation of
fibro-connective tissue and inflammatory infiltrates, and no foreign
body granulomas, giant cells, or birefringent material [13].
Long term prognosis appears to be excellent. There have been no
reports of recurrence to date.
Conclusion
Recognition of congenital peritoneal encapsulation is fundamental in insightful management to avoid surgical misadventure which may result in unwarranted enterotomies, bowel resection or stoma creation.
Chief Educational Objective
Congenital peritoneal encapsulation is an uncommon and poorly
understood condition. This article endeavors to revisit the topic and
provide clarity to the issues surrounding the abdominal cocoon.
- Congenital peritoneal encapsulation has been described to present as all of the following, except:
- Intestinal obstruction
- Abdominal mass
- Loss of appetite and loss of weight
- Enterocutaneous fistula
- Tenesmus
- Described risk factors for secondary sclerosing encapsulating peritonitis include all of the following, except:
- Beta blockers
- Peritoneal dialysis
- Radiotherapy
- Tuberculosis
- Previous abdominal surgery
- Typical radiological features of congenital peritoneal encapsulation include:
- Dilated bowel with a transition point
- Football sign
- Silver’s sign
- Congregated bowel loops encased in a thick membrane of soft tissue density
- Rigler’s sign
Correct answer: e. Tenesmus.
Explanation: Congenital peritoneal encapsulation results in the encasement of variable lengths of bowel by dense fibrocollagenous membranes, which may be asymptomatic, result in vague symptoms such as early satiety, loss of appetite, loss of weight, intestinal obstruction, an abdominal mass, or complications such as malnutrition, ischemia, perforation, or enterocutaneous fistulae. Tenesmus has not been described.
Correct answer: c. Radiotherapy.
Explanation: While there have been various associated risk factors for secondary sclerosing encapsulating peritonitis, radiotherapy has not been described to be amongst them.
Correct answer: d. Congregated bowel loops encased in a thick membrane of soft tissue density.
Explanation: Typical features of dilated but congregated bowel loops encased in a thick membrane of soft tissue density may be evident on computed-tomography scan.
References
- Foo KT, Ng KC, Rauff A, Foong WC, Sinniah R. Unusual small intestinal obstruction in adolescent girls: the abdominal cocoon. Br J Surg. 1978; 65: 427-430.
- Gupta RK, Chandra AS, Bajracharya A, Sah PL. Idiopathic sclerosing encapsulating peritonitis in an adult male with intermittent subacute bowel obstruction, preoperative multidetector-row CT (MDCT) diagnosis. BMJ Case Rep. 2011; 2011.
- Cleland J. On an Abnormal Arrangement of the Peritoneum, with Remarks on the Development of the Mesocolon. J Anat Physiol. 1868; 2: 201-206.
- Xu P, Chen LH, Li YM. Idiopathic sclerosing encapsulating peritonitis (or abdominal cocoon): a report of 5 cases. World J Gastroenterol. 2007; 13: 3649-3651.
- Costantinides F, Di Nunno N, Bernasconi P, Jonjic N, Melato M. A new type of peritoneal encapsulation of the small bowel. Am J Surg Pathol. 1998; 22: 1297-1298.
- Kim BS, Choi HY, Ryu DR, Yoo TH, Park HC, Kang SW, et al. Clinical characteristics of dialysis related sclerosing encapsulating peritonitis: multi-center experience in Korea. Yonsei Med J. 2005; 46: 104-111.
- M U, Kumar V, R RA, Kamath S. Perforated GIST in Jejunum - A Rare Cause of Abdominal Cocoon. J Clin Diagn Res. 2014; 8: 132-133.
- Caldwell J, Dyer A. Sclerosing encapsulating peritonitis (cocoon bowel) presenting after laparotomy for splenic abscess. J Radiol Case Rep. 2013; 7: 17-23.
- Kalra S, Atia A, McKinney J, Borthwick TR, Smalligan RD. Sclerosing encapsulating peritonitis associated with propranolol usage: a case report and review of the literature. J Dig Dis. 2009; 10: 332-335.
- Kaur R, Chauhan D, Dalal U, Khurana U. Abdominal cocoon with small bowel obstruction: two case reports. Abdom Imaging. 2012; 37: 275-278.
- Wright B, Summers A, Fenner J, Gillott R, Hutchinson CE, Spencer PA, et al. Initial observations using a novel "cine" magnetic resonance imaging technique to detect changes in abdominal motion caused by encapsulating peritoneal sclerosis. Perit Dial Int. 2011; 31: 287-290.
- Qasaimeh GR, Amarin Z, Rawshdeh BN, El-Radaideh KM. Laparoscopic diagnosis and management of an abdominal cocoon: a case report and literature review. Surg Laparosc Endosc Percutan Tech. 2010; 20: e169- 171.
- Clatworthy MR, Williams P, Watson CJ, Jamieson NV. The calcified abdominal cocoon Lancet. 2008; 371: 1452.