Case Report
Combined Transfemoral Aortic Valve Replacement and Endovascular Repair of Abdominal Aortic Aneurysm in Nonagenarians is Feasible
Faisal Aziz1*, Mark Kozak2 and Walter E Pae3
1Division of Vascular Surgery, Pennsylvania State University College of Medicine, USA
2Division of Interventional Cardiology, Pennsylvania State University College of Medicine, USA
3Division of Cardiothoracic Surgery, Pennsylvania State University College of Medicine, USA
*Corresponding author: Faisal Aziz, Division of Vascular Surgery, Pennsylvania State University College of Medicine, 500 University Drive, Mail Code H053, Room C4632 Hershey, PA 17033, USA
Published: 13 Oct, 2016
Cite this article as: Aziz F, Kozak M, Pae WE. Combined
Transfemoral Aortic Valve Replacement
and Endovascular Repair of Abdominal
Aortic Aneurysm in Nonagenarians is
Feasible. Clin Surg. 2016; 1: 1153.
Abstract
Geriatric population is the fastest growing age group in the United States. Traditionally, high risk
procedures are not offered to elderly, high risk patients. With increasing number of aging patients
and evolution of endovascular technology, more and more minimally invasive procedures are being
offered to geriatric patients. Transfemoral aortic valve repair and endovascular repair of abdominal
aortic aneurysms have been successfully performed in patients above 90 years of age. We describe
a unique case of a nonagenarian, who presented with severe aortic stenosis and a large, infrarenal
abdominal aortic aneurysm. She underwent a successful combined endovascular repair of both
these pathologies at the same time. This case highlights the feasibility of combined procedures in
select group of nonagenarians.
Keywords: Nonagenarian; Abdominal aortic aneurysm; Aortic stenosis; Endovascular repair of abdominal aortic aneurysm; Transfemoral aortic valve replacement
Introduction
Improvements in healthcare quality have allowed the geriatric population to live and remain independent longer [1]. Population demographics are changing, and it is expected that nonagenarian population will quadruple by year 2050 [2]. Calcific aortic stenosis is the most common structural cardiac disease in the elderly population, and medical management of severe aortic stenosis is associated with poor outcomes as compared to surgical treatment [3]. The incidence of Abdominal Aortic Aneurysm (AAA) increases with increasing age and its rupture is associated with significantly high morbidity and mortality rates. Both of these disease entities can be life threatening. The majority of patients in the tenth decade of life are so frail that physicians are hesitant to offer them surgical therapy. We present a case of a nonagenarian, who presented with worsening of severe aortic stenosis and an enlarging AAA. We treated her with simultaneous Transfemoral Aortic Valve Replacement (TAVR) and Endovascular Repair of Abdominal Aortic Aneursym (EVAR).
Case Presentation
A 94-year-old female with a past medical history significant for aortic stenosis and abdominal
aortic aneurysm presented with worsening shortness of breath and fatigue. Previously, she was
asymptomatic and was deemed not a surgical candidate for treatment of either of these conditions
due to her advanced age. Now for the past year, she was becoming more and more short of breath
upon exertion and had four hospitalizations for the treatment of dyspnea. On physical examination,
she was awake and alert and hemodynamically stable. Her jugular venous distension was 8cm
above the sternal notch in the sitting position. On auscultation of her chest, she had a loud systolic
murmur in the aortic area. Her abdominal examination revealed a large, pulsatile aortic aneurysm.
Her echocardiogram showed an ejection fraction of 55% with peak aortic gradient of 70 mmHg with
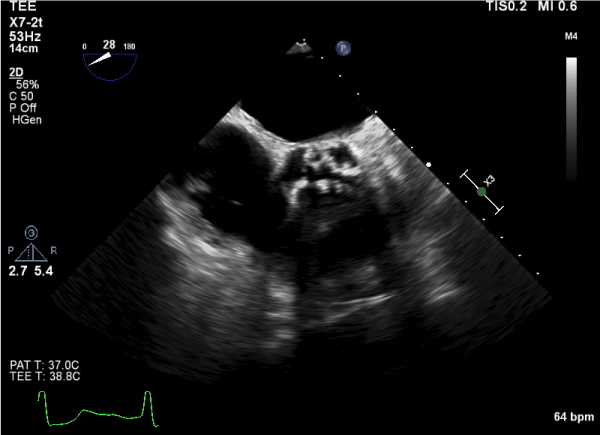
a mean gradient of 40 mmHg. Her aortic annulus was about 2cm (Figure 1 and 2). Her abdominal
CT scan showed a 7.8 cm infrarenal abdominal aortic aneurysm (Figure 3), which was a dramatic
increase, as compared to diameter of 6 cm the year before. The patient and her family had multiple
long discussions with her physicians. Options for management included conservative management
and minimally invasive surgical treatment. After long deliberations regarding risks, benefits and
alternatives, the patient and her family opted for surgical management. From a technical standpoint,
she was a candidate for TAVR and EVAR. The next decision point
was whether to perform these procedures simultaneously, in the same
setting, or to do them in a staged fashion. Each approach has its own
pros and cons. It was decided to perform both procedures in the same
setting.
The patient was brought to the operating room and placed
in supine position. Right internal jugular central venous line was
inserted and right radial arterial line was placed. Due to patient’s
advanced age, we opted to choose Monitored Care Anesthesia
(MAC) with judicious use of intravenous fentanyl and versed. A
transverse cut-down was performed on the left groin. The skin was
incised with scalpel. Dissection was continued until the femoral
sheath was reached, which was incised longitudinally to expose the
common femoral artery. Now, under ultrasound guidance, a micro
puncture needle was inserted into patient’s right common femoral
artery and using Seldinger’s technique and a wire was placed followed
by placement of 5 French sheaths. After angiographic confirmation
that the entry was in the common femoral artery, a 7 French 24
cm sheath was placed in the right femoral artery. Using a modified
Seldinger technique, a 7 French 24 cm sheath was placed in the right
femoral vein. A 5 French, balloon-tipped pacing wire was advanced
into the right ventricular apex and adequate pacing thresholds were
confirmed. An angled Glide catheter was inserted through the right
femoral sheath, and advanced to the ascending aorta with the aid
of a Bentson wire. The Bentson wire was then replaced with a 260
cm J-tipped wire, and the wire was advanced to the aortic valve.
The Glide catheter was removed and replaced by a 5 French pigtail
catheter. The pigtail catheter was positioned in the right coronary
sinus. Diagnostic aortography was performed to identify a coplanar
view. The left femoral artery was then punctured with a needle under
direct visualization. A soft wire was advanced to the distal aorta, and
a 7 French 24 cm sheath was inserted and flushed. Heparin was given
as a 7500 unit bolus with Activated Clotting Time (ACT) monitoring,
and the ACT kept above 250 seconds. A pigtail catheter was inserted
into the sheath in the left femoral artery, and advanced over a soft
wire into the abdominal aorta. The catheter was used to steer the
wire into the thoracic aorta, and the pigtail catheter was advanced
to the aortic arch. The soft wire was removed and replaced with a
Lunderquist wire. After removal of the 7 French sheath, 16 French
and 18 French dilators were introduced, after which an Edwards 18
French eSheathTM was inserted under fluoroscopic guidance with the
end placed in the upper abdominal aorta.
Simultaneously, a 23 mm Edwards Sapien XTTM valve was
prepared on the back table. The aortic valve was crossed with an AL1
catheter. An exchange wire was placed in the apex of the left ventricle,
and a pigtail catheter was advanced over the wire and positioned in
the left ventricular apex. The wire was then exchanged for a 260 cm
Safari wire, and the pigtail catheter was removed. We then performed
aortic valvuloplasty and deployed the 23 mm Edwards Sapient XTTM
valve during rapid ventricular pacing (170 bpm). Transthoracic
echocardiography showed motion of the leaflets and trivial aortic
insufficiency. The guidewire was then removed from the LV. The
valve appeared stable by echocardiography and fluoroscopy. The
pigtail catheter was withdrawn.
The patient tolerated the TAVR procedure without any problems
and without any change in hemodynamics. Therefore, we decided to
proceed with EVAR. An exchange-length wire was inserted via the
pigtail catheter in the right femoral sheath, after which the pigtail
was removed. A 1 cm skin incision was made over the sheath in the
right femoral artery, and the tract was spread with hemostats. Two
Perclose ProGlide vascular closure devices (Abbott Vascular, Abbott
Park, IL) were deployed at 10 o'clock and 2 o'clock positions, after
which the wire was reinserted, and a 7 French 14cm sheath was
inserted. The soft wire was removed and replaced by a Lunderquist
wire, and a 16 French sheath was inserted. An Omniflush catheter
was inserted via the left femoral sheath. The 18 French sheaths in the
left femoral artery was withdrawn so that its tip was in the aneurysm.
Now using the 20-French sheath on the left side, we placed a marking
pigtail catheter into the aorta. Aortography showed the location
of the renal arteries and location of the neck. Next, the sheath was
withdrawn, and the main body of the endograft (Medtronic Endurant
device, Medtronic (Minneapolis, MN) was then placed just below
the origin of the lowest renal artery. The device was then deployed
keeping the gate at 10 o'clock position and the main body was then
deployed until the contralateral gate opened. Next, due to tortuosity
of the iliac, we kept a large sheath in the left iliac over a stiff wire and
used a buddy wire to cannulate the contralateral gate. The catheter
was confirmed to be inside the endograft by swirling it multiple
times inside the aortic graft. Now the contralateral gate was deployed
via the left side. A Reliant balloon (Medtronic, Minneapolis, MN)
was then used to perform inflation at the proximal neck as well as
at the junction points. Completion angiography was performed,
which showed patent renal arteries and patent bilateral hypogastric
arteries. The aneurysm was successfully excluded (Figure 4). There
was some delayed, most likely a type 2, endoleak, but overall flow in
the aneurysm was remarkably diminished. At this point in time, the
sheath from the right side was then removed and closure devices were
then deployed. Adequate hemostasis was achieved. The left femoral
artery was repaired primarily with 5-0 prolene sutures. The incision
was closed in multiple layers and skin was closed with dermabond.
The overall operative time was around 120 minutes. Total
amount of contrast used was about 50 ml. The patient tolerated the
procedure well. On postoperative day number one, patient developed
asymptomatic atrial fibrillation, which was treated with cardioversion.
Patient was given regular diet on postoperative day one. Due to her
advanced age and frailty, she required physical therapy and was
discharged to a rehabilitation facility on ninth post-operative day.
She progressed well in the rehabilitation center and was discharged
to home after spending about twelve days in the rehabilitation center.
She was seen in outpatient clinic a month after surgery and was found
to be doing well. About 2 months after surgery, she was found by her
primary care physician to be getting lethargic. Initially, her physicians
felt that it was due to increasing azotemia, which was most likely
due to poor oral intake. However, within next few days, her BUN
and Creatinine returned to normal, but she persistently remained
confused. She denied evaluation or transportation to a hospital and
family supported this. Primary care physicians offered her hospice
and she passed way peacefully.
Figure 1
Figure 2
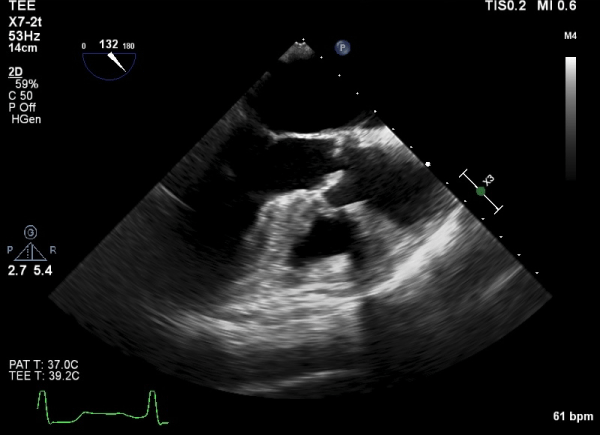
Figure 2
Transesophageal echocardiogram showing severe aortic valve stenosis in the 120 degrees view.
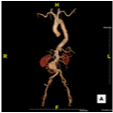
Figure 3
Figure 3
Three- Dimensional reconstruction of Computed Tomography scan showing infra renal abdominal aortic aneurysm.
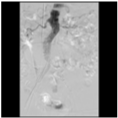
Figure 4
Figure 4
Aortogram status post deployment of endograft, showing successful exclusion of abdominal aortic aneurysm.
Discussion
The geriatric population is a growing segment of US population
[2]. In year 2000, people above 85 years of age constituted 1.5% of
entire US population and it is projected that by year 2050, this ratio
will increase to 5% [2]. Current life expectancy for people aged 85 is
about 6.4 years, implying that majority of these people will be enter
the tenth decade of their lives [4]. Before the advent of minimally
invasive procedures, the only treatment options for the treatment
of aortic stenosis and AAA were open aortic valve replacement
and open abdominal aortic aneurysm repair respectively. The
geriatric population has routinely been denied open operations of
this magnitude due to their frailty. The emergence or endovascular
technique has changed the face of modern surgery with increasing
numbers of patients being treated with TAVR for severe aortic
stenosis and EVAR for AAA.
Calcific aortic stenosis is found in about 20% of geriatric
population [5]. Operative repair of severe aortic stenosis in this
population is associated with significant mortality [3]. Traditionally,
about one third of patients with severe, symptomatic aortic stenosis
were not deemed to be surgical candidates due to presence of
significant comorbidities, advanced age being one of them [6 and 7]. The
introduction of TAVR offers hope for high-risk patients who were
traditionally denied surgical valve repair. Large clinical trials have
clearly shown that TAVR is superior to medical treatment of aortic
stenosis and is not inferior to open surgical repair [8-10].
Abdominal aortic aneurysms are a significant cause of morbidity
and mortality. The risk of rupture increases with increasing diameter.
The standard of care requires repair of AAA prophylactically to
prevent rupture in patients who are deemed appropriate candidates
[11]. EVAR has shown to improve short-term morbidity and
mortality, as compared to open repair, without any difference in longterm
survival [12-14]. EVAR has the advantage of avoiding the need
for exploratory laparotomy with associated fluid shifts. It also avoids
the need for aortic cross clamping with associated hemodynamic
changes. Operative blood loss is lower with EVAR. EVAR can be
performed with either femoral cut-downs or percutaneous femoral
access under local anesthetic and is physiologically less stressful to
the body. Recent literature shows that the incidence of complications
after groin cut-down and percutaneous femoral access for EVAR is
similar [15].
The geriatric population will especially benefit from endovascular
operations, given the fact that these operations can be performed
without much physiologic stress and in a much shorter operation
time. Both TAVR and EVAR can be performed via Transfemoral
access. The advantage of doing both operations at the same time is
that they can be performed using the same access site and without
the need for a second anesthetic for the second operation. The
disadvantages of combined procedure are increased operative time
and higher (combined) contrast dose. The key issue in offering
these operations to nonagenarians is to determine if the patients
will truly benefit from them. Traditionally, prophylactic operations,
such as AAA repair, have not been offered to elderly patients on the
assumption that their life expectancy would not justify the immediate
operative risk. As life spans increase and newer operative techniques
become less risky, this evaluation will have to be individualized, rather
than mad on the basis of age alone. There will still be patients whose
procedural risks will remain excessive in relation to any expected
benefit in symptoms or longevity, but there will be fewer such patients
than previously. Lindman et al. [16] have discussed the principle of
therapeutic futility when dealing with high surgical risk in an elderly
patient population. Therapeutic futility has been defined as “a lack of
medical efficacy, particularly when the therapy is unlikely to produce
its intended clinical result, as judged by the physician; or lack of a
meaningful survival, as judged by the personal values of the patient”
[16]. Careful patient selection is the key–patients who are physically
active and are expected to live a healthy, meaningful life should
be offered the surgical operations. This case highlights the real life
dilemma of practicing physicians who have to make decisions based
on the information they have. Despite having multiple discussions
with this patient and family to explain risks, benefits and alternatives,
patient wanted to pursue all surgical options to prevent mortality.
Yet, after a few months after surgery, she refused to get diagnostic
work up for lethargy and ultimately chose hospice. Certainly, if she
had expressed these wishes before, physicians involved in her care
would have offered her no surgical intervention. This case depicts
the real life scenario, in which patients play an active role in clinical
decision-making and have the right to change their minds whenever
they wish.
Our case shows that both TAVR and EVAR can be performed
safely in nonagenarians. After careful screening and thoughtful
process, suitable patients with severe aortic stenosis and large
abdominal aortic aneurysms can be treated by simultaneous TAVR
and EVAR.
References
- Manton KG. Recent declines in chronic disability in the elderly U.S. population: risk factors and future dynamics. Annu Rev Public Health. 2008; 29: 91-113.
- US Census Bureau ACsr, ACS-17, 90+ in the United States. http://www. census.gov/prod/2011pubs/acs-17.pdf.
- Thourani VH, Myung R, Kilgo P, Thompson K, Puskas JD, Lattouf OM, et al. Long-term outcomes after isolated aortic valve replacement in octogenarians: a modern perspective. Ann Thorac surg. 2008; 86: 1458- 1464./a>
- Reports NVS. http://www.cdc.gov/nchs/data/nvsr/nvsr57/nvsr57_14.pdf.
- Aronow WS, Kronzon I. Prevalence and severity of valvular aortic stenosis determined by Doppler echocardiography and its association with echocardiographic and electrocardiographic left ventricular hypertrophy and physical signs of aortic stenosis in elderly patients. Am J Cardiol. 1991; 67: 776-777.
- Iung B, Cachier A, Baron G, Messika-Zeitoun D, Delahaye F, Tornos P, et al. Decision-making in elderly patients with severe aortic stenosis: why are so many denied surgery? Eur Heart J. 2005; 26: 2714-2720.
- Bach DS, Siao D, Girard SE, Duvernoy C, McCallister BD Jr, Gualano SK. Evaluation of patients with severe symptomatic aortic stenosis who do not undergo aortic valve replacement: the potential role of subjectively overestimated operative risk. Circ Cardiovasc Qual Outcomes. 2009; 2: 533-539.
- Cribier A, Eltchaninoff H, Bash A, Borenstein N, Tron C, Bauer F, et al. Percutaneous transcatheter implantation of an aortic valve prosthesis for calcific aortic stenosis: first human case description. Circulation. 2002; 106: 3006-3008.
- Joint Task Force on the Management of Valvular Heart Disease of the European Society of C, European Association for Cardio-Thoracic S, Vahanian A, Alfieri O, Andreotti F, Antunes MJ, Barón-Esquivias G, et al. Guidelines on the management of valvular heart disease (version 2012). Eur Heart J. 2012; 33: 2451-2496.
- Holmes DR Jr., Mack MJ, Kaul S, Agnihotri A, Alexander KP, Bailey SR, et al. 2012 ACCF/AATS/SCAI/STS expert consensus document on transcatheter aortic valve replacement: developed in collabration with the American Heart Association, American Society of Echocardiography, European Association for Cardio-Thoracic Surgery, Heart Failure Society of America, Mended Hearts, Society of Cardiovascular Anesthesiologists, Society of Cardiovascular Computed Tomography, and Society for Cardiovascular Magnetic Resonance. J Thorac Cardiovasc surg. 2012; 144: e29-e84.
- Lederle FA, Johnson GR, Wilson SE, Ballard DJ, Jordan WD Jr, Blebea J, et al. Rupture rate of large abdominal aortic aneurysms in patients refusing or unfit for elective repair. JAMA. 2002; 287: 2968-2972.
- Prinssen M, Verhoeven EL, Buth J, Cuypers PW, van Sambeek MR, Balm R, et al. A randomized trial comparing conventional and endovascular repair of abdominal aortic aneurysms. N Engl J M. 2004; 351: 1607-1618.
- Goueffic Y, Becquemin JP, Desgranges P, Kobeiter H. Midterm survival after endovascular versus open repair of infrarenal aortic aneurysm s. J Endovasc Ther. 2005; 12: 47-57.
- Greenhalgh RM, Brown LC, Kwong GP, Powell JT, Thompson SG, EVAR trial participants. Comparison of endovascular aneurysm repair with open repair in patients with abdominal aortic aneurysm (EVAR trial 1), 30-day operative mortality results: randomised controlled trial. Lancet. 2004; 364: 843-848.
- Kauvar DS, Martin ED, Givens MD. Thirty-Day Outcomes after Elective Percutaneous or Open Endovascular Repair of Abdominal Aortic Aneurysms. Ann of vasc surg. 2016; 31: 46-51.
- Lindman BR, Alexander KP, O'Gara PT, Afilalo J. Futility, benefit, and transcatheter aortic valve replacement. JACC Cardiovasc interv. 2014; 7: 707-716.