Review Article
Smoking, Colorectal Disease and Colorectal Surgical Practice
David G1#, West MA2,3#, Magee CJ1 and Walsh CJ1*
1Department of Surgery, Wirral University Teaching Hospital NHS Foundation Trust, UK
2Academic Unit of Cancer Sciences, University of Southampton, UK
3Department of Surgery, University Hospital Southampton NHS Foundation Trust, UK
#Both authors have contributed equally to this work
*Corresponding author: Walsh CJ, Department of Surgery, Wirral University Teaching Hospital NHS Foundation Trust, Wirral, UK
Published: 15 Sep, 2016
Cite this article as: David G, West MA, Magee CJ, Walsh
CJ. Smoking, Colorectal Disease and
Colorectal Surgical Practice. Clin Surg.
2016; 1: 1115.
Abstract
Background: Cigarette smoking adversely affects the natural history of a number of common
colorectal conditions and their treatment. Colorectal cancer is now recognised as a smoking
attributable cancer.
Aim: This is a narrative review of the effects of cigarette smoking on colorectal disease, colorectal
surgery and colorectal surgical outcomes.
Methods: Literature search was carried out for relevant articles highlighting relationship between
smoking and variety of colorectal conditions (cancer, diverticular disease, inflammatory bowel
disease and perianal diseases). As we wanted to highlight the breadth of the problem to current
colorectal practice, we have deliberately kept this as a narrative review.
Results: Smoking is associated with higher all-cause mortality and colorectal cancer–specific
mortality in non-metastatic colorectal cancer and it worsens the outcomes of a number of
inflammatory colorectal conditions including diverticular disease and Crohn’s disease. Smoking
impairs the inflammatory response, tissue repair and consequently impairs wound healing. Smoking
during radiotherapy worsens acute toxicity and reduces disease free survival in anal cancer.
Conclusion: Approximately 10 million adults smoke cigarettes in Great Britain currently and
therefore there is arguably no single greater reversible patient related parameter that affects
outcomes in colorectal practice. Surgeons must advocate and facilitate smoking cessation in their
patients putting particular emphasis on the likelihood of increased complication rates and poorer
outcomes if an active smoker undergoes colorectal surgery or radiotherapy.
Keywords: Smoking; Colorectal Disease; WHO
Introduction
In 1887 Thomas Morton of Philadelphia arguably ushered in the age of modern colorectal
surgery by performing the first appendicectomy for appendicitis [1]. He was probably not aware
that 400 miles away another significant, though malign, milestone in history had taken place six
years earlier. In 1881 James Duke and James Bonsack of Raleigh, North Carolina created the world’s
first cigarette-rolling machine. Their machine rolled 120,000 cigarettes per day thus revolutionizing
tobacco smoking as a habit.
According to the WHO report on the global tobacco epidemic (2013), tobacco kills approximately
6 million people worldwide and causes more than half a trillion dollars of economic damage each
year. It is the leading global cause of preventable death and is predicted to kill 1 billion people this
century. About 10 million adults smoke cigarettes in Great Britain, approximately 22% of adult men
and 19% of adult women are smokers (www.ash.org.uk April 2014). Smoking caused 60,000 cases of
cancer in 2010 and 43,000 cancer deaths in 2009 [2]. Approximately 1.6 million hospital admissions
in 2012/13 among adults aged >35 were primarily smoking related [3]. According to the report by
Surgeon General report in 2013, the annual cost for direct medical care of adults attributable to
smoking between the years 2009 and 2012 was $132.5 to 175.9 billion. More than 2.3 billion people
are now covered by at least one of the WHO Framework Convention on Tobacco Control (WHO
FCTC) but this covers mostly high income countries. Although substantial progress has been made
in the global tobacco use since the adoption of WHO FCTC 10 years
ago, more work is needed in the low and middle income countries [4].
The medical profession appreciate these devastating statistics
and the general ill effects of smoking, in particular lung cancer and
cardiovascular disease, but there is paucity of data on the effects of
smoking in patients with colorectal disease. The aim of this narrative
is to present a general review of smoking and its effect on colorectal
disease with special reference to contemporary colorectal surgery.
Table 1
Table 2
Smoking and Appendicitis
Appendicectomy is currently the commonest general surgical emergency operation. In 1999 Montgomery et al. [5] showed that maternal smoking and or patient smoking was positively associated with appendicectomy by the age of 26 years. The unadjusted odds ratio for appendicectomy associated with cigarette smoking was 2.34 (95% CI 1.52–3.59) [5]. A more recent study found associations between tobacco use and risks of perforation of an inflamed appendix [6]. Smoking therefore increases the incidence of appendicitis and also the likelihood of complications.
Smoking and Diverticular Disease
Smoking is a risk factor for the development of complicated
diverticular disease at a young age (Table 1). Turunen et al. [7] reported
on 261 patients undergoing sigmoid colectomy for complicated
diverticulitis. Smokers were younger, had more evidence of strictures,
histological perforations and more episodes of diverticulitis following
surgery. A smaller study comparing complicated diverticular
disease and asymptomatic diverticular disease showed a greater
proportion of smokers in the former group. Regression modelling
demonstrated smoking to be an independent risk factor predisposing
to postoperative complications [8].
Hjern et al. [9] concluded that in women, smoking appears
to increase the risk of symptomatic diverticular disease even after
controlling for confounding factors such as age, diabetes and obesity.
Of particular note smokers had almost double the risk of perforation
or abscess formation. Why smoking should be associated with
increased complicated disease in women is uncertain, but it is known
that smoking increases VIP (vasointestinal polypeptide) levels
in the colonic mucosa [10], thus increasing colonic motility and
intraluminal pressure [11], while impairing blood supply [12,13].
A contradictory study including only male health professionals
concluded that smoking was not a substantial risk for symptomatic
diverticular disease although less than 10% of the study population
smoked (RR 1.25, 95%CI 0.75-2.09) [14].
Smoking, Colorectal Cancer and Colorectal Cancer Treatments
The International Association for Research on Cancer added
Colorectal cancer to the list of smoking attributable cancers in
2009 [15,16]. The US Surgeon General did the same in 2014 [17].
According to several recent meta-analyses, the relationship between
smoking and colorectal cancer incidence is likely dose dependent [18-
21]. The association seems strongest for three correlated molecular
phenotypes of the disease: microsatellite instability (MSI) [22-
25], CpG island methylator phenotype (CIMP) [26-28] and BRAF
mutation positive [25-27].
The association between colorectal polyps, cancer and smoking
habits has been extensively studied (Table 2). The Cancer Prevention
Study II was a large prospective cohort established by the American
Cancer Society in 1982. Smokers of 20 or more years experienced
higher colorectal cancer death rates, even when adjusted for multiple
potential confounders. Consistent with a causal relationship with
smoking, risk was higher in current than in former smokers and
increased with smoking duration, number of cigarettes smoked per
day, and pack-years. Among former smokers, risk decreased with
the number of years since smoking cessation [29]. The association
between smoking and increased all causes mortality and disease
specific mortality is especially pronounced in colorectal cancer with
micro satellite instability (MSI). Across sex, tumour site, stage, and
MSI status, and for both all-cause and CRC-specific mortality, an
increased mortality was found in patients who were smokers than in
former smokers and highest in patients with a >40 pack-year history
[30].
Using the cohort of patients who participated in the Cancer
and Leukaemia group B phase 3 trials, a spin off study evaluated
the impact of smoking on colon cancer recurrence and survival.
Surgically treatable stage III colon cancer patients with no evidence of
distant spread were included. Smoking status or time since smoking
cessation did not impact on the disease-free survival, overall survival
or recurrence-free survival, but a dose-response association was noted
for smoking intensity, particularly for the risk of death or recurrence
in higher quartiles of pack-years smoked before age 30 years when
compared with non-smokers [31].
Yang et al. [32] have recently shown that in colorectal cancer
survivors smoking is associated with a nearly two-fold higher risk
of death compared with never smoking. These studies indicate that
cigarette smoking is associated with higher all-cause and colorectal
cancer–specific mortality in non-metastatic colorectal cancer.
In treatment of metastatic colorectal cancer with Cetuximab,
current smoking was found to be associated with poorer response,
quicker time to progression and decreased overall survival. There
was no dose relationship among current smokers [33]. In another
study observing survival among patients with a potentially curative
colorectal resection and referred for consideration for adjuvant
chemotherapy/radiotherapy, smoking was found to significantly
lower cause-specific survival by around 20% at 5 years [34].
There is little data on the effect of smoking on outcomes after
radiotherapy for rectal cancer in smokers versus non-smokers, but
there is compelling evidence from other diseases. In head and neck
cancer patients having radiotherapy active smokers had significantly
more complications and significantly poorer 5 years overall survival,
locoregional control and disease free survival [35]. Prostate cancer
patients who were current smokers had a 40% relative increase of
cancer relapse and were twice as susceptible to disease spread and
death due to prostate cancer compared to non-smokers [36].
Smoking and Anal Cancer
Risk factors for anal cancer include HPV infection, multiple sexual partners and ano-receptive intercourse. Smoking, as a risk factor is less well known (Table 3). In a retrospective review of about 300 patients with anal cancer, current smokers were at higher risk for anal cancer, independent of age and other risk factors [37]. Two studies using standard chemoradiotherapy found that smoking adversely affected local disease control, however this did not affect disease specific survival and colostomy free survival [38,39]. The European Society of Medical Oncology Clinical practice guidelines state that smoking may worsen acute toxicity during treatment and lead to a poorer disease free and colostomy free survival. Every effort should be made to ensure patients stop smoking before therapy [40].
Table 3
Table 4
Smoking and Inflammatory Bowel Disease
There is a dichotomy in the relationship between smoking,
ulcerative colitis and Crohn’s disease (Table 4). The British Cohort
Study in the 1980’s concluded that smoking protects against Ulcerative
Colitis (UC). The risk of developing UC is decreased in current
smokers compared with lifetime non-smokers (OR 0.41 95%CI 0.34–
0.48). In contrast smoking increased the risk of developing Crohn’s
Disease (CD) (OR 2.0 95%CI 1.65–2.47) [41].
In a meta-analysis of 16 studies that included 2962 patients,
there was almost twice the rate of clinical relapse of CD, following
surgery among smokers. The risk of re-operation was also twice as
high at 10-year follow up. There was no significant difference in reoperation
rate or postoperative acute relapses between ex-smokers
and non-smokers, indicating that smoking cessation reduces clinical
and surgical recurrence rates [42]. A more recent review article also
suggests an increased risk of clinical recurrence rates among smokers
with CD [43]. In smaller studies, where risk factors for re-do surgery
for CD were analysed, smoking was found to increase the risk of redo
[44] and secondary ileocolic resection [45].
Smoking has also been shown to increase the risk of endoscopic
recurrence, with macroscopic lesions found in the neo-terminal ileum
of 70% of smokers 1 year after surgery compared with 35% of non
smokers and 27% of ex-smokers [46]. Endoscopic recurrence rates
appear similar for ex-smokers and non smokers [47-49]. After ileorectal
anastomoses for CD smoking was an independent risk factor
which predicted further intervention and proctectomy [50]. In recent
studies involving Ileal Pouch Anal Anastomosis (IPAA) patients,
active smoking was a risk factor associated with development of CD
in patients who underwent this procedure for indeterminate colitis
[51]. Smoking was also found to be associated with pouch related
hospitalization and pouch failure [52].
In a large retrospective cohort study of 3000 patients,
evaluating the effect of smoking on current medical treatment
(immunosuppressant’s and anti-TNF drugs) for CD, smokers were
found to have a significantly decreased survival free of structuring
disease, perianal complications and had a higher risk for requiring
thiopurine therapy. Moreover, smokers were more frequently
treated with steroids, immunosuppressants and anti-TNF drugs
[53]. These results suggest that, despite the widespread use of
immunosuppressant’s and anti-TNF drugs, smokers with Crohn's
disease still have a more severe disease course with increased
therapeutic requirements. In a smaller study of about 300 patients
with CD, there was no statistically significant difference between
smokers (current and ex) and non-smokers in relation to disease
characteristics or post-operative complications. The smokers, however
had a lower Cleveland Quality of Life score [54]. In a more recent
study from Sydney IBD Database Cohort comprising about 1200
patients, smoking in CD was associated with increased proportional
surgery rate (45.8% vs. 37.8%, P = 0.045), requirement for IBD-related
hospitalisation (P = 0.009) and incidence of peripheral arthritis
(29.8% vs. 22.0%, P = 0.027). Current smokers with UC demonstrated
reduced corticosteroid utilisation (24.1% vs. 37.5%, P = 0.045), yet
no statistically significant reduction in the rates of colectomy (3.4%
vs. 6.6%, P = 0.34) or hospital admission (P = 0.25) relative to nonsmokers.
Ex-smokers with UC required proportionately greater
immunosuppressive (36.2% vs. 26.3%, P = 0.041) and corticosteroid
(43.7% vs. 34.5%, P = 0.078) therapies compared with current and
never smokers [55].
Smoking and Perianal Disease
In an epidemiological study of US veterans (74 with anal abscess
or fistula and 816 without) recent smoking was found to be a risk
factor for anal abscess/fistula development [56] (Table 5). Current
smokers developed more perianal disease than smokers who quit
less than a year ago. Increased OR 1.72 (95% CI 1.03–2.86, p=0.0375)
was also observed when comparing current smokers and smokers
who quit within the past 5 years compared with non-smokers. An
accompanying editorial questioned our understanding of the origin of
perianal sepsis and explores a plausible case for a causal relationship
between smoking and perianal sepsis [57].
Hidradenitis suppurativa (HS) is a chronic, recurrent,
inflammatory disorder of hair follicles in apocrine gland-bearing sites.
In a population based study of 200 patients, smokers (current and
past) were found to have more severe HS compared to non-smokers
(OR 2.0, 95% C.I 1.1–3.5) [58]. In a pair matched case-control study
between patients presenting with HS and other dermatological
conditions (atopic dermatitis, melanoma, varicose veins etc), the rate
of active smokers was higher in the HS group (90% vs. 46%) [59].
The percentage of smokers among patients with HS was found to be
higher in the above German and Polish study (73%) [60] compared to
the national average (27% and 23% respectively).
Table 5
Figure 1
Figure 2
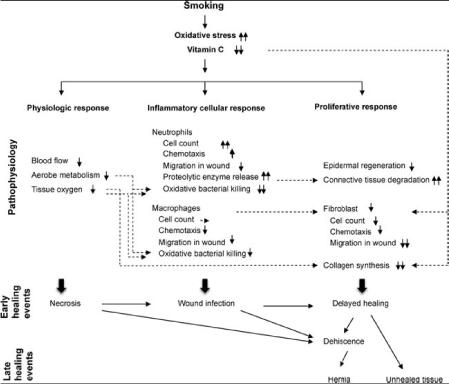
Smoking and Wound Healing
Smoking causes changes in all three phases of wound healing.
Smoking enhances the initial haemostatic phase through elevated
levels of circulating fibronectin; it reduces blood flow, aerobic
metabolism and tissue oxygenation. It impairs the inflammatory
phase with reduced neutrophil phagocytosis and abnormal monocytemacrophage
responses whilst the proliferative phase is deranged with
reductions in fibroblast function, epithelial regeneration and collagen
metabolism. These events are manifested clinically as delayed healing,
wound infection and necrosis [61-67] with subsequent anastomotic
breakdown or herniation [68].
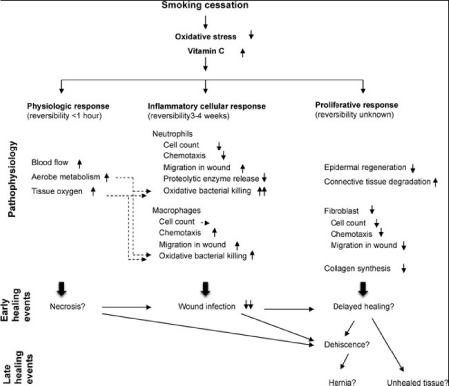
In a systematic review of 177 articles, smoking cessation was
seen to reverse certain adverse factors on wound healing. Cessation
restores tissue oxygen levels rapidly and reduces oxidative stress.
The number of circulating neutrophils and the negative impact on
neutrophil and monocyte-macrophage function are normalized. In
the healing wound, inflammatory cell infiltration increases and wound
contraction is partially reversed. In contrast, smoking cessation does
not appear to reverse epidermal regeneration, fibroblast proliferation,
and collagen synthesis and deposition, indicating that in the wound
the detrimental effect of smoking on proliferation and remodeling is
prolonged [69]. The pathophysiological effects of both smoking and
smoking cessation are summarized in Figure1 and 2.
In a post hoc analysis of data from PROXI trial, including
1386 patients (emergency and elective laparotomy for surgical
and gynaecological causes), smoking was found to be significantly
associated with higher frequency of surgical site infections (SSI) (25%
versus 17%, p value< 0.001) and burst abdomen (3.8% vs. 2.4%, p
value 0.04) [70].
Cigarette Smoking and Outcomes after Colorectal Surgery
Smokers are more likely to suffer surgical, as well as, cardiovascular
and respiratory complications [71,72]. Smokers are more likely to be
admitted to an intensive care unit [73], have longer lengths of stay
and higher mortality.
Two large retrospective studies from the Veterans Associations
Surgical Quality Improvement Programme (VA SQIP) showed that
smoking was associated with more post-operative complications
following both general and cancer related surgical procedures [74,75].
In a further large study of patients undergoing curative resection
for colorectal cancer, smoking (as a preoperative risk factor) was
associated with more post-operative complications poorer overall
survival and systemic recurrence [76] (Table 6).
In a more recent retrospective database (VA SQIP) study of
almost 48,000 patients undergoing colorectal surgery for cancer,
diverticular disease or IBD; current smokers were at a significantly
increased risk of post-operative morbidity and mortality [77]. In a
small study involving 38 patients, non-healing perineal wounds after
abdominoperineal resection were observed in 26%. On principal
component analysis of seven other factors, smoking was found to
contribute 13.8% of delayed wound healing [78].
In another small, but well conducted randomised clinical trial,
where an experimental wound was made and followed up, between
non-smokers and smokers, wound infection was commoner among
smokers (12% vs. 2%, p< 0.05). After four weeks of abstinence the
wound infection levels were similar to never smokers [79]. In a
retrospective case matched study of patients who developed acute
fascial wound dehiscence requiring return to theatre following an
earlier laparotomy, incidence of smoking was more common (46%)
compared to those who did not develop the complication (16%,
p=0.0002) [80].
In two large retrospective studies of over 3000 cases undergoing
ventral hernia repair [81] and abdominal wall reconstruction [82],
smoking was the only modifiable risk factor associated with wound
infection and major operative complication. Current smoking
was approximately 1.5 times more prevalent in subjects with
postoperative wound infections. In 2010, the Ventral Hernia Working
Group actively recommended smoking cessation as part of patient
optimisation prior to elective surgery [83].
Transanal advancement flap repair (TAFR) has been advocated
as a possible treatment of choice for trans-sphincteric fistula in
ano. Initially promising reports [84-87] have been followed by less
favourable results [88,89]. To examine this further 105 patients with
crypto glandular perianal fistulae underwent TAFR and were assessed
for healing outcomes. TAFR was successful in 69% at a median of 14
months. The healing rate varied between 60% in smokers and 79%
in non smokers with a significant correlation between number of
cigarettes smoked per day and reduced healing [90]. The same group
went on to show significantly decreased blood flow in the rectal
mucosa among smokers prior to TAFR [13], mainly demonstrated at
the apex of the flap. In another study smoking was again found to be a
factor adversely affecting healing of advancement flaps and increased
recurrence rates [91]. In a study evaluating “staged fibrin sealant
(FS)” technique and TAFR for treatment of complex perianal fistulae,
100% of smokers in the FS group developed recurrent fistulae [92].
Table 6
Table 7
Smoking and Anastomotic Leaks
Anastomotic leakage is a very serious complication after colorectal
surgery [93,94], leak rates vary between 1.5% and 16% being highest
in low rectal anastomoses [95]. Anastomotic complications cause
increased morbidity and mortality [96], poor long term outcomes
and impaired functional results [97] (Table 7).
A combined clinical and histopathological study, correlating
smoking, hypertension and anastomotic dehiscence, among 147
patients, found a positive correlation between microvascular disease
and anastomotic dehiscence, with an increased incidence among
smokers [12]. In a retrospective analysis of 215 patients who underwent
low anterior resections for rectal cancer multivariate analysis revealed
the presence of a protective stoma and history of heavy smoking to
be significantly and independently associated with anastomotic
complications [98]. Similar studies of patients undergoing colonic
or rectal resections used regression analyses to identify smoking,
metastatic disease and alcohol abuse to be significantly associated
with higher anastomotic leak rates [99,100]. A recent systematic
review showed smoking to be an important adjustable risk factor for
anastomotic leak [101]. It is also reasonable to hypothesise that there
might be a cumulative effect of smoking and radiotherapy in particular
on anastomotic healing [101]. A more recent clinical study of 246
patients undergoing left sided colonic resections, the percentage of
anastomotic leak requiring an intervention, was significantly higher
among smokers (17%) compared to non-smokers (5%) [102]. A
much larger multicentre study from Germany including almost
18,000 patients over a 10 year period, also found smoking (reliant on
patient history) to be an independent factor in both univariate (leak
rate of 17% vs. 11%, p value >0.001) and multivariate analysis (O.R
1.332, 95% C.I 1.106 – 1.604) [103].
Conclusion
Cigarette smoking adversely affects the natural history of common
colorectal conditions, their treatment and surgical outcomes. As
well as the deleterious cardiorespiratory effects on blood supply,
pulmonary function and tissue oxygenation there is also a smoking
related impairment of the inflammatory response and tissue repair.
The latter, directly associated with impaired wound healing, an
increased incidence of anastomotic leakage, hernia formation and
flap failure. Smoking is associated with poorer cancer outcomes and
increased complications after colorectal surgery. Smoking is also
associated with poorer outcomes after radiotherapy.
Smoking history should be an integral part of the colorectal
patient’s assessment. There is an obligation to explain the effects
of smoking on the natural history of the disease and its treatment.
Surgeons must advocate and facilitate smoking cessation in order to
empower patients to alter the course of their disease for their benefit.
Whilst aware of the health risks of smoking in general terms many
patients don’t appreciate the specific risk to their colorectal disease
or their proposed surgery or radiotherapy. Smoking cessation should
become an integral part of enhanced recovery protocols which is not the
case at present in the UK. The optimal duration of smoking cessation
and success of smoking cessation programmes has been a subject of
few studies. A systematic review and Meta analysis concludes that
at least four weeks of abstinence from smoking reduces respiratory
complications and abstinence of at least three to four weeks reduces
wound healing complications [104]. Another systematic review and
meta analysis of smoking cessation interventions among oncology
patients (counselling, nicotine replacement therapy, buproprion and
varenicline) concludes that interventions in the perioperative period
had a pooled odds ratio of 2.31 (95% C.I 1.32 – 4.07), making this an
important teachable moment [105].
If smoking cessation advice is unsuccessful then the
appropriateness of proceeding with elective surgery in the face of
active smoking should be reconsidered [106]. If proceeding with
surgery, then alteration in surgical strategy with reconsideration of
anastomoses or flaps needs to be undertaken, particularly if other risk
factors are present.
References
- Seal A. Appendicitis: a historical review. Can J Surg. 1981; 24: 427-433.
- Brandt AM. The Cigarette Century: The Rise, fall and Deadly persistence of the Product that Defined America. Basic Books, New York. 2007.
- Statistics on Smoking: England 2011. The NHS Information Centre. 2011.
- WHO report on the global tobacco epidemic 2013.
- Montgomery SM, Pounder RE, Wakefield AJ. Smoking in adults and passive smoking in children are associated with acute appendicitis. Lancet. 1999; 353: 379.
- Sadr Azodi O, Lindstorm D, Adami J, Bellocco R, Linder S, Wladis A. Impact of body mass index and tobacco smoking on outcome after open appendicectomy. Br J Surg 2008; 95: 751-757.
- Turunen P, Wikstrom H, Carpelan-Holmstrom M, Kairaluoma P, Kruuna O, Scheinin T. Smoking increases the incidence of complicated diverticular disease of the sigmoid colon. Scand J Surg. 2010; 99: 14-17.
- Papagrigoriadis S, Macey L, Bourantas N, Rennie JA. Smoking may be associated with complications in diverticular disease. Br J Surg. 1999; 86: 923-926.
- Hjern F, Wolk A, Hakansson N. Smoking and the risk of diverticular disease in women. Br J Surg. 2011; 98: 997-1002.
- Miotto D, Boschetto P, Bononi I, Zeni E, Cavallesco G, Fabbri LM, et al. Vasoactive intestinal peptide receptors in the airways of smokers with chronic bronchitis. Eur Respir J. 2004; 24: 958-963.
- Milner P, Crowe R, Kamm MA, Lennard-Jones JE, Burnstock G. Vasoactive intestinal polypeptide levels in sigmoid colon in idiopathic constipation and diverticular disease. Gastroenterology. 1990; 99: 666-675.
- Fawcett A, Shembekar M, Church JS, Vashisht R, Springall RG, Nott DM. Smoking, hypertension, and colonic anastomotic healing; a combined clinical and histopathological study. Gut. 1996; 38: 714-718.
- Zimmerman DD, Gosselink MP, Mitalas LE, Delemarre JB, Hop WJ, Briel JW, et al. Smoking impairs rectal mucosal bloodflow--a pilot study: possible implications for transanal advancement flap repair. Dis Colon Rectum. 2005; 48: 1228-1232.
- Aldoori WH, Giovannucci EL, Rimm EB, Wing AL, Trichopoulos DV, Willett WC. A prospective study of alcohol, smoking, caffeine and the risk of symptomatic diverticular disease in men. Ann Epidemiol. 1995; 5: 221-228.
- Secretan B, Straif K, Baan R, Grosse Y, El Ghissassi F, Bouvard V, et al. A review of human carcinogens--Part E: tobacco, areca nut, alcohol, coal smoke, and salted fish. Lancet Oncol. 2009; 10: 1033-1034.
- International Agency for Research on Cancer: IARC Monograph on the Evaluation of Carcinogenic Risks to Humans. International Agency for Research on Cancer, Lyon, France. 2012.
- The Health Consequences of Smoking: 50 Years of Progress - A Report of the Surgeon General. National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. 2014.
- Tsoi KK, Pau CY, Wu WK, Chan FK, Griffiths S, Sung JJ. Cigarette smoking and the risk of colorectal cancer: a meta-analysis of prospective cohort studies. Clin Gastroenterol Hepatol. 2009; 7: 682-688. e1-5.
- Huxley RR, Ansary-Moghaddam A, Clifton P, Czernichow S, Parr CL, Woodward M. The impact of dietary and lifestyle risk factors on risk of colorectal cancer: a quantitative overview of the epidemiological evidence. Int J Cancer. 2009; 125: 171-180.
- Liang PS, Chen TY, Giovannucci E. Cigarette smoking and colorectal cancer incidence and mortality: systematic review and meta-analysis. Int J Cancer. 2009; 124: 2406-2415.
- Botteri E, Iodice S, Bagnardi V, Raimondi S, Lowenfels AB, Maisonneuve P. Smoking and colorectal cancer: a meta-analysis. Jama. 2008; 300: 2765-2778.
- Slattery ML, Curtin K, Anderson K, Ma KN, Ballard L, Edwards S, et al. Associations between cigarette smoking, lifestyle factors, and microsatellite instability in colon tumors. J Natl Cancer Inst. 2000; 92: 1831-1836.
- Chia VM, Newcomb PA, Bigler J, Morimoto LM, Thibodeau SN, Potter JD. Risk of microsatellite-unstable colorectal cancer is associated jointly with smoking and nonsteroidal anti-inflammatory drug use. Cancer Res. 2006; 66: 6877-6883.
- Poynter JN, Haile RW, Siegmund KD, Campbell PT, Figueiredo JC, Limburg P, et al. Associations between smoking, alcohol consumption, and colorectal cancer, overall and by tumor microsatellite instability status. Cancer Epidemiol Biomarkers Prev. 2009; 18: 2745-2750.
- Curtin K, Samowitz WS, Wolff RK, Herrick J, Caan BJ, Slattery ML. Somatic alterations, metabolizing genes and smoking in rectal cancer. Int J Cancer. 2009; 125: 158-164.
- Limsui D, Vierkant RA, Tillmans LS, Wang AH, Weisenberger DJ, Laird PW, et al. Cigarette smoking and colorectal cancer risk by molecularly defined subtypes. J Natl Cancer Inst. 2010; 102: 1012-1022.
- Samowitz WS, Albertsen H, Sweeney C, Herrick J, Caan BJ, Anderson KE, et al. Association of smoking, CpG island methylator phenotype, and V600E BRAF mutations in colon cancer. J Natl Cancer Inst. 2006; 98: 1731-1738.
- Nishihara R, Morikawa T, Kuchiba A, Lochhead P, Yamauchi M, Liao X, et al. A prospective study of duration of smoking cessation and colorectal cancer risk by epigenetics-related tumor classification. Am J Epidemiol. 2013; 178: 84-100.
- Chao A, Thun MJ, Jacobs EJ, Henley SJ, Rodriguez C, Calle EE. Cigarette smoking and colorectal cancer mortality in the cancer prevention study II. J Natl Cancer Inst. 2000; 92: 1888-96.
- Phipps AI, Baron J, Newcomb PA. Prediagnostic smoking history, alcohol consumption, and colorectal cancer survival: the Seattle Colon Cancer Family Registry. Cancer. 201; 117: 4948-4957.
- McCleary NJ, Niedzwiecki D, Hollis D, Saltz LB, Schaefer P, Whittom R, et al. Impact of smoking on patients with stage III colon cancer: results from Cancer and Leukemia Group B 89803. Cancer. 2010; 116: 957-966.
- Yang B, Jacobs EJ, Gapstur SM, Stevens V, Campbell PT. Active Smoking and Mortality among Colorectal Cancer Survivors: The Cancer Prevention Study II Nutrition Cohort. J Clin Oncol. 2015; 33: 885-893.
- Vincenzi B, Santini D, Loupakis F, Addeo R, Rojas Llimpe FL, Baldi GG, et al. Cigarettes smoking habit may reduce benefit from cetuximab-based treatment in advanced colorectal cancer patients. Expert Opin Biol Ther. 2009; 9: 945-949.
- Munro AJ, Bentley AH, Ackland C, Boyle PJ. Smoking compromises cause-specific survival in patients with operable colorectal cancer. Clin Oncol (R Coll Radiol). 2006; 18: 436-440.
- Chen AM, Chen LM, Vaughan A, Sreeraman R, Farwell DG, Luu Q, et al. Tobacco smoking during radiation therapy for head-and-neck cancer is associated with unfavorable outcome. Int J Radiat Oncol Biol Phys. 2011; 79: 414-419.
- Steinberger E, Kollmeier M, McBride S, Novak C, Pei X, Zelefsky MJ. Cigarette smoking during external beam radiation therapy for prostate cancer is associated with an increased risk of prostate cancer-specific mortality and treatment-related toxicity. BJU Int. 2015; 116: 596-603.
- Daling JR, Madeleine MM, Johnson LG, Schwartz SM, Shera KA, Wurscher MA, et al. Human papillomavirus, smoking, and sexual practices in the etiology of anal cancer. Cancer. 2004; 101: 270-280.
- Mai SK, Welzel G, Haegele V, Wenz F. The influence of smoking and other risk factors on the outcome after radiochemotherapy for anal cancer. Radiat Oncol. 2007; 2: 30.
- Ramamoorthy S, Luo L, Luo E, Carethers JM. Tobacco smoking and risk of recurrence for squamous cell cancer of the anus. Cancer Detect Prev. 2008; 32: 116-120.
- Glynne-Jones R, Nilsson PJ, Aschele C, Goh V, Peiffert D, Cervantes A, et al. Anal cancer: ESMO-ESSO-ESTRO clinical practice guidelines for diagnosis, treatment and follow-up. Eur J Surg Oncol. 2014; 40: 1165-1176.
- Calkins BM. A meta-analysis of the role of smoking in inflammatory bowel disease. Dig Dis Sci. 1989; 34: 1841-1854.
- Reese GE, Nanidis T, Borysiewicz C, Yamamoto T, Orchard T, Tekkis PP. The effect of smoking after surgery for Crohn's disease: a meta-analysis of observational studies. Int J Colorectal Dis. 2008; 23: 1213-1221.
- Buisson A, Chevaux JB, Allen PB, Bommelaer G, Peyrin-Biroulet L. Review article: the natural history of postoperative Crohn's disease recurrence. Aliment Pharmacol Ther. 2012; 35: 625-633.
- Papay P, Reinisch W, Ho E, Gratzer C, Lissner D, Herkner H, et al. The impact of thiopurines on the risk of surgical recurrence in patients with Crohn's disease after first intestinal surgery. Am J Gastroenterol. 2010; 105: 1158-1164.
- Unkart JT, Anderson L, Li E, Miller C, Yan Y, Gu CC, et al. Risk factors for surgical recurrence after ileocolic resection of Crohn's disease. Dis Colon Rectum. 2008; 51: 1211-1216.
- Cottone M, Rosselli M, Orlando A, Oliva L, Puleo A, Cappello M, et al. Smoking habits and recurrence in Crohn's disease. Gastroenterology. 1994; 106: 643-648.
- Martin G, Heyen F, Dube S. Factors of recurrence in Crohn disease. Ann Chi r. 1994; 48: 685-690.
- Timmer A, Sutherland LR, Martin F. Oral contraceptive use and smoking are risk factors for relapse in Crohn's disease. The Canadian Mesalamine for Remission of Crohn's Disease Study Group. Gastroenterology. 1998; 114: 1143-1150.
- Medina C, Vergara M, Casellas F, Lara F, Naval J, Malagelada JR. Influence of the smoking habit in the surgery of inflammatory bowel disease. Rev Esp Enferm Dig. 1998; 90: 771-778.
- O'Riordan JM, O'Connor BI, Huang H, Victor JC, Gryfe R, MacRae HM, et al. Long-term outcome of colectomy and ileorectal anastomosis for Crohn's colitis. Dis Colon Rectum. 2011; 54: 1347-1354.
- Liu Z, Lu H, Kiran RP, Ni R, Remzi FH, Shen B. Pre-colectomy appendectomy and risk for Crohn's disease in patients with ileal pouch-anal anastomosis. J Gastrointest Surg. 2012; 16: 1370-1378.
- Shen B, Remzi FH, Nutter B, Bennett AE, Lashner BA, Lavery IC, et al. Association between immune-associated disorders and adverse outcomes of ileal pouch-anal anastomosis. Am J Gastroenterol. 2009; 104: 655-664.
- Nunes T, Etchevers MJ, Domenech E, Garcia-Sanchez V, Ber Y, Penalva M, et al. Smoking does influence disease behaviour and impacts the need for therapy in Crohn's disease in the biologic era. Aliment Pharmacol Ther. 2013; 38: 752-760.
- Joyce MR, Hannaway CD, Strong SA, Fazio VW, Kiran RP. Impact of smoking on disease phenotype and postoperative outcomes for Crohn's disease patients undergoing surgery. Langenbecks Arch Surg. 2013; 398: 39-45.
- Lunney PC, Kariyawasam VC, Wang RR, Middleton KL, Huang T, Selinger CP, et al. Smoking prevalence and its influence on disease course and surgery in Crohn's disease and ulcerative colitis. Aliment Pharmacol Ther. 2015; 42: 61-70.
- Devaraj B, Khabassi S, Cosman BC. Recent smoking is a risk factor for anal abscess and fistula. Dis Colon Rectum. 2011; 54: 681-685.
- Zimmerman DD. The impact of smoking on perianal disease. Dis Colon Rectum. 2011; 54: 658-659.
- Vazquez BG, Alikhan A, Weaver AL, Wetter DA, Davis MD. Incidence of hidradenitis suppurativa and associated factors: a population-based study of Olmsted County, Minnesota. J Invest Dermatol. 2013; 133: 97-103.
- Konig A, Lehmann C, Rompel R, Happle R. Cigarette smoking as a triggering factor of hidradenitis suppurativa. Dermatology. 1999; 198: 261-264.
- Matusiak L, Bieniek A, Szepietowski JC. Hidradenitis suppurativa and associated factors: still unsolved problems. J Am Acad Dermatol. 2009; 61: 362-365.
- Jones RM. Smoking before surgery: the case for stopping. Br Med J (Clin Res Ed). 1985; 290: 1763-1764.
- Jorgensen LN, Kallehave F, Christensen E, Siana JE, Gottrup F. Less collagen production in smokers. Surgery. 1998; 123: 450-455.
- Nickelsen TN, Jorgensen T, Kronborg O. Lifestyle and 30-day complications to surgery for colorectal cancer. Acta Oncol. 2005; 44: 218-223.
- Jones JK, Triplett RG. The relationship of cigarette smoking to impaired intraoral wound healing: a review of evidence and implications for patient care. J Oral Maxillofac Surg. 1992; 50: 237-239.
- Silverstein P. Smoking and wound healing. Am J Med. 1992; 93: 22S-24S.
- Spear SL, Ducic I, Cuoco F, Hannan C. The effect of smoking on flap and donor-site complications in pedicled TRAM breast reconstruction. Plast Reconstr Surg. 2005; 116: 1873-1880.
- Sorensen LT, Hemmingsen U, Kallehave F, Wille-Jorgensen P, Kjaergaard J, Moller LN, et al. Risk factors for tissue and wound complications in gastrointestinal surgery. Ann Surg. 2005; 241: 654-658.
- Sorensen LT, Hemmingsen UB, Kirkeby LT, Kallehave F, Jorgensen LN. Smoking is a risk factor for incisional hernia. Arch Surg. 2005; 140: 119-123.
- Sorensen LT. Wound healing and infection in surgery: the pathophysiological impact of smoking, smoking cessation, and nicotine replacement therapy: a systematic review. Ann Surg. 2012; 255: 1069-1079.
- Dahl RM, Wetterslev J, Jorgensen LN, Rasmussen LS, Moller AM, Meyhoff CS. The association of perioperative dexamethasone, smoking and alcohol abuse with wound complications after laparotomy. Acta Anaesthesiol Scand. 2014; 58: 352-361.
- Moller AM, Pedersen T, Villebro N, Munksgaard A. Effect of smoking on early complications after elective orthopaedic surgery. J Bone Joint Surg Br. 2003; 85: 178-181.
- Walker NM, Morris SA, Cannon LB. The effect of pre-operative counselling on smoking patterns in patients undergoing forefoot surgery. Foot Ankle Surg. 2009; 15: 86-89.
- Moller AM, Maaloe R, Pedersen T. Postoperative intensive care admittance: the role of tobacco smoking. Acta Anaesthesiol Scand. 2001; 45: 345-348.
- Hawn MT, Houston TK, Campagna EJ, Graham LA, Singh J, Bishop M, et al. The attributable risk of smoking on surgical complications. Ann Surg. 2011; 254: 914-920.
- Gajdos C, Hawn MT, Campagna EJ, Henderson WG, Singh JA, Houston T. Adverse effects of smoking on postoperative outcomes in cancer patients. Ann Surg Oncol. 2012; 19: 1430-1438.
- Richards CH, Platt JJ, Anderson JH, McKee RF, Horgan PG, McMillan DC. The impact of perioperative risk, tumor pathology and surgical complications on disease recurrence following potentially curative resection of colorectal cancer. Ann Surg. 2011; 254: 83-89.
- Sharma A, Deeb AP, Iannuzzi JC, Rickles AS, Monson JR, Fleming FJ. Tobacco smoking and postoperative outcomes after colorectal surgery. Ann Surg. 2013; 258: 296-300.
- Artioukh DY, Smith RA, Gokul K. Risk factors for impaired healing of the perineal wound after abdominoperineal resection of rectum for carcinoma. Colorectal Dis. 2007; 9: 362-367.
- Sorensen LT, Karlsmark T, Gottrup F. Abstinence from smoking reduces incisional wound infection: a randomized controlled trial. Ann Surg. 2003; 238: 1-5.
- Abbas SM, Hill AG. Smoking is a major risk factor for wound dehiscence after midline abdominal incision; case-control study. ANZ J Surg. 2009; 79: 247-250.
- Finan KR, Vick CC, Kiefe CI, Neumayer L, Hawn MT. Predictors of wound infection in ventral hernia repair. Am J Surg. 2005; 190: 676-681.
- Fischer JP, Wink JD, Nelson JA, Kovach SJ, 3rd. Among 1,706 cases of abdominal wall reconstruction, what factors influence the occurrence of major operative complications? Surgery. 2014; 155: 311-319.
- Breuing K, Butler CE, Ferzoco S, Franz M, Hultman CS, Kilbridge JF, et al. Incisional ventral hernias: review of the literature and recommendations regarding the grading and technique of repair. Surgery. 2010; 148: 544-558.
- Oh C. Management of high recurrent anal fistula. Surgery. 1983; 93: 330-332.
- Aguilar PS, Plasencia G, Hardy TG, Jr., Hartmann RF, Stewart WR. Mucosal advancement in the treatment of anal fistula. Dis Colon Rectum. 1985; 28: 496-498.
- Wedell J, Meier zu Eissen P, Banzhaf G, Kleine L. Sliding flap advancement for the treatment of high level fistulae. Br J Surg. 1987; 74: 390-391.
- Kodner IJ, Mazor A, Shemesh EI, Fry RD, Fleshman JW, Birnbaum EH. Endorectal advancement flap repair of rectovaginal and other complicated anorectal fistulas. Surgery. 1993; 114: 682-689.
- Schouten WR, Zimmerman DD, Briel JW. Transanal advancement flap repair of transsphincteric fistulas. Dis Colon Rectum. 1999; 42: 1419-1122.
- Ozuner G, Hull TL, Cartmill J, Fazio VW. Long-term analysis of the use of transanal rectal advancement flaps for complicated anorectal/vaginal fistulas. Dis Colon Rectum. 1996; 39: 10-14.
- Zimmerman DD, Delemarre JB, Gosselink MP, Hop WC, Briel JW, Schouten WR. Smoking affects the outcome of transanal mucosal advancement flap repair of trans-sphincteric fistulas. Br J Surg. 2003; 90: 351-354.
- Ellis CN, Clark S. Effect of tobacco smoking on advancement flap repair of complex anal fistulas. Dis Colon Rectum. 2007; 50: 459-463.
- van der Hagen SJ, Baeten CG, Soeters PB, van Gemert WG. Staged Mucosal Advancement Flap versus Staged Fibrin Sealant in the Treatment of Complex Perianal Fistulas. Gastroenterol Res Pract. 2011: 186350: 7.
- Tuson JR, Everett WG. A retrospective study of colostomies, leaks and strictures after colorectal anastomosis. Int J Colorectal Dis. 1990; 5: 44-48.
- Bokey EL, Chapuis PH, Fung C, Hughes WJ, Koorey SG, Brewer D, et al. Postoperative morbidity and mortality following resection of the colon and rectum for cancer. Dis Colon Rectum. 1995; 38: 480-486.
- Iancu C, Mocan LC, Todea-Iancu D, Mocan T, Acalovschi I, Ionescu D, et al. Host-related predictive factors for anastomotic leakage following large bowel resections for colorectal cancer. J Gastrointestin Liver Dis. 2008; 17: 299-303.
- Moran BJ, Heald RJ. Risk factors for and management of anastomotic leakage in rectal surgery. Colorectal Dis. 2001; 3: 135-137.
- Kruschewski M, Rieger H, Pohlen U, Hotz HG, Buhr HJ. Risk factors for clinical anastomotic leakage and postoperative mortality in elective surgery for rectal cancer. Int J Colorectal Dis. 2007; 22: 919-927.
- Kim MJ, Shin R, Oh HK, Park JW, Jeong SY, Park JG. The impact of heavy smoking on anastomotic leakage and stricture after low anterior resection in rectal cancer patients. World J Surg. 2011; 35: 2806-2810.
- Sorensen LT, Jorgensen T, Kirkeby LT, Skovdal J, Vennits B, Wille-Jorgensen P. Smoking and alcohol abuse are major risk factors for anastomotic leakage in colorectal surgery. Br J Surg. 1999; 86: 927-931.
- Richards CH, Campbell V, Ho C, Hayes J, Elliott T, Thompson-Fawcett M. Smoking is a major risk factor for anastomotic leak in patients undergoing low anterior resection. Colorectal Dis. 2012; 14: 628-633.
- McDermott FD, Heeney A, Kelly ME, Steele RJ, Carlson GL, Winter DC. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br J Surg. 2015; 102: 462-479.
- Baucom RB, Poulose BK, Herline AJ, Muldoon RL, Cone MM, Geiger TM. Smoking as dominant risk factor for anastomotic leak after left colon resection. Am J Surg. 2015; 210: 1-5.
- Jannasch O, Klinge T, Otto R, Chiapponi C, Udelnow A, Lippert H, et al. Risk factors, short and long term outcome of anastomotic leaks in rectal cancer. Oncotarget. 2015; 6: 36884-36893.
- Wong J, Lam DP, Abrishami A, Chan MT, Chung F. Short-term preoperative smoking cessation and postoperative complications: a systematic review and meta-analysis. Can J Anaesth. 2012; 59: 268-279.
- Nayan S, Gupta MK, Strychowsky JE, Sommer DD. Smoking cessation interventions and cessation rates in the oncology population: an updated systematic review and meta-analysis. Otolaryngol Head Neck Surg. 2013; 149: 200-211.
- Bartsch RH, Weiss G, Kastenbauer T, Patocka K, Deutinger M, Krapohl BD, et al. Crucial aspects of smoking in wound healing after breast reduction surgery. J Plast Reconstr Aesthet Surg. 2007; 60: 1045-1049.