Research Article
Narrow Band Imaging Endoscopy: Novel Diagnostic Method in the Hypertrophy of Inferior Turbinates
Stoelzel K*, Dommerich S, Bandelier M, Olze H and Szczepek AJ
Department of Otorhinolaryngology, Head and Neck Surgery, Charité - Medical University, Germany
*Corresponding author: Katharina Stölzel, Department of Otorhinolaryngology, Head and Neck Surgery, Charité - Medical University, Campus Charité Mitte, Chariteplatz 1, 10117 Berlin, Germany
Published: 26 Oct, 2016
Cite this article as: Stoelzel K, Dommerich S, Bandelier
M, Olze H, Szczepek AJ. Narrow Band
Imaging Endoscopy: Novel Diagnostic
Method in the Hypertrophy of Inferior
Turbinates. Clin Surg. 2016; 1: 1163.
Abstract
Objectives: The narrow band imaging endoscopy (NBI) is an imaging method used by otolaryngologists for the examination of oral cavity, pharynx and larynx. Our present study was designed to determine the effectiveness of NBI in the examination of the inferior turbinate hypertrophy status.
Study Design: Individual cohort study.
Methods: A hundred and nine patients with enlarged inferior nasal conchae were enrolled. All subjects were examined prior to surgical intervention of conchae nasals inferior and were followed up to 6 months after the intervention. During the appointments, nasal endoscopy with white light, NBI endoscopy and the anterior rhinomanometry were performed. In addition, all subjects were asked about subjective nasal obstruction.
Results: Following surgery, the number of blood vessels in the nasal concha inferior was found to be reduced in all cases studied. The vascular imaging with NBI endoscopy produced significantly better results as the white light endoscopy. The decrease in the objective concha component correlated stronger with the NBI endoscopy than with white light endoscopy; however, the difference has not reached statistical significance.
Conclusion: NBI endoscopy allows fine tuning of the endoscopy scores and can therefore
contribute to a standardized evaluation of the nasal inferior conchae by improving the diagnosis
and monitoring of nasal mucosal vascular lesions.
Keywords: Narrow band imaging endoscopy; Concha nasalis inferior; Blood vessels; Nasal obstruction
Introduction
Nasal obstruction is one of the most common symptoms with which the ORL specialists are
confronted in clinical practice. Common reason of nasal obstruction is the enlargement of inferior
nasal concha (inferior turbinates). Conditions that contribute to the enlargement include allergic
and non-allergic rhinitis, chronic hypertrophic rhinitis and a compensatory enlargement being a
consequence of septal deviation [1,2]. Chronic inferior turbinate enlargement is not associated with
cellular hypertrophy but rather with cellular hyperplasia, tissue oedema and vascular congestion
[3]. One of the important hallmarks of turbinate enlargement is the increased number of blood
vessels within the nasal mucosal tissues and dilatation of venous sinusoids within lamina propria.
The number of laminar blood vessels correlates with positive therapy outcome, as it was reduced one
month after electrocautery [4]. Enlarged sinusoids are beside fibrosis and inflammation the reason
for an enlargement of inferior turbinates [5].
Clinical examination of nasal obstruction is performed with a use of anterior rhinoscopy and
nasal endoscopy. Despite the attempts of Camacho et al. [6] to introduce the size of nasal concha as
a marker of disease classification, no standard scoring system of the anterior rhinoscopy is so far in
use. The endoscopic description of the inferior nasal concha, which was introduced by Meltzer et
al. [7-8] as a so-called "endoscopy score", is based on the swelling status and on the color of affected
tissue. Unfortunately, no further observations can be made with the use of white-light endoscopy.
The narrow band imaging (NBI) is an optical technology, which uses two specific wavelengths
in order to improve the surface and the vascular representation of mucosa. NBI is based on the
fact, that the penetration depth of light is wavelength-dependent and that specific spectral regions
are particularly well absorbed by hemoglobin. The favored penetration wavelengths lay between
440 and 460 nm (blue light), where the penetration of light is low
and therefore the mucosal surface and feeding capillaries are clearly
visible. The specific spectral region within 540-560 nm (green) is
absorbed by hemoglobin, therefore the submucosal blood vessels can
be imaged in an optimal way [9-10].
In the recent years, the NBI has been used for imaging of various
internal organs [11-12]. In addition to the tumor evaluation and
the determination of tumor-dependent neovascularization in the
gastrointestinal tract, lungs, bladder, and many other organs, NBI has
been also applied in ORL for the early detection of larynx, oropharynxand
hypopharynx- carcinomas [13-16]. Only few records about the
use of NBI in the nasal cavity endoscopy have been published to date:
one about the examination of hereditary hemorrhagic telangiectasia
and another one about the examination of granulomatosis with
polyangiitis [17-18]. However, to our best knowledge, no studies
using NBI-examination of blood vessels in the enlarged inferior nasal
concha or in the associated nasal obstruction were published so far.
Our study evaluated the application of NBI nasal endoscopy
for the diagnosis and monitoring of enlarged inferior nasal concha.
Moreover, we wanted to determine if the imaging results obtained
with NBI nasal endoscopy correlate with the nasal breathing.
We studied patients with nasal obstruction who were scheduled
for a surgery of enlarged inferior nasal concha. Three surgical
techniques were included in the study: lateralized submucosal
turbinectomy electrocautery and laser cautery.
Figure 1
Figure 2
Figure 2
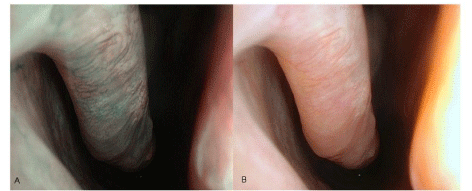
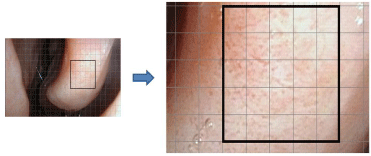
Exemplary image obtained with white light endoscopy (A). Magnified field used for counting of vessels within the grid (n=80) (B).
Figure 3
Figure 3
Exemplary image obtained with NBI endoscopy (A). Magnified field used for counting of vessels within the grid (n=145) (B).
Materials and Methods
Patients
This randomized, three-arm prospective study was approved by a
local Ethics Committee (permit number EA1/188/13). One hundred
and nine patients were initially enrolled in the study. The inclusion
criteria were age between 18 and 70 years, both genders and a positive
diagnosis of inferior nasal concha enlargement. The exclusion
criteria included chronic rhinosinusitis, nasal surgery prior to this
study, diabetes, nasal polyps, autoimmune diseases and coagulation
disorders. All patients underwent surgical reduction of inferior nasal
concha and the allocation to the surgical technique was randomized.
For 75 subjects, full evaluation over six months (four appointments)
could be done. Of these 75 patients, 19 were treated with lateralized
submucosal turbinectomy, 26 with electrocautery and 30 with laser
cautery.
Methods
During the first appointment prior to surgery, medical history
was collected and the nasal endoscopy with flexible endoscope
(Olympus ENF-P4 fiber scope) equipped with the EVIS EXERA III
Video System Center CV-190 was performed with a viewing angle
of 110°. The white light endoscopy was directly followed by the NBI
endoscopy with the same distance to the concha (Figure 1A, B). For
the statistical analyses, a specific grid was placed in the endoscopic
image, which was of the same size in every shot. The blood vessels were
counted in the grid of each picture of white light endoscopy (Figure 2)
and then compared with the grid count from NBI endoscopy (Figure
3). In order to objectively determine the number of blood vessels,
each a congruent cut out of 6.5 x 6.5 cm was selected. At a resolution
of 96 x 96 DPI, the vessel Number of inferior turbinate per visual field
was determined using a Neubauer chamber.
As an objective comparison parameter we used the results of
anterior rhinomanometry (Rhino 4000, Homoth Medizinelektronik
GmbH & Co KG, Hamburg, Germany). The total inspiration in
milliliters per second was determined before and after decongestion
of the nasal mucosa. The comparison of various parameters has been
performed for each subject over time as well as between the various
surgical procedures.
Following the rhinomanometry, the patients were asked to
report their subjective nasal obstruction. Scoring ranging from 0 (no
symptoms) to 4 (very severe symptoms) was applied, according to
Likert scale. During follow up, the same question and scales were
used during the appointments one month, three months and six
months after surgical reduction of the inferior nasal concha.
Statistical analysis
The statistical analysis was performed with the IBM SPSS Statistics
22 for Windows. For the statistical test procedures a significance level
of 0.05 was used (alpha = 5%). The treatment groups were also tested
for differences in the time course. Because there is an incomplete
repeated measures design (not all patients showed up for all 3
postoperative time points; not all questions were answered), the twofactor
experimental design was checked for statistical significance
using a between-subjects factor and a within-subjects factor according
to the Generalized Estimating Equations (GEE) methodology [19-20].
Results
Comparison of the white-light endoscopy with the NBI endoscopy results
The visibility of blood vessels was significant better when using
NBI, as compared to the white light endoscopy (p=0.00; t-test).
Significantly more blood vessels could be identified by NBI (Table 1).
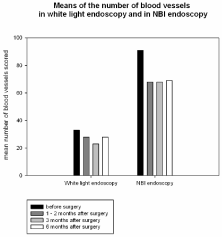
Changes observed using white light endoscopy during the consecutive appointments
During the first appointment (prior to surgery), the majority
of blood vessels were localized on the surface of the inferior nasal
concha. One to two months following surgery, only few blood vessels
could be visualized using white light endoscopy, mainly due to the
nasal secretion. Six months after the surgery, the visualization of the
vessels improved and the number of visible blood vessels was lower
than that on the first appointment (Figure 4).
Changes observed using NBI endoscopy during the consecutive appointments
During the first appointment, most blood vessels were identified
on the surface of the mucosa and submucosa in the inferior nasal
conchae. Despite the nasal secretion, the blood vessels could
be clearly identified with NBI after surgery. Their number was
significantly lower than before surgery and nearly constant during all
appointments (Figure 4).
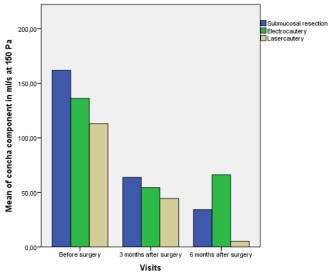
Post-operative, temporal changes in the objective nasal obstruction – individual and between-group comparison
The objective changes in the nasal obstruction were measured
using differences of mean values determined by the total inspiration
(in ml/s at a pressure of 150 Pa) with anterior rhinomanometry
before and after decongestion with a topical nasal decongestant
xylometazoline. So we measured the concha component. Six months
after surgery, the concha components have significantly decreased in
all three groups as compared with the preoperative findings (Figure 5).
Post-operative, temporal changes in the subjective nasal obstruction
In all three groups studied (lateralized submucosal turbinectomy,
electrocautery and laser acutery), the patients reported a significant
improvement of symptoms in terms of nasal obstruction following
surgery. However, two and three months following surgery there was
a significant difference between the groups in favor of laser acutery
(p=0.032). Six months after surgery, the differences between the
groups were no longer significant.
Figure 4
Figure 5
Figure 5
Mean mucous membrane component as a difference of total inspiration in ml/s at 150 Pa before and after decongestion with a topical nasal decongestant xylometazoline.
Table 1
Discussion
In the neoplastic tissues, typical composition of the mucosal
and submucosal layers is altered in the nasal cavity and especially
in inferior turbinates’. Moreover, the presence of capillaries in
the lamina propria and venous sinusoids of lamina submucosa
significantly contribute to the tissue enlargement [3]. Berger et al.
[5] has suggested that the increase in the number of capillaries may
be responsible for the enlargement of venous sinusoids whereas
Talaat et al. [4] hypothesized that the number of small vessels in
the mucosal and submucosal tissues causatively associates with the
tissue enlargement. Our evidence supports these hypotheses, because
we have demonstrated significant difference in the number of blood
vessels between the white light- and NBI endoscopies.
The narrow band imaging was first described in 2004 by Yoshidi
et al. [21] as a diagnostic measure of oesophageal lesions. Ever
since, narrow band imaging has been used in many studies as an
indispensable part of gastrointestinal tract endoscopy [12] and the
lung endoscopy [22]. Also in the otorhinolaryngology, the narrow
band imaging found its application in the early diagnostic of the oral
cavity- and oropharynx carcinomas [23,24]. In the present study,
we have determined the usefulness of NBI for the examination of
nasal mucosa in cases of nasal obstruction. To exclude the effect of
septoplasty, we measured the mean value of conchae components and
found that it significantly decreases after surgery in all intervention
groups. Likewise, the number of blood vessels scored in the NBI
endoscopy has decreased over the time in all intervention groups.
NBI endoscopy uses filtered white light to imagine blood vessels in
the mucosal and submucosal tissues [3-5]. However, to date, the
non-invasive diagnostic methods used to determine pathologies in
inferior nasal concha was mainly done using non-filtered, whitelight
endoscopy and so called “Meltzer score” or “endoscopy score”
[8]. The use of NBI in determination of chronic enlargement of
inferior turbinates has never been described before. In our study,
the diagnostic use of standard white light endoscopy was extended
by means of complementing measures, which can easily be added to
conventional endoscopy. In addition to the color and the grade of
mucosal tissue swelling obtained with the white-light endoscopy, the
NBI endoscopy offers important information about the number of
blood vessels in inferior nasal concha. This information can be used
to estimate vascularization index in nasal tissues, which cannot be
obtained with the white-light endoscopy. Importantly, the results
of NBI endoscopy proved to be unaffected to the presence of nasal
secretion, which was not the case for white light endoscopy. In the
present study, we demonstrated that the visualization of blood vessels
during first few months after surgical intervention is compromised if
not impossible when using the conventional, white light endoscopy.
At the same time, the blood vessels could be easily monitored with
the NBI endoscopy.
Our study not only demonstrated feasibility of using NBI
endoscopy for the diagnosis and monitoring of patients with inferior
nasal concha enlargement, but also detected a trend in the outcome
of various surgical techniques used for treatment of this common
condition. This trend points at beneficial use of the laser cautery in
terms of objective and subjective measurements. However, larger
study sample representing all three groups are required to come to
concise clinical conclusions.
Conclusion
In the present work, we demonstrate the usefulness of NBI endoscopy in the diagnosis and post-surgical monitoring of inferior nasal concha enlargement. The quick change between white light and NBI during the nasal endoscopy is useful and time-saving. Extended specification of endoscopy scores and the ability to determine typical morphology of nasal mucosa by NBI endoscopy could improve the diagnosis and treatment of nasal mucosa pathologies.
References
- Baumann I, Baumann H. A new classification of septal deviations. Rhinology. 2007; 45: 220-223.
- Larrabee YC, Kacker A. Which inferior turbinate reduction technique best decreases nasal obstruction? Laryngoscope. 2014; 124: 814-815.
- Farmer SEJ, Eccles R. Chronic inferior turbinate enlargement and the implications for surgical intervention. Rhinology. 2006; 44: 234-238.
- Talaat M, el-Sabawy E, Baky FA, Raheem AA. Submucous diathermy of the inferior turbinates in chronic hypertrophic rhinitis. J Laryngol Otol. 1987; 101: 452-460.
- Berger G, Gass S, Ophir D. The histopathology of the hypertrophic inferior turbinate. Arch Otolaryngol Head Neck Surg. 2006; 132: 588-594.
- Camacho M, Zaghi S, Certal V, Abdullatif J, Means C, Acevedo J, et al. Inferior turbinate classification system, grades 1 to 4: development and validation study. Laryngoscope. 2015; 125: 296-302.
- Hirunwiwatkul P, Udomchotphruet P. Efficacy study of nasal irrigation after radiofrequency tissue volume reduction for inferior turbinate hypertrophy: An equivalence randomized controlled trial. American journal of rhinology & allergy. 2012; 26: 497-503.
- Meltzer EO, Hamilos DL, Hadley JA, Lanza DC, Marple BF, Nicklas RA, et al. Rhinosinusitis: Developing guidance for clinical trials. Otolaryngology- -head and neck surgery. 2006; 135: S31-S80.
- Gono K, Obi T, Yamaguchi M, Ohyama M, Machida H, Sano Y, et al. Appearance of enhanced tissue features in narrow-band endoscopic imaging. J Biomed Opt. 2004; 9: 568-577.
- Uedo N, Ishihara R, Iishi H, Yamamoto S, Yamamoto S, Yamada T, et al. A new method of diagnosing gastric intestinal metaplasia: narrow-band imaging with magnifying endoscopy. Endoscopy. 2006; 38: 819-824.
- Larghi A, Lecca PG, Costamagna G. High-resolution narrow band imaging endoscopy. Gut. 2008; 57: 976-986.
- Louie JS, Richards-Kortum R, Anandasabapathy S. Applications and Advancements in the Use of High-resolution Microendoscopy for Detection of Gastrointestinal Neoplasia. Clin Gastroenterol Hepatol. 2014; 12: 1789-1792.
- Goda K, Dobashi A, Tajiri H. Perspectives on narrow-band imaging endoscopy for superficial squamous neoplasms of the orohypopharynx and esophagus. Dig Endosc. 2014; 26: 1-11.
- Irjala H, Matar N, Remacle M, Georges L. Pharyngo-laryngeal examination with the narrow band imaging technology: early experience. European archives of oto-rhino-laryngology. 2011; 268: 801-806.
- Kraft M, Fostiropoulos K, Gurtler N, Arnoux A, Davaris N, Arens C. Value of narrow band imaging in the early diagnosis of laryngeal cancer. Head Neck. 2014; 38: 15-20.
- Piazza C, Dessouky O, Peretti G, Cocco D, De Benedetto L, Nicolai P. Narrow-band imaging: a new tool for evaluation of head and neck squamous cell carcinomas. Review of the literature. Acta Otorhinolaryngol Ital. 2008; 28: 49-54.
- Pagella F, Pusateri A, Chu F, Caputo M, Danesino C, Matti E. Narrowband imaging in the endoscopic evaluation of hereditary hemorrhagic telangiectasia patients. Laryngoscope. 2013; 123: 2967-2968.
- Trimarchi M, Bozzolo E, Pilolli F, Bertazzoni G, Campochiaro C, Sabbadini MG, et al. Nasal mucosa narrow band imaging in granulomatosis with polyangiitis (Wegener granulomatosis): A preliminary study. American journal of rhinology & allergy. 2015; 29: 170-174.
- Dobson AJ. An Introduction to Generalized Linear Models. 2002.
- Hardin JW. Generalized Estimating Equations. 2001..
- Yoshida T, Inoue H, Usui S, Satodate H, Fukami N, Kudo SE. Narrow-band imaging system with magnifying endoscopy for superficial esophageal lesions. Gastrointest Endosc. 2004; 59: 288-295.
- Hanna WC, Yasufuku K. Bronchoscopic staging of lung cancer. Ther Adv Respir Dis. 2013; 7: 111-118.
- Vu AN, Farah CS. Efficacy of narrow band imaging for detection and surveillance of potentially malignant and malignant lesions in the oral cavity and oropharynx: a systematic review. Oral Oncol. 2014; 50: 413-420.
- Vergez S, Moriniere S, Dubrulle F, Salaun PY, De Monès E, Bertolus C, et al. Initial staging of squamous cell carcinoma of the oral cavity, larynx and pharynx (excluding nasopharynx). Part I: Locoregional extension assessment: 2012 SFORL guidelines. Eur Ann Otorhinolaryngol Head Neck Dis. 2013; 130: 39-45.