Case and Review
Posterior Pelvic Reconstruction: A Case and Review of the Literature
Jill P Stone1, Mieke Heyns2, Rebecca Hartley1, Heyns M2, Marcio Barreto3 and Claire Temple-Oberle1,3*
1Division of Plastic Surgery, University of Calgary, Canada
2University of Calgary, Canada
3Division of Surgical Oncology, University of Calgary, Canada
*Corresponding author: Claire Temple-Oberle, Division of Surgical Oncology, Department of Surgery, University of Calgary, 1331 29 St NW Calgary, Canada, T2A4N2
Published: 30 Sep, 2016
Cite this article as: Stone JP, Heyns M, Hartley R, Barreto
M, Temple-Oberle C. Posterior Pelvic
Reconstruction: A Case and Review of
the Literature. Clin Surg. 2016; 1: 1147.
Abstract
Introduction: En-bloc tumor resection of the sacrum can leave the posterior pelvis devoid of
structural support and soft tissue coverage, putting patients at risk of postoperative complications
and sacral herniation. Compared to total sacrectomies, partial sacrectomy defects can often be
managed without instrumentation to reconstruct the structural support of the spinopelvic junction.
However, lack of skeletal support and often large soft tissue defects lead to challenges in decisionmaking
for posterior pelvic reconstruction (PPR).
Objective: The objective of this paper is to share a reconstructive method for posterior pelvic
reconstruction following a partial sacrectomy and present a systematic review of the current
literature.
Material and Methods: A systematic review identified relevant studies published through Medline
until July 2015. Search terms focused on (1) reconstruction (2) primary sacral tumors. Data
collection included the following: (1) Patient demographics (2) Sacral resection (3) Reconstruction
of defect (4) Tumor pathology (5) Patient outcome complications
and follow-up.
Result: A total of twenty-three articles highlight soft tissue reconstruction in posterior sacral
defects following partial sacrectomies. Fifty-nine patients met our inclusion criteria. Age ranged
from 23 71.
There were 28 male and 28 female (3 unknown). The most common pathology was
chordoma (n=49, 83%). Other pathology included giant cell tumor, myxopapillary ependymoma,
and chondrosarcoma. Follow-up time ranged from 696
months. Eighteen articles highlighted used
of soft tissue flaps. Gluteal based and vertical rectus flaps and were most common. Other closures
included omental, paraspinous, gluteal thigh and free latissimus flaps. Complications included
infection, deep vein thrombosis, flap necrosis, wound dehiscence, seroma and rectal perforation
secondary to mesh.
Discussion: Partial sacrectomy reconstruction is varied in its approach. Few published case reports
and series are available to guide the reconstructive surgeon.
Keywords: Traumatic lung herniation; Chest wall; Vertical thoracotomy
Introduction
En-bloc tumor resection of the sacrum can leave the posterior pelvis devoid of structural support and soft tissue coverage, putting patients at risk of postoperative complications and sacral herniation [1]. Compared to total sacrectomies, partial sacrectomy defects can often be managed without instrumentation to reconstruct the structural support of the spinopelvic junction [2]. However, lack of skeletal support and often large soft tissue defects lead to challenges in decisionmaking for posterior pelvic reconstruction (PPR) [3]. A variety of tissue flaps have been previously described including gluteal muscle flaps, VRAM flaps, paraspinous flaps and omental flaps [4]. Other support includes use of synthetic materials and acellular dermal matrices [5,6]. The objective of this systematic review was to examine the current literature on PPR of partial sacrectomy defects involving soft tissue reconstruction.
Case Presentation
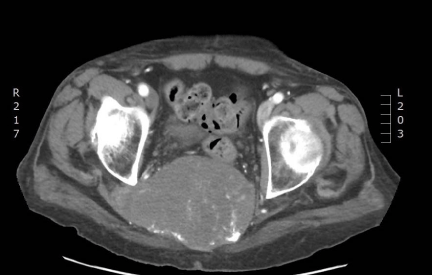
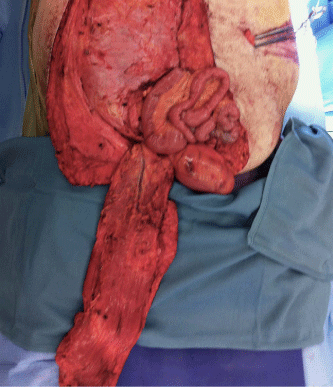
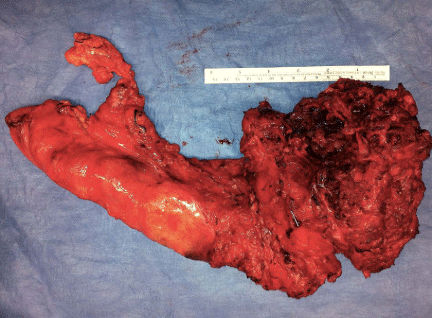
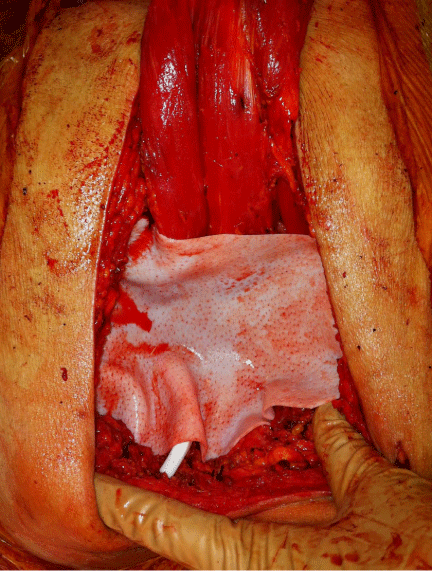
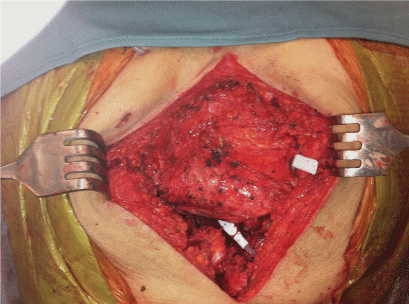
A retrospective review of the chart was initiated. A 75yearold male with presented with a one-year history of lower back pain. Computed tomography imaging demonstrated an 8 x 12 x 7 cm destructive lesion in his upper sacrum later confirmed by biopsy to be a classic type chordoma (Figure 1). The procedure began with the reconstructive team raising an inferiorly based vertical rectus flap and the general surgery team performing accessing and releasing the anterior portion of the tumor (Figure 2). In the second stage of the procedure, the spinal surgery team resected the remaining tumor requiring sacrifice of SGAP and IGAP perforators obviating gluteal based flaps as a potential reconstruction (Figure 3). The resultant defect measured 20x20cm. To isolate the small bowel from herniating posteriorly, a 16 x 20 cm ACD was secured to remaining bone and gluteus muscle residuum, leaving a superior opening for the pedicled VRAM (Figure 4). The VRAM flap was deepithelized and positioned over the ACD thus obliterating dead space (Figure 5). Primary closure was achievable. The patient was reassessed at 13 months post operation and had no evidence of sacral herniation. The patient passed away 15 months post-operation.
Figure 1
Figure 2
Figure 3
Figure 4
Figure 5
Methods
Search strategy and selection criteria
A systematic review identified relevant studies published through
Medline from 1970 until July 2015. Search terms focused on (1)
reconstruction (2) primary sacral tumors (Appendix). All study types
were included in the initial search, including abstracts and pending
publications available. Non-electronic versions were requested.
Inclusion criteria required the publications to be pertaining to
reconstruction of partial sacral defects from a primary sacral lesion.
Articles were excluded if they had no reconstructive data or were not
clinical cases (Figure 6). Two authors (J.S. and B.H.) independently
reviewed the articles. Disagreements on consensus of inclusion or
exclusion were arbitrated by a third author (M.B.).
Data extraction and outcomes
Title, author, journal, publication year and study type were
recorded. Data collection included the following when available:
(1) Patient demographics (2) Sacral resection (3) Reconstruction of
defect (4) Tumor pathology (5) Patient outcome–complications and
follow-up.
Results
Study selection
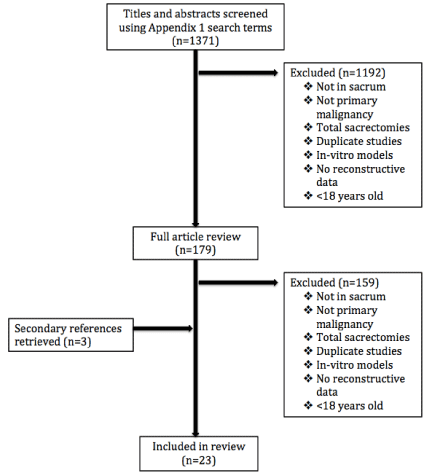
Using the previously described search method, 1371 articles were
identified (Figure 1). Articles were screened by title and abstract using
our exclusion criteria, 1192 articles were removed from the search.
The remaining 179 articles were reviewed in full further excluding
159. Secondary resources revealed an additional four studies.
Demographics
A total of twenty-three articles highlight soft-tissue reconstruction
in posterior sacral defects following partial sacrectomies. Articles
included case reports (n=9) and case series (n=14) highlighting a
variety of reconstructions for sacral defects. A total of 59 patients met
our inclusion criteria (Table 1). Eight articles reported on patients
undergoing partial and total sacrectomies for primary neoplasms.
Five of these include reconstructive data for secondary neoplasms of
the sacrum. Papers recording total sacrectomy and partial sacrectomy
patients and/or secondary pathology of the sacrum in total numbers
(specific data for partial sacrectomy patients could not be extracted)
were excluded from the demographic calculations (denoted with * &
ν). The remaining 15 articles accounted for 28 male and 28 female who
underwent soft tissue reconstruction for partial sacrectomies (3 not
recorded). Age ranged from 23–71. There most common pathology
was chordoma (n=49, 83%). Other pathology included giant cell
tumor, myxopapillary ependymoma, and chondrosarcoma. The most
common pathology reported in all articles was chordoma (18 of 23
articles) (Table 1). Follow-up time ranged from 6-96 months.
Soft tissue reconstruction
Soft tissue reconstruction was commonly utilized in partial
sacrectomies. Eighteen articles used soft tissue flaps. Gluteal-based
and vertical rectus flaps were most common (11 articles & 7 articles,
respectively). Other closures included omental, paraspinous, gluteal
thigh and free latissimus flaps. Five articles highlighted use of
synthetic materials including Dacryl© mesh, Polypropylene, and an
unknown mesh brand for structural support, with it being used in two
patients for repair of late sacral herniation at 4 and 72 months (Atkin,
Junge). Another late sacral herniation was treated with acellulardermal matrix one-year post resection (Brizidene). Acellular dermal
matrices combined with gluteal-based reconstruction were used in
29 cases. Complications included infection, deep vein thrombosis,
flap necrosis, wound dehiscence, seroma, and rectal perforation
secondary to mesh. Complications are listed in Table 1 and include
undifferentiated complications of total and partial sacrectomies
as well as secondary malignancy reconstruction that could not be
delineated for some articles.
Figure 6
Discussion
Compared to total sacrectomies, partial sacrectomy defects
can often be managed without instrumentation to reconstruct the
structural support of the spinopelvic junction provided the sacroiliac
(SI) joint is spared. However, lack of skeletal support and often large
soft tissue defects lead to challenges in decision-making for posterior
pelvic reconstruction (PPR). Without adequate reconstruction
following partial sacrectomy, post-operative complications including
infection, herniation, and fistula formation may ensue [7]. Resection
volume has previously been determined to be a factor in choice
of flap reconstruction [4]. Other factors that may determine flap
reconstruction may include previous radiation to the site or sacrifice
of the gluteal vessels secondary to reconstruction. Prior laparotomy
or previous ostomy site once precluded the vertical rectus flap as an
option but this is no longer supported [8,9].
The most common flap reconstructions following partial
sacrectomies were gluteal-based flaps. Reconstruction included
unilateral or bilateral and sliding/advancement and turn-over
methods. Advantages of this flap include adequate bulk, robust
blood supply, and proximity to the defect [9 and 10]. Additionally, it
allows for a posterior-only approach [11]. Disadvantages include
possible disruption of gluteal perforators during tumor resection
and risk of gluteal weakness affecting gait [7 and 9]. Vertical rectus flowthrough
flaps offer the advantage of having a robust blood supply as
well as providing sufficient soft tissue bulk and long pedicle length
[12]. Donor site complications including abdominal herniation and
weakness should be factored into decision-making.
Marichevi et al. [13] present evidence that acellular dermal matrix
(ADM) reconstruction decreased the number of intra-abdominal
complications when compared to a control group. The use of ADM
restores the continuity of the posterior abdominal wall as well as
provides an additional barrier between intraperitoneal space and
instrumentation if present. Addition of flap reconstruction helps
to obliterate the dead space and minimizes seroma and infection.
Sciubba et al. [3] present a decrease trend in surgical site infection with
presence of soft-tissue flap reconstruction, further supporting other’s
recommendations of utilizing flaps for sacrectomy defects. There are
few published studies presenting partial sacrectomy reconstruction
and their possible complications. The current published level of
evidence available to guide the reconstructive surgeon in tackling
partial sacrectomy defects is limited. A summary of options are
presented in this manuscript.
Table 1
Conclusion
A review of the literature on soft tissue reconstruction of the posterior pelvis following partial sacrectomy reveals that VRAM and gluteal perforator flaps are appropriate choices for obliterating dead space and closure of skin deficiencies, and ACD provides structural support and fewer intra abdominal complications.
Appendix
Search terms applied to Medline Ovid database.
General scheme
[(‘‘tumor terminology’’ AND ‘‘sacral area terminology’’)
OR (‘‘sacral resection terminology’’)] AND [(‘‘reconstruction
terminology’’)].
Search terms
Tumor terminology
“primary tumor” OR “neoplasm” OR “bone neoplasms”
OR “chondrosarcoma” OR “chondrosarcoma” OR “giant cell
tumor” OR “lymphoma” OR “multiple myeloma” OR “myeloma”
OR “plasmacytoma” OR “Ewing sarcoma” OR “chordoma” OR
“osteosarcoma” OR “osteogenic sarcoma” OR “spinal neoplasms” OR
“bone cysts” OR “aneurysmal bone cysts”.
Sacral area terminology
“sacrum” OR “sacral” OR “Sacrum” OR “lumbosacral”
OR “lumbo-sacral” OR “spinal pelvic” OR “spinal-pelvic” OR
“spinopelvic” OR “sacroiliac” OR “sacro-iliac” OR “iliosacral” OR
“ilio-sacral” OR “lumbo-pelvic” OR “lumbopelvic” OR “lumbo-iliac”
OR “lumbosacropelvic” OR "posterior pelvis" OR "posterior pelvic
wall".
Sacral resection terminology
("partial sacrectomy" OR "en bloc resection" OR "enbloc resection"
OR (("sacral" OR"sacrum") and resection)).
Reconstruction terminology
"reconstruction" OR "flap" OR "mesh" OR "acellular dermal
matrix" OR "Strattice" OR "Alloderm" OR "musculocutaneous flap"
OR "cutaneous flap" OR "fasciocutaneous flap" OR "gluteal flap" OR
"soft tissue reconstruction" OR "soft tissue coverage" OR "soft-tissue
reconstruction" OR "reconstructive procedures" OR "reconstructive
surgical procedures" OR "free flap" OR "myocutaneous flap" OR
"rectus flap" OR "vertical rectus flap" OR "flap" OR "woundclosure".
References
- Abhinav K, Shaaban M, Raymond T, Oke T, Gullan R, Montgomery AC. Primary reconstruction of pelvic floor defects following sacrectomy using Permacol graft. Eur J Surg Oncol. 2009; 35: 439-443.
- Bederman SS, Shah KN, Hassan JM, Hoang BH, Kiester PD, Bhatia NN. Surgical techniques for spinopelvic reconstruction following total sacrectomy: a systematic review. Eur Spine J. 2014; 23: 305-319.
- Sciubba DM, Nelson C, Gok B, McGirt MJ, McLoughlin GS, Noggle JC, et al. Evaluation of factors associated with postoperative infection following sacral tumor resection. J Neurosurg Spine. 2008; 9: 593–599.
- Garvey PB, Rhines LD, Feng L, Gu X, Butler CE. Reconstructive strategies for partial sacrectomy defects based on surgical outcomes. Plast Reconstr Surg. 2010; 127: 190-199.
- Atkin G, Nmathur P, Ahrrion R. Mesh repair of sacral hernia following sacrectomy. J R Soc Med. 2003; 96: 28–30.
- Korn JM, Connolly MM, Walton RL. Single-stage sacral coccygectomy and repair using human acellular dermal matrix (AlloDerm) with bilateral gluteus maximus flaps for hernia prophylaxis. Hernia. 2009; 13: 329-332.
- Furukawa H, Yamamoto Y, Igawa H, Sugihara T. Gluteus maximus adipomuscular turnover sliding flap in the surgical treatment of extensive sacral chordomas. Plast Reconstr Surg. 2000; 105: 1013–1016.
- Glatt BS, Dissa JJ, Mehrara BJ, Pusic AL, Boland P, Cordeiro PG. Reconstruction of extensive partial or total sacrectomy defects with a transabdominal vertical rectus abdominis myocutaneous flap. Ann Plast Surg. 2006; 56: 526–531.
- Weitao Y, Qiqing C, Sontao G, Kiaqiang W. Use of gluteus maximus adipomuscular sling flaps in the reconstruction of sacral defect tumor resection. World J Surg Oncol. 2013; 11: 110.
- Sahakitrungruang C, Chantra K, Dusitanond N, Atittharnsakul P, Rojanasakul A. Sacrectomy for primary sacral tumors. Dis Colon Rectum. 2009; 52: 913-918.
- Clarke MJ, Dasenbrock H, Bydon A, Sciubba DM, McGirt MJ, Hsieh PC, et al. Posterior-only approach for en bloc sacrectomy: clinical outcomes in 36 consecutive patients. Neurosurg. 2012; 71: 357–364.
- Cheong YW, Sulaiman WA, Halim AS. Reconstruction of large sacral defects following tumor resection: a report of two cases. J Orthop Surg. 2009; 16: 351-354.
- Maricevich M, Maricevich R, Chim H, Moran SL, Rose PS, Mardini S. Reconstruction following partial and total sacrectomy defects: an analysis of outcomes and complications. J Plas, Recon, & Aesth Surg. 2014; 67: 1257-1266.
- Gillis CC, Street JT, Boyd MC, Fisher CG. Pelvic reconstruction after subtotal sacrectomy for sacral chondrosarcoma using cadaveric and vascularized fibula autograft: Technical note. J Neurosurg Spine. 2014; 21: 623-627.
- Asavamongkolkul A, Waikakul S. Wide resection of sacral chordoma via a posterior approach. Int Orthop. 2012; 36: 607–612
- Althausen PL, Schneider PD, Bold RJ, Gupta MC, Goodnight JE, Khatri VP. Multimodality management of giant cell tumor arising in the proximal sacrum. Spine. 2002; 27: E361–365.
- Brizendine JB, LeFaivre JF, Yost MJ, Fann SA. Reconstruction of parasacral hernia with acellular human dermis graft. 2006; 10: 360-363.
- Dasenbrock HH, Clarke M, Bydon A, Witham TF, Sciubba DM, Simmons OP, et al. Reconstruction of extensive defects from posterior en bloc resection of sacral tumors with human acellular dermal matrix and gluteus maximus myocutaneous flaps. Neurosurg. 2011; 69: 1240-1247.
- Di Benedetto G, Bertani A, Pallua N. The free latissimus dorsi flap revisited: a primary option for coverage of wide recurrent lumbosacral defects. Plast Reconstr Surg. 2002; 109: 1960-1965.
- Junge K, Krones CJ, Rosch R, Fackeldey V, Schumpelick V. Mesh reconstruction preventing sacral herniation. Hernia. 2003; 7: 224-226.
- Koh PK, Tan BK, Hong SW, Tan MH, Tay AG, Song C, et al. The gluteus maximus muscle flap for reconstruction of sacral chordoma defects. Ann Plast Surg. 2004; 53: 44–49.
- Korn JM, Connolly MM, Walton RL. Single-stage sacral coccygectomy and repair using human acellular dermal matrix (AlloDerm) with bilateral gluteus maximus flaps for hernia prophylaxis. 2009; 13: 329-332.
- RamamurthyR, BoseJC, MuthusamyV, NatarajanM, Kunjithapatham D. Staged sacrectomy—an adaptive approach. J Neurosurg Spine. 2009; 11: 285–294.
- Schwab JH, Healey JH, Rose P, Casas-Ganem J, Boland PJ. The surgical management of sacral chordomas. Spine. 2009; 34: 2700-2704.
- Solini A, Gargiulo G, Fronda G, De Paolis P, Ruggieri N, Garino M. Emisacrectomy, experience in 11 cases. Eur Spine J. 2009; 18: 109-114.
- Varga PP, Lazary A. Chordoma of the sacrum: “En bloc” high partial sacrectomy. Eur Spine J. 2010; 19: 1037-1038.