Research Article
Learning Curve of Robot-Assisted Laparoscopy in Gynecologic Oncology Surgery: Team Training and Impact on Morbidity
Jauffret C1*, Lambaudie E1, Esterni B2, Bannier M1, Buttarelli M1 and Houvenaeghel G1
1Department of Surgical Oncology, Paoli-Calmettes Institute, France
2Department of Biostatistics Unit, Paoli-Calmettes Institute, France
*Corresponding author: Camille Jauffret, Department of Surgical Oncology, Paoli Calmettes Institute, 232 Bd Sainte Marguerite, 13009 Marseille, France
Published: 14 Sep, 2016
Cite this article as: Jauffret C, Lambaudie E, Esterni B,
Bannier M, Buttarelli M, Houvenaeghel
G. Learning Curve of Robot-Assisted
Laparoscopy in Gynecologic Oncology
Surgery: Team Training and Impact on
Morbidity. Clin Surg. 2016; 1: 1112.
Abstract
Objectives: The aim of this study was to evaluate the learning curve of robot-assisted laparoscopy
for gynecologic oncologic surgical procedures, regarding post-operative morbidity.
Methods: Between February 2007 and October 2010, 225 robot-assisted procedures has been
performed by a single team specialized in gynecologic oncology. Dates were prospectively collected.
Isolated and poorly reproducible procedures were excluded, so a total of 187 procedures were finally
included to assess the learning curve. Three parameters have been used: overall rate of postoperative
complications, operative time and number of lymph nodes.
CUSUM statistical analysis was used to investigate the learning curve. After determining the number
of cases necessary for learning, a comparative analysis was conducted to compare the learning
period to the efficiency period.
Results: Fifty cases were necessary to master postoperative complications. The operative time evolves
in three phases: the learning phase, with a gradual decrease in operative time; the consolidation
phase, steady; and the final phase, increasing, corresponding to the introduction of more difficult
cases and to the beginning of training program. There was a growing trend in the number of nodes
removed. Comparative analysis of the learning period (senior surgeons) and the efficiency period
(with increasing involvement of juniors) showed a significant evolution of procedures, and did not
reveal any difference in terms of complications (44% vs. 31%, non significant), or mean number of
lymph nodes removed (12.3 vs. 13.2, non significant).
Conclusion: Team training of robot-assisted laparoscopy requires an average of fifty cases looking
at per operative complications.
Keywords: Robot-assisted laparoscopy; Gynecologic; Oncology
Introduction
Conventional laparoscopy has demonstrated its interest in various gynecologic surgical
procedures and particularly in oncology. But this minimally invasive approach presents several
difficulties due to its relatively long learning curve, which can explain its slow development in our
countries.
In this context, in order to overcome the difficulties, robotic assistance appeared in 1994, with
the development of the first robotic arm holding a camera (AESOP, Computer Motion, California,
1994). Later, in the 2000s, the 3-armed Zeus robot (Computer Motion, California) and the Da Vinci
robot (Intuitive Surgical, California) appeared. This new robot-assisted technique is currently being
evaluated, both clinically and financially. This evolution of conventional laparoscopy has its own
learning curve which should be evaluated.
Objective
The aim of our study was to evaluate the learning curve of a single surgical team, considering all gynecologic oncology procedures conducted by robot-assisted laparoscopy, since the beginning of our robotic program. The primary endpoint was postoperative morbidity, and secondary endpoints were operative time and number of lymph node removed.
Table 1
Materials and Methods
Patients
From February 2007 to October 2010, 193 patients underwent
225 robotic assisted laparoscopic procedures in our institution (Paoli-
Calmettes Institute, Marseille, France). Six surgeons, three seniors
and three fellows in training performed all the procedures.
Data collection
Socio-demographic, clinical, and intraoperative data were
prospectively collected, including type of surgery, docking time,
operative time, intraoperative complications, blood loss and
transfusions, conversion to conventional laparoscopy or laparotomy,
and definitive pathology.
Operative time was defined as the time elapsed between the first
incision and the end of the skin suture. Thus, it also included robot
docking time and surgeon console time.
The anatomopathological data collected were histological type
and tumor size, margin status, number of lymph nodes removed, and
lymph node involvement.
Postoperative complications were prospectively collected.
Postoperative morbidity was graded according to the Clavien and
Dindo’s classification [1]. Minor postoperative complications were
defined by a grade 1 or 2, and severe postoperative complications by
a grade 3 or 4.
Patient follow-up was ensured by a systematic visit at one month,
six months, and then annually. Late post operative complications and
recurrence rate has been recorded.
Learning curve analysis
We decided not to take into account exceptional or poorly
reproducible interventions, such as pelvectomy, isolated lymph
node removal, or ovarian cancer restaging. A total of 187 cases were
analyzed and divided into 5 homogenous sub groups in terms of
indications and surgical procedures (Table 1).
The effect of learning was studied by analyzing the following
criteria: postoperative morbidity, severe postoperative morbidity,
operative time, and number of pelvic lymph nodes removed. We
chose the number of complications as a primary endpoint. The
conversion to laparotomy was not included as a learning factor
because it was a rare event in our series. Intraoperative data were
collected individually by each surgeon and were not exhaustive.
Thus, in the data collected, blood loss information was too missing
to be analyzed. The extent of bleeding during the intervention was
then assessed by the rate of transfusion. Concerning oncological
outcomes, we did not considered recurrence rate and overall survival
in our analysis, because our robotic program only began 4 years ago.
Our objective was to determine the learning curve of the surgical
team, and not to study individual learning curve. Surgical procedures
were analyzed according to whether they were performed by a senior
surgeon, or a junior surgeon associated to a senior. The analysis was
not performed in previously determined periods, but by observing the
learning curve, defined by the complications rate. A shift in the curve
was considered the end of the learning period. To define the learning
period, we decided to follow the evolution of the complications rate.
Statistical analysis
To study the learning curve, a univariate analysis of factors
deemed relevant a priori was first conducted on the rate of surgical
complications, the operative time, and the number of pelvic lymph
nodes removed. These factors were related to the patient (age, BMI), the
intervention performed (indication, procedure, chemoradiotherapy
antecedents), and the surgeon (intervention conducted by a senior
surgeon, or “accompanied” intervention conducted in whole or in
part by a junior surgeon, under supervision of a senior surgeon).
The cumulative sum technique (CUSUM) was used for continuous
variables (operative time and number of pelvic lymph nodes removed),
and for binary variables (overall postoperative morbidity and severe
postoperative morbidity) [2-4]. It consists in expressing each case by
the difference between the value of a measured parameter (operative
time in minutes, or presence/absence of complications) and the mean
of the entire series. The cases were first classified in chronological
order, depending on the date of surgery.
Regarding continuous variables, the point representing the
1st case is the difference between the operating time of the 1st case
and the mean operating time of the entire series. To represent the
2nd case, we calculated the difference between the operating time of
the 2nd case and the mean operating time of the entire series, added
to the previous value, and so on, until the last case, for which the
CUSUM value is necessarily 0. Regarding binary variables such as
complications, the curve moves upward for each complication, and
downward for each case free of complications. The end of the learning
process was defined as the point where the curve ceases to rise, and
stabilizes or descends.
Finally, Fischer and X2 tests were used to compare clinical data,
type of procedure, complication rates, operative times, participation
of surgeons in training, and number of lymph nodes removed between
the so-called “learning period” and the so-called “efficiency period.”
Ethics statement
Prior to analysis, patients were anonymized. Neither their names
nor their initials were recorded, but a number was assigned to each. At
the time of the first admission, all patients provided written consent
to the use of their clinical and histological data by anonymous way.
Our institutional review board (COS, Comitéd’ Orientation
Stratégique) approved the study and the consent procedure.
Table 2
Results
Descriptive results
To analyze the learning curve of 187 surgical procedures we
defined five homogenous sub groups as follow:
Group 1 (n=46): Adjuvant surgery for locally advanced cervical
cancer after para-aortic lymph node staging (surgical in 85% of
cases), concomitant chemoradiotherapy and brachytherapy. In this
subgroup, patients underwent an extra facial hysterectomy + bilateral
oophorectomy + pelvic lymphadenectomy, Most patients were
treated for stage IB2 (48 %) or IIB (28 %).
Group 2 (n=41): Colpohysterectomy + bilateral oophorectomy +
pelvic lymphadenectomy for endometrial or cervical cancer (less than
4 cm), before treatment, or after brachytherapy for cervical cancers.
Group 3 (n=54): Hysterectomy + bilateral oophorectomy without
lymph node removal and without prior treatment (intraepithelial
cervical neoplasia, fibromas, ovarian cysts, and endometrial or
cervical cancers).
Group 4 (n=17): Pelvic lymphadenectomy.
Group 5 (n=32): Para-aortic lymphadenectomy (retroperitoneal
or transperitoneal).
Clinical and intraoperative data are reported in Table 1.
Two intraoperative complications occurred during two paraaortic
lymphadenectomies: an inferior mesenteric artery injury
controlled by suture laparoscopically, and a bowel injury (duodenojejunal
angle), with a conversion to open.
The median hospital stay in each group, as well as the rate
of postoperative complications, is summarized in Table 2. The
overall postoperative complications rate was 35%, including
lymphatic complications rate (20%). Seventeen severe postoperative
complications were described (9%). They were mainly infected or
symptomatic lymphoceles requiring radiological drainage.
In our series, the most important complications rate was observed
in the sub group 1 (66%).
Postoperative morbidity
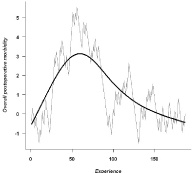
Regarding the overall rate of postoperative complications,
the CUSUM curve revealed a significant decrease in the rate of
complications beyond the 50th procedure (Figure 1).
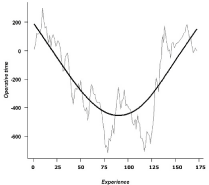
Operative time
From the CUSUM curve representing the evolution of operative
time, three phases were identified: a first phase going downward,
corresponding to the learning stage; a consolidation phase, with a
plateau phase; and an ascending phase, corresponding in our activities
to the inclusion of more challenging cases and to the beginning of
training program for our 3 fellows (Figure 2).
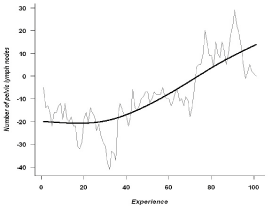
Number of pelvic lymph nodes removed
The mean number of lymph nodes removed during pelvic
lymphadenectomies (104 procedures) was 12.8 (4 – 33) (Table 3),
The CUSUM analysis showed an increase mean lymph node number,
without reaching any specific threshold (Figure 3).
Comparisons between the “learning period” and the
“efficiency period”
Looking at the curve representing the overall complications rate,
we defined the point of inflection of the curve (50th case), as the end
of the “learning period”. The following cases (n=137) represented
the “efficiency period.” Comparative analysis of these two periods
is reported in Table 3. There was no difference regarding age, BMI,
and the patients’ history of radio- and chemotherapy. The indications
and the integration of fellows were significantly different between
these two periods. Indeed, during the learning period, all surgical
procedures were performed by experienced surgeons in conventional
laparoscopy, whereas 36% of subsequent procedures included
fellows. We did not observe any significative difference concerning
complications rate between the 2 periods (44% vs. 31%), the rate
decreased by 44% during the learning period, to 31% thereafter (nonsignificant).
The mean operative time was 161.6 and 171.7 minutes,
respectively (non-significant). There was no significant difference
in the number of pelvic lymph nodes removed. Two patients in the
series were transfused: one during the learning period and one during
the efficiency period. The length of stay was also similar in the two
groups.
Figure 1
Figure 2
Table 3
Figure 3
Discussion
The concept of minimally invasive surgery in gynecology emerged
in the 1980s and has been progressively developed in gynecologic
oncology surgery. Conventional laparoscopy is now validated for
various procedures, and its advantages compared to open surgery
are now well described (less blood loss, less postoperative pain, and
reduced hospital stay with a faster recovery, and better quality of life)
[5-16]. However, laparoscopy has several limitations, such as the twodimensional
view, the image instability, and the instruments, which
offer only four degrees of freedom. These limitations are associated
with a specific and longer learning curve compared to open surgery,
and may limit the development of this minimally invasive approach.
Robotic-assisted laparoscopy emerged in 2000, and has gradually
expanded to the field of pelvic surgery. On July 11, 2000, the Food and
Drug Administration approved robot-assisted laparoscopy for pelvic
surgery, and specifically approved it for hysterectomy in 2005. It has
been validated by numerous comparative studies [13,17-28]. Robotic
assistance was developed to overcome the drawbacks of conventional
laparoscopy, by allowing 3D visualization, tremor filtration, increased
accuracy (thanks to motion scaling), ergonomics, allowing surgeon
performance optimization; and low intra-abdominal pressure.
However, this new robotic tool requires specific training. Indeed,
the surgeon must acquire a control on several elements associated
with robot-assisted surgery: he must learn to incorporate the spatial
relationships of the instruments outside the operative field to mobilize
them while minimizing conflicts between the robot’s arms; master the
operation of the robot by combining specific manual controls with
pedal controls; and compensate for the loss of force feedback through
a visual assessment of the tension applied to the tissue.
Choice of statistical method
We chose the CUSUM method to express our results. This is a
method adopted by the medical profession in the 1970s [2-4,33].
It allowed us to express our results in deviations from the mean,
providing data non-identified by other approaches (such as the
comparison of chronological cases split into predefined segments).
Similarly, Bokhari et al. [34] in 2010 investigated robot-assisted
laparoscopy learning curve in rectosigmoid and rectal surgery. Three
phases of learning on a CUSUM curve representing the operative
time was demonstrated: the learning phase, the consolidation phase,
and the expertise phase. Duration of hospitalization was stable, setup
time and blood loss decreased, and operative time increased (which
they attributed to the management of increasingly difficult cases).
In our study, the curve representing operative time shows the
same triphasic aspect: the first phase represent the learning stage,
with an improvement in operative time throughout the procedures;
the second phase correspond to the consolidation of this learning;
and the third phase, going upward, correspond to the contribution
of junior training surgeons and the inclusion of more challenging
cases. Indeed, the majority of cervical and endometrial cancers were
included during the second phase of the study (Table 4).
Choice of primary endpoint
Unlike most studies, which analyze preferentially operative
time, we chose the rate of complications as the primary endpoint.
This could be more relevant and more representative of mastery of
surgical procedure, given the heterogeneity of ourserie. This have
been previously reported in conventionnal laparoscopy by Altgassen
et al. [35] in 2004.
Learning curve
Several studies have been published concerning the learning
curve in robotic surgery. Seamon et al. [38] in 2008, in a descriptive
analysis of their 92 first interventions for endometrial cancer, noted a
threshold at the 20th case (considering the evolution of operative time).
They compared the learning phase to the next phase. They didn’t find
any difference in the number of lymph nodes removed or blood loss.
No comparison was done considering the rate of complications. In
2009, the same authors reported a prospective analysis on 79 cases
of endometrial cancer, carried out by two surgeons. Interventions
were splited into chronological quartiles of 20 cases (because of the
threshold observed in the preliminary study). There was a difference
in terms of operative time when comparing the 20 first cases to the
20 next cases, which was not found in subsequent cases. There was a
significant difference regarding blood loss, number of lymph nodes
removed, and duration of hospitalization [39].
In 2009, Bell et al. [40] published a retrospective review of the
first 100 robot-assisted laparoscopic hysterectomies performed by
a single surgeon. Cases were analysed after division into quartiles
of 20. No significant difference was noted in operative time. The
rate of complications was 15% for the first 20 cases, and 5% for the
following cases, although this difference was not significant. Lim et
al. [41] in 2010 compared the 122 first robot-assisted procedures for
endometrial cancer, to a historical cohort of the 122 first procedures
by conventional laparoscopy. Learning curve was 24 cases for
robotic assistance, against 49 cases for conventional laparoscopy.
Comparisons before and after this threshold did not show any
difference in conventional laparoscopy, and showed increased
operative time and blood loss during robot-assisted laparoscopy
learning.
Robot-assisted laparoscopy was evaluated in other specialties,
and notably in urology. Patel et al. [42] analyzed data from the
200 first prostate cancer procedures, managed by two surgeons. A
progressive decrease was noted in operative time and blood loss, and
they described a learning curve of 20 to 25 cases.
In our study, 50 procedures were necessary to observe a threshold
in the rate of complications. This is reflecting a longer learning curve
than most published studies. This may be due to the fact that we
chose the rate of complications as our primary endpoint (instead of
operative time), or because the patients carried out by our team were
mainly affected by malignant tumors, with a majority of advanced
stages of cervical cancer. Moreover, we analyzed the learning of a
whole team, and not individual surgeons, and we analyzed several
different procedures, including after preliminary treatment with
concomitant chemoradiotherapy. Similarly, Lenihan et al. [36]
reported the learning curve of a two gynecological surgeon’s team by
analyzing data from all procedures performed with robotic assistance
and for various benign pathologies. They analyzed 113 procedures
and concluded that the learning curve required 50 cases, in agreement
with our results.
Junior’s contribution
During the first phase of our study, there was no accompanied
procedure.Our training program began at the sixty fourth procedures,
according to a step by steplearning method: the trainee performs one
step of the procedure, and when this step is mastered, he performs
the next one.
Comparative analysis of these two periods - learning and
efficiency - is therefore similar to analyzing the effects of the training
surgeons’ involvement. However, we didn’t find any difference
regarding the rate of complications, transfusions, and number of
lymph nodes removed. Operative time increased but this wasn’t
significant, and probably due, as mentioned before, to the inclusion
of more challenging cases, but also to the implication of more junior
surgeons (Table 4).
In our experience, robot-assisted surgery can thus be taught
without risk and without impairing the quality of the procedures. The
curve corresponding to the number of pelvic lymph nodes removed
even showed an increasing trend. This suggests that there is a positive
effect of the senior surgeons’ experience on the performance of their
students. Once the team training is achieved, fellowship is sufficient
to train juniors without increased risk or decreased quality of surgery.
Ali et al. [37] have studied this, in 2007. Learning curve in robotassisted
laparoscopy was evaluated by analysis of the performance of
the junior staff during bariatric surgery. Three phases were identified,
and the junior was the main surgeon during the first phase, then for
the two first phases, and then for all three phases. These procedures
were compared to data from the original series on senior surgeons.
There was no difference in total time for all three phases, no matter
the junior’s contribution. More importantly, there was no difference
in operative time when the three phases were performed by the
junior, in comparison with the original series. Similarly, during
“accompanied” procedures, where a senior surgeon performs one
or more phases, operative times were shorter than in the original
series (seniors). Moreover, robot docking time was better than in the
original series, reflecting the learning of the entire team, including
nurses, anesthesiologists, etc.
A prospective study of the learning curve in robot-assisted
laparoscopy of training surgeons is currently in progress in our team.
A standardization of procedures seems interesting to accelerate and
evaluate learning. This standardization is an unavoidable step to teach
robot-assisted laparoscopy (because of the impossibility to do “four
hands surgery”), unlikely to conventional laparoscopy. This includes
standardization of pre operative patient care, patient positioning,
position of trocars, and surgical procedures. This standardization
might be one of the reasons why there is no increase in morbidity
when involving junior surgeons.
Reducing the learning curve would allow access to minimally
invasive surgery to an increasing number of patients. Indeed, only
20% of patients eligible for this approach are actually supported by
laparoscopy in France (data from the Program for Medicalization
of Information Systems). Robotic assistance, once its cost is
overcome, can be the answer to this too-limited access to minimally
invasive approaches. The advantages of robot-assisted compared to
conventional laparoscopy are currently under evaluation in France in
a randomized trial (PHRC ROBO-GYN).
Conclusion
Robot-assisted laparoscopy appears to be an evolution of
minimally invasive surgery, with an important place in gynecologic
oncology. While some parameters, such as financial impact and
oncologic follow-up remain to be evaluated, it appears that this
technique is feasible in the majority of gynecologic oncology
procedures, and could give access to minimally invasive surgery to an
increasing number of patients.
This study suggests that team training needs 50 cases to master
the rate of complications, and to allow integration of juniors and
advanced procedures without negative impact on level of safety and
quality of results. A prospective validation of this learning curve is
necessary to confirm these data.
References
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004; 240: 205-213.
- Chaput de Saintonge, DM Vere WD. Why don't doctors use CUSUM's? Lancet. 1974; 1: 120-121.
- Tekkis PP, Senagore AJ, Delaney CP, Fazio VW. Evaluation of the learning curve in laparoscopic colorectal surgery: comparison of right-sided and left-sided resections. Ann Surg. 2005; 242: 83-91.
- Wohl H. The cusum plot: its utility in the analysis of clinical data. N Engl J Med. 1977; 296: 1044-1045.
- Colombo PE, Bertrand MM, Gutowski M, Mourregot A, Fabbro M, Saint-Aubert B, et al. Total laparoscopic radical hysterectomy for locally advanced cervical carcinoma (stages IIB, IIA and bulky stages IB) after concurrent chemoradiation therapy: surgical morbidity and oncological results. Gynecol Oncol. 2009; 114: 404-409.
- Frumovitz M, dos Reis R, Sun CC, Milam MR, Bevers MW, Brown J, et al. Comparison of total laparoscopic and abdominal radical hysterectomy for patients with early-stage cervical cancer. Obstet Gynecol. 2007; 110: 96-102.
- Ghezzi F, Cromi A, Ciravolo G, Volpi E, Uccella S, Rampinelli F, et al. Surgicopathologic outcome of laparoscopic versus open radical hysterectomy. Gynecol Oncol. 2007; 106: 502-506.
- Malzoni M, Tinelli R, Cosentino F, Perone C, Vicario V. Feasibility, morbidity, and safety of total laparoscopic radical hysterectomy with lymphadenectomy: our experience. J Minim Invasive Gynecol. 2007; 14: 584-590.
- Morgan DJ, Hunter DC, McCracken G, McClelland HR, Price JH, Dobbs SP. Is laparoscopically assisted radical vaginal hysterectomy for cervical carcinoma safe? A case control study with follow up. BJOG. 2007; 114: 537-542.
- Sharma R, Bailey J, Anderson R, Murdoch J. Laparoscopically assisted radical vaginal hysterectomy (Coelio-Schauta): A comparison with open Wertheim/Meigs hysterectomy. Int J Gynecol Cancer. 2006; 16: 1927-1932.
- Uccella S, Laterza R, Ciravolo G, Volpi E, Franchi M, Zefiro F, et al. A comparison of urinary complications following total laparoscopic radical hysterectomy and laparoscopic pelvic lymphadenectomy to open abdominal surgery. Gynecol Oncol. 2007; 107: S147-S149.
- Bell MC, Torgerson J, Seshadri-Kreaden U, Suttle AW, Hunt S. Comparison of outcomes and cost for endometrial cancer staging via traditional laparotomy, standard laparoscopy and robotic techniques. Gynecol Oncol. 2008; 111: 407-411.
- Boggess JF, Gehrig PA, Cantrell L, Shafer A, Ridgway M, Skinner EN, et al. A comparative study of 3 surgical methods for hysterectomy with staging for endometrial cancer: robotic assistance, laparoscopy, laparotomy. Am J Obstet Gynecol. 2008; 199: 360. e1-9.
- Malur S, Possover M, Michels W, Schneider A. Laparoscopic-assisted vaginal versus abdominal surgery in patients with endometrial cancer--a prospective randomized trial. Gynecol Oncol. 2001; 80: 239-244.
- Malzoni M, Tinelli R, Cosentino F, Perone C, Rasile M, Iuzzolino D, et al. Total laparoscopic hysterectomy versus abdominal hysterectomy with lymphadenectomy for early-stage endometrial cancer: a prospective randomized study. Gynecol Oncol. 2009; 112: 126-133.
- Veljovich DS, Paley PJ, Drescher CW, Everett EN, Shah C, Peters WA, 3rd. Robotic surgery in gynecologic oncology: program initiation and outcomes after the first year with comparison with laparotomy for endometrial cancer staging. Am J Obstet Gynecol. 2008; 198: 679 e1-9.
- Boggess JF, Gehrig PA, Cantrell L, Shafer A, Ridgway M, Skinner EN, et al. A case-control study of robot-assisted type III radical hysterectomy with pelvic lymph node dissection compared with open radical hysterectomy. Am J Obstet Gynecol. 2008; 199: 357. e1-7.
- Estape R, Lambrou N, Diaz R, Estape E, Dunkin N, Rivera A. A case matched analysis of robotic radical hysterectomy with lymphadenectomy compared with laparoscopy and laparotomy. Gynecol Oncol. 2009; 113: 357-361.
- Ko EM, Muto MG, Berkowitz RS, Feltmate CM. Robotic versus open radical hysterectomy: a comparative study at a single institution. Gynecol Oncol. 2008; 111: 425-430.
- Magrina JF, Kho RM, Weaver AL, Montero RP, Magtibay PM. Robotic radical hysterectomy: comparison with laparoscopy and laparotomy. Gynecol Oncol. 2008; 109: 86-91.
- Nam EJ, Kim SW, Kim S, Kim JH, Jung YW, Paek JH, et al. A case-control study of robotic radical hysterectomy and pelvic lymphadenectomy using 3 robotic arms compared with abdominal radical hysterectomy in cervical cancer. Int J Gynecol Cancer. 2010; 20: 1284-1289.
- Nezhat F. Minimally invasive surgery in gynecologic oncology: laparoscopy versus robotics. Gynecol Oncol. 2008; 111: S29-S32.
- Persson J, Reynisson P, Borgfeldt C, Kannisto P, Lindahl B, Bossmar T. Robot assisted laparoscopic radical hysterectomy and pelvic lymphadenectomy with short and long term morbidity data. Gynecol Oncol. 2009; 113: 185-190.
- Cardenas-Goicoechea J, Adams S, Bhat SB, Randall TC. Surgical outcomes of robotic-assisted surgical staging for endometrial cancer are equivalent to traditional laparoscopic staging at a minimally invasive surgical center. Gynecol Oncol. 2010; 117: 224-228.
- Gehrig PA, Cantrell LA, Shafer A, Abaid LN, Mendivil A, Boggess JF. What is the optimal minimally invasive surgical procedure for endometrial cancer staging in the obese and morbidly obese woman? Gynecol Oncol. 2008; 111: 41-45.
- Paley PJ, Veljovich DS, Shah CA, Everett EN, Bondurant AE, Drescher CW, et al. Surgical outcomes in gynecologic oncology in the era of robotics: analysis of first 1000 cases. Am J Obstet Gynecol. 2011; 204: 551.e1-9.
- Seamon LG, Cohn DE, Henretta MS, Kim KH, Carlson MJ, Phillips GS, et al. Minimally invasive comprehensive surgical staging for endometrial cancer: Robotics or laparoscopy? Gynecol Oncol. 2009; 113: 36-41.
- Reza M, Maeso S, Blasco JA, Andradas E. Meta-analysis of observational studies on the safety and effectiveness of robotic gynaecological surgery. Br J Surg. 2010; 97: 1772-1783.
- Prasad SM, Maniar HS, Soper NJ, Damiano RJ, Jr., Klingensmith ME. The effect of robotic assistance on learning curves for basic laparoscopic skills. Am J Surg. 2002; 183: 702-707.
- Yohannes P, Rotariu P, Pinto P, Smith AD, Lee BR. Comparison of robotic versus laparoscopic skills: is there a difference in the learning curve? Urology. 2002; 60: 39-45.
- Chang L, Satava RM, Pellegrini CA, Sinanan MN. Robotic surgery: identifying the learning curve through objective measurement of skill. Surg Endosc. 2003; 17: 1744-1748.
- Geller EJ, Schuler KM, Boggess JF. Robotic surgical training program in gynecology: how to train residents and fellows. J Minim Invasive Gynecol. 2011; 18: 224-229.
- Bege T, Lelong B, Esterni B, Turrini O, Guiramand J, Francon D, et al. The learning curve for the laparoscopic approach to conservative mesorectal excision for rectal cancer: lessons drawn from a single institution's experience. Ann Surg. 2010; 251: 249-253.
- Bokhari MB, Patel CB, Ramos-Valadez DI, Ragupathi M, Haas EM. Learning curve for robotic-assisted laparoscopic colorectal surgery. Surg Endosc. 2011; 25: 855-860.
- Altgassen C, Michels W, Schneider A. Learning laparoscopic-assisted hysterectomy. Obstet Gynecol. 2004; 104: 308-313.
- Lenihan JP, Jr., Kovanda C, Seshadri-Kreaden U. What is the learning curve for robotic assisted gynecologic surgery? J Minim Invasive Gynecol. 2008; 15: 589-594.
- Ali MR, Rasmussen J, BhaskerRao B. Teaching robotic surgery: a stepwise approach. Surg Endosc. 2007; 21: 912-915.
- Seamon LG, Cohn DE, Richardson DL, Valmadre S, Carlson MJ, Phillips GS, et al. Robotic hysterectomy and pelvic-aortic lymphadenectomy for endometrial cancer. Obstet Gynecol. 2008; 112: 1207-1213.
- Seamon LG, Fowler JM, Richardson DL, Carlson MJ, Valmadre S, Phillips GS, et al. A detailed analysis of the learning curve: robotic hysterectomy and pelvic-aortic lymphadenectomy for endometrial cancer. Gynecol Oncol. 2009; 114: 162-167.
- Bell MC, Torgerson JL, Kreaden U. The first 100 da Vinci hysterectomies: an analysis of the learning curve for a single surgeon. S D Med. 2009; 62: 91, 3-5.
- Lim PC, Kang E, Park do H. A comparative detail analysis of the learning curve and surgical outcome for robotic hysterectomy with lymphadenectomy versus laparoscopic hysterectomy with lymphadenectomy in treatment of endometrial cancer: a case-matched controlled study of the first one hundred twenty two patients. Gynecol Oncol. 2011; 120: 413-418.
- Patel VR, Tully AS, Holmes R, Lindsay J. Robotic radical prostatectomy in the community setting--the learning curve and beyond: initial 200 cases. J Urol. 2005; 174: 269-272.