Case Report
Giant Malignant Melanomas and their Clinical Implications: Review of Literature and Case Report
Gishen K1, Maria DJP2, Khanlari M3, Cho-Vega HJ3, Feun L2, Thaller S1 and Möller MG1*
1Department of Surgery, University of Miami - Miller School of Medicine, USA
2Department of Medicine, University of Miami - Miller School of Medicine, USA
3Department of Pathology, University of Miami - Miller School of Medicine, USA
*Corresponding author: Mecker G Möller, Department of Surgery, Division of Surgical Oncology, University of Miami - Miller School of Medicine, Sylvester Comprehensive Cancer Center, 1120 NW 14th Street, CRB C232 Miami, Florida 33136, USA
Published: 01 Sep, 2016
Cite this article as: Gishen K, Maria DJP, Khanlari M, ChoVega HJ, Feun L, Thaller S, et al. Giant Malignant Melanomas and their Clinical Implications: Review of Literature and Case Report. Clin Surg. 2016; 1: 1096.
Abstract
Background: Cutaneous malignant melanoma is the sixth most commonly diagnosed cancer in the United States with an incidence that is increasing more quickly than any other type of cancer.
Fortunately, the majority of the cases are diagnosed at early stage, conferring a better prognosis.
However, there are rare giant destructive melanomas >10 cm that are described in the literature,
most metastatic at time of presentation. Here we report the clinical case and genomics of the largest
giant melanoma without evidence of metastatic disease described to date.
Methodology: Literature search of Scopus and Pubmed from 1960-2016 was performed using the
terms “giant, melanoma, cutaneous melanoma, and non-metastatic.” Cases were reviewed for
location, tumor size, Breslow depth, presence of nodal and distant metastases, and surgical and
medical treatments. In addition, we report the case of an 84-year-old female who presented with
a giant melanoma of the shoulder without evidence of metastatic disease. We describe the clinical
characteristics of the tumor and pathologic profile, including immunohistochemistry and genetic
analysis.
Results: 18 cases of primary giant melanoma were identified in addition to our case. Female: 9. Male:
10. Age 1-88. Most common location was the trunk followed by the extremities. Average largest
diameter was 13.3 cm with a range of 5-23 cm. Breslow thickness ranged from 0.45 to 100 mm. Four
(21%) of patients were diagnosed with localized disease, four (21%) with loco-regional metastases
and eight (42%) with distant metastases. Our patient’s tumor genetic analysis with Amplicon next
generation sequencing for melanoma biomarkers was positive for NRAS Q61L but negative for
BRAF, CTNNB1, GNA11, GNAQ, and KIT.
Conclusion: Giant malignant melanomas are extremely rare, particularly those without evidence
of metastatic disease. While they represent a surgical challenge due to their locally destructive
nature, wide local excision and sentinel node biopsy are still considered the standard of care and
may be utilized in conjunction with chemotherapy, immunotherapy, or radiotherapy. Additional
genetic analysis of particular phenotypes may help us to elucidate differences in behavior patterns
of melanomas to better target therapy.
Keywords: Malignant melanoma; Giant melanoma; Melanoma metastases; Melanoma genetics
Introduction
Since the mid 1950’s, the incidence of melanoma has increased more quickly than any other type of cancer [1],making cutaneous malignant melanoma the sixth most commonly diagnosed cancer in the United States [2].Cutaneous melanomas are thought to arise either from the slow progression of a common nevus, first growing radially and then vertically to invade the vasculature and metastasize, or they arise rapidly into aggressive lesions without origin from nevus [3].Fortunately, the majority of cutaneous melanoma is diagnosed early [4]. Lesions diagnosed later with increased thickness generally have worse prognosis, as Breslow thickness, or the depth of the lesions from the basement membrane of the epidermis to the deepest margin, remains the single most important predictor of long term survival. Other less predictive prognostic factors include ulceration, rate of mitosis, and nodal involvement [5]. Women generally have a higher rate of cutaneous melanoma [6] and these lesions are more frequently found in the lower extremities. Men present more often with lesions of the head, neck and trunk [7].Truncal lesions are associated with the BRAF proto-oncogene [8]. While chemotherapeutic and immunologic agents are available for the treatment of systemic disease, surgical excision of early stage melanoma remains the standard of care for curative intent when disease is localized [3],with diameter of excision margin determined by Breslow depth [9]. This report describes the identification, staging, and treatment of a localized giant cutaneous melanoma, which is a rarely described phenomenon.
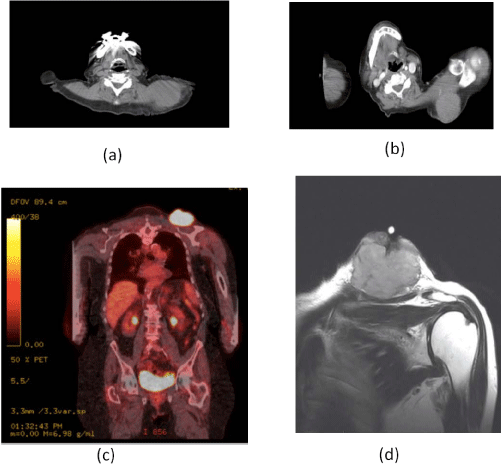
Figure 1
Figure 1
A, B: Computer tomography showing left shoulder tumor without
clear tissue planes. C: Positron emission computer tomography demonstrating
the melanoma tumor to be the only site of disease. D: Magnetic Resonance
of the left shoulder demonstrating the complex tumor with significant skin
edema and inflammation.
Case Presentation
The patient is an 84-year-old Caucasian female nursing home
resident with history significant for mild dementia and 20 pack-year
tobacco uses. The patient presented with a left shoulder malodorous,
fungating massto a local community hospital where she was admitted
for MRSA cellulitis requiring parenteral antibiotics. Computerized
Axial Tomography (CT) scan showed a 6.2 cm solid enhancing mass
posterior to the left trapezius muscle (Figure 1A and 1B). CT-guided
biopsy initially was read as poorly differentiated malignancy favoring
lymphoma, but pathology review at the University of Michigan
was diagnostic of malignant melanoma. Immunohistochemistry
was positive for S100, Cyclin D1, SOX 10, and MTF; rare cells were
positive for HMB-45 and MART-1; negative for CD3, CD4, CD5,
CD7, CD8, CD10, CD20, CD30, CD34, CD45, CD79A, CD138,
PAX5, MPO, MUM1, ALK, BCL-2, BCL-6, Pancytokeratin, and
P63. In situ hybridization was negative for EBER. Staging Positron
Emission Tomography/CT showed 7.2 cm lesion with max SUV of
32.0 g/ml with no evidence of diffuse disease (Figure 1C). MRI of the
left shoulder demonstrated muscular and soft tissue involvement but
no osseous involvement (Figure 1D). MRI brain was also negative for
metastatic disease.
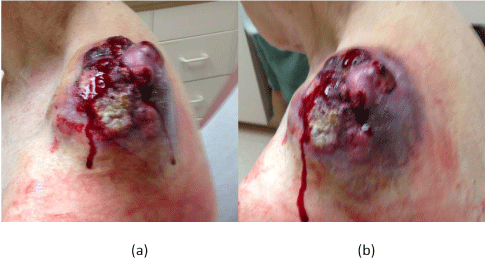
On exam, the tumor was noted to be ulcerated and exophytic
with lobulations. It was actively bleeding and measured 20 x 20 cm
with surrounding erythema (Figure 2A and 2B). Nodal involvement
was clinically negative. Sentinel node biopsy was discussed but due
to the patient’s general condition, the patient’s guardian declined.
Ultrasound of the auxiliary and cervical nodes was negative for
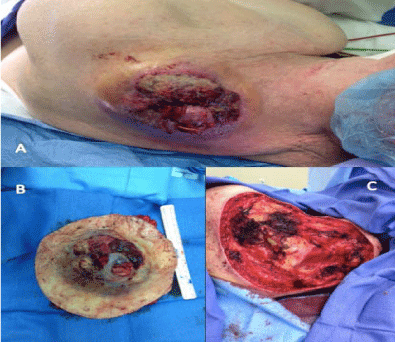
nodal involvement. She underwent en bloc resection of the tumor,
skin, subcutaneous tissue and muscle down to the scapular bone
protuberance with an additional 2 cm margin. (Figure 3A and 3B).
Tumor was lifted off of the left acromion and trapezius muscles,
leaving a 22 cm by 25 cm defect (Figure 3C), requiring plastic surgery
closure with bilaminar Integra and VAC dressing as an interim
closure pending final surgical pathology. The patient tolerated the
procedure well and returned ten days later for closure with split
thickness skin graft.
Surgical pathology noted invasive malignant melanoma,
unclassifiable type; Clark Level 5 and Breslow Depth of 70 mm
surgical margins were negative. No radial growth, microsatellitosis, or
associated melanotic nevus was identified. Vascular invasion, tumorinfiltrating
lymphocytes, and vertical growth phase were present.
Cytology was predominantly epithelioid. Immunohistochemistry
with melanocytic markers revealed expression of SOX 10 of 80%,
MiTF of 70%, S100 of 40%, pan-melanoma cocktail of 10%, and
MART-1 of less than 1% (Figure 3). Results indicate that a significant
number of melanoma cells lost MART-1 expression during tumor
progression. Pathology demonstrated a very high mitotic rate with
12 mitosis/mm2
on HE staining and 18 PHH3 positive cells/mm2
on
two colors IHC with PHH3 and MART-1. (Figure 4A, 4B and 4C).
Genetic analysis with Amplicon next generation sequencing for
melanoma biomarkers was positive for NRAS Q61L but negative for
BRAF, CTNNB1, GNA11, GNAQ, and KIT.
The patient was evaluated by medical oncology but was not
a candidate for adjuvant immunotherapy with interferon given
her old age, dementia, and general performance status. While
new immunotherapy agents such as the PD-1 inhibitors Keytruda
(pembrolizumab) and Opdivo (nivolumab) are promising, their side
effect profiles render them better suited for treatment of recurrence.
The patient was advised to follow up with routine surveillance. She
was with no clinical evidence of recurrence at 6 months follow up.
Patient has since deceased of unknown cause.
Figure 2
Figure 2
A, B Exophytic, ulcerated, hemorrhagic left shoulder melanoma
tumor measuring 20 x 20 cm in diameter.
Figure 3
Figure 3
A: Melanoma tumor with surrounding erythema. B. Excised
specimen. C: Large surgical defect with exposure of osseus structures due to
need of obtaining oncological negative deep margins.
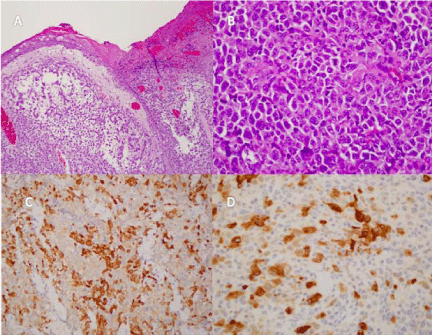
Figure 4
Figure 4
A. Histological section of the malignant melanoma ( H&E staining 25
X), with surface ulceration. B. Histological section representing the melanoma
cells in the dermis (H&E staining 40 X): Cells are large and have pleomorphic
nuclei that contain prominent nucleoli. C. Immunohistochemistry for S100 in
melanoma cells (25 X). D. Immunohistochemistry for panmelanoma cocktail
(40 X).
Table 1
Table 1
Reported cases of giant malignant melanomas.
Table Key: WLE: Wide Local Excision; AND: Axillary Node Dissection; SND: Sentinel Node Dissection; NR: Not Reported; INF: Interferon; anti-TNF: Anti Tumor
Necrosis Factor
Literature Review
Eighteen previous cases of primary giant melanoma from 1960 to June 2016 were identified in addition to our case. Analysis of all 19 cases revealed 9 females and 10 males, with median age of 57, ranging from 1-88 years old. The most common location was the trunk, followed by the extremities. The average largest diameter was 13.3 cm (5-23 cm). Breslow thickness ranged from 0.45 to 100 mm. Four (21%) of patients were diagnosed with localized disease, 4 (21%) with loco-regional disease, and 8 (42%) with distant metastases. The majority of patient underwent wide local excision and auxiliary node dissection. Two patients receive systemic adjuvant treatment with chemotherapy.
Discussion
While melanoma is the sixth most common cause of cancer in
the United States, giant primary melanomas, defined as lesions of at
least 10cm diameter [10], are rare. Of the previous 18 cases reported,
the majority is associated with significant local regional metastatic
disease [10]. Interestingly, there was no evidence of nodal or systemic
disease in our case. In an earlier literature review by Ching et al. [11]
that identified nine cases of giant melanoma, only one case reported
no regional lymphadenopathy and no evidence of metastatic disease.
Previously documented cases of giant primary melanoma report
lesions occurring on the scalp [12], arm [13,14], abdomen [15], and back [4,16,17]. Our patient’s tumor location on the shoulder
presented a unique challenge in terms of surgical reconstruction; a
conservative approach with Integra grafting and VAC dressing was
chosen to await for final pathology results prior to skin grafting. Per
the treatment guidelines published by the National Comprehensive
Cancer Network for those melanomas with Breslow depth of 4 mm
and greater, the principal treatment of wide local excision remains
valid for giant melanomas [18]. These tumors necessitate a minimum
of recommended 2 cm excision margin, with no survival benefit
noted with a greater than two-centimeter margin in melanomas
with thickness greater than 4 millimeters because the risk of death is
more likely from metastatic disease than from recurrence [19]. In the
case presented in this report the lesion was successfully excised with
eventual skin graft reconstruction.
While radiation has not been used historically as a primary
treatment for melanoma due to radio-resistance, radiation therapy
should be considered for giant melanomas. Radiation therapy has
been shown to help palliate approximately 50 percent of patients with
unrespectable, recurrent, or metastatic disease. Radiotherapy may be
used after complete excision, after lymphadenectomy, or to prevent
local recurrence, although evidence is limited [20].
Based on literature review, the likelihood of regional and
metastatic disease is exceptionally high with giant melanomas. Only
three known cases of giant melanoma with no evidence of metastasis
have previously been reported [10,12]. This case report is the 4th case
in the medical literature. Chong et al. [10] reported 2 cases of giant
melanoma without metastasis. The two lesions described by Chong
measured 6.0 × 8.2 cm and 8 × 7 cm, respectively. Additionally, a scalp
melanoma measuring 12 x 10 cm without metastatic disease has been
described [12]. The lesion described in this case report measured 20 x
20 cm, making this tumor the largest known giant melanoma without
metastatic disease. The occurrence of such lesions might suggest a less
aggressive variant of the disease.
As the relationship between melanoma and the expression
of genetic markers is better understood, the current four subtype
histopathalogic classification schemata is challenged, particularly
because genetic profile may prove more predictive of tumor behavior
than histology. For example, desmoplastic melanoma presents with
more advanced Breslow depth but fewer regional metastatic lesions.
The more recently classified primary dermal melanoma, which
by definition is confined to the dermis, shows better survival rates
when compared with nodular melanoma of equal thickness [10].
The surgical pathology of the lesion presented in our case study
demonstrated an invasive malignant melanoma of unclassifiable
type, Clark Level 5 up to the skeletal muscle with Breslow depth of
70 mm. Ulceration and vascular invasion were both present, which
is remarkable in light of the fact that despite vascular invasion no
metastatic lesions were noted wither clinically or radiographically.
The tumor’s slow growth over the course of ten years with
sudden rapid expansion over the course of several months prior to
excision with no proven metastatic disease demonstrates the need for
continued research to elucidate what determines the growth phase,
rate, directionality and metastatic potential of giant melanomas. The
complexity of diagnosing and treating malignant melanoma in part
is due to the fact that melanoma has more gene mutations per cell
than any other type of cancer. In order to more accurately diagnose
and treat the disease, we need to better understand the complex
genomic profiles, signaling pathways, and immune checkpoints that
drive tumor progression and cause some lesions to advance quickly
and metastasize while others grow slowly to an impressive size. Aside
from the four main subtypes of melanoma, as many as seventeen
rare variants have been described. Nevoid melanomas are often
mistaken for benign dermal nevi but do have a metastatic potential.
Desmoplastic melanoma, which appear scar-like, have a significant
amount of mucin and most often appear poorly circumscribed with
an impressive vertical growth phase [21]. Solitary dermal melanoma
is a variety of tumor confined to the dermis. Regardless of subtype,
hematoxylin and eosin staining remains the gold standard of
diagnosis. However, pathologists often measure antibodies directed
against S-100, MART-1/Melan-A, and HMB45 [22,23]. They utilize
reverse transcriptase polymerase chain reaction (RT-PCR) to uncover
melanoma-specific tumor markers such as tyrosinase messenger
RNA [24]. Specifically, additional research is required to elucidate
the underlying genetic profiles that drive the local growth of giant
melanomas, and to better treat these tumors when their size prevents
surgical resection. Genetic profiling with Comparative Genomic
Hybridization (CGH) or Fluorescent Iin Situ Hybridization (FISH)
can guide therapy for giant melanomas that prove unrespectable.
In the presence of mutated BRAF, combination therapy with BRAF
and MEK inhibitors is standard of care. First line treatment options
include Dabrefenib/trametinib or Vemurafenib/cobimetinib, or
monotherapy with either Vemurafenib or Dabrefenib. The choice of
therapy is based on the individual patient. In the absence of a BRAF
mutation, immunotherapy with anti-PD1 agents Pembrolizumab
or Nivolumab as monotherapy or in combination is a category 2A
recommendation [19].
Conclusion
In conclusion, giant malignant melanomas are extremely rare and represent a surgical challenge due to their destructive nature. Even though they have significant metastatic potential, a subset of these tumors could have a more indolent clinical course. Genetic analysis may prove a valuable clinical tool to predict phenotype and to provide more individualized treatment options.
References
- Hall H, Miller D, Rogers J, Bewerse B. Update on the incidence and mortality from melanoma in the United States. J Am AcadDermatol. 1999; 40: 35-42.
- Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, et al. Cancer statistics, 2006. CA Cancer J Clin. 2006; 56: 106-130.
- Houghton AN, Polsky D. Focus on melanoma. Cancer Cell. 2002; 2: 275-278.
- Kruijff S, Vink R, Klaase J. Salvage surgery for a giant melanoma on theb back. Rare Tumors. 2011; 3: e28.
- Baade PD, English DR, Youl PH, McPherson M, Elwood JM, Aitken JF. The relationship between melanoma thickness and time to diagnosis in a large population-based study. Arch Dermatol. 2006; 142: 1422-1427.
- Buettner PG, MacLennan R. Geographical variation of incidence of cutaneous melanoma in Queensland. Aust J Rural Health. 2008; 16: 269-277.
- Bulliard JL, Cox B, Elwood JM. Comparison of the site distribution of melanoma in New Zealand and Canada. Int J Cancer. 1997; 72: 231-235.
- Perez-Gomez B, Aragones N, Pollan M. Divergent cancer pathways for early onset and late onset cutaneous malignant melanoma. A role for sex– site interaction. Cancer. 2010; 115: 2499.
- Haigh P, DiFronzo L, McCready D. Optimal excision margins for primary cutaneous melanoma: a systematic review and meta-analysis. Can J Surg. 2003; 46: 419-426.
- Chong T, Strazzula L, Hoang MP, Arakaki R, Kroshinsky D. Giant Primary Melanoma With No Apparent Metastases: A Report of 2 Cases. JAMA Dermatol. 2014; 150: 574-575.
- Ching JA, Gould L. Giant Scalp Melanoma: A Case Report and Review of the Literature. Eplasty. 2012; 12: e51.
- Panajotovic L, Dordevic B, Pavlovic MD. A giant primary cutaneous melanoma of the scalp—can it be that big? J Eur Acad Dermatol Venereol. 2007; c 21: 1417-1418.
- Del Boz J, Garcia JM, Martinez S, Gomez M. Giant melanoma and depression. Am J Clin Dermatol. 2009; 10: 419-420.
- Tseng WW, Doyle JA, Maguiness S, Horvai AE, Kashani-Sabet M, Leong SP. Giant cutaneous melanomas: evidence for primary tumour induced dormancy in metastatic sites? BMJ Case Rep. 2009; 2009.
- De Giorgi V, Massi D, Carli P. Giant melanoma displaying gross features reproducing parameters seen on dermoscopy. Dermatol Surg. 2002; 28: 646-647.
- Grisham AD. Giant melanoma: novel problem, same approach. South Med J. 2010; 103: 1161-1162.
- Eisen DB, Lack EE, Boisvert M, Nigra TP. Giant tumor of the back. Arch Dermatol. 2002; 138: 1245-1250.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Melanoma. Fort Washington, PA: National Comprehensive Cancer Network. 2011.
- Balch CM, Soong S, Ross MI, Urist MM, Karakousis CP, Temple WJ, et al. Long-term results of a multi institutional randomized trial comparing prognostic factors and surgical results for intermediate thickness melanomas (1.0 to 4.0 mm). Intergroup Melanoma Surgical Trial. Ann Surg Oncol. 2000; 7: 87-97.
- National Collaborating Centre for Cancer (UK). Melanoma: Assessment and Management. London: National Institute for Health and Care Excellence (UK). 2015.
- Li W, Stall A, Shivers SC, Lin J, Haddad F, Messina J, et al. Clinical relevance of molecular staging for melanoma: comparison of RT-PCR and immunohistochemistry staining in sentinel lymph nodes of patients with melanoma. Ann Surg. 2000; 231: 795-803.
- Blessing K, Sanders DS, Grant JJ. Comparison of immunohistochemical staining of the novel antibody melan-A with S100 protein and HMB-45 in malignant melanoma and melanoma variants. Histopathology. 1998; 32: 139-146.
- Donato R. Functional roles of S100 proteins, calcium-binding proteins of the EF-hand type. Biochim Biophys Acta. 1999; 1450: 191-231.
- Scoggins CR, Ross MI, Reintgen DS, Noyes RD, Goydos JS, Beitsch PD, et al. Prospective multi-institutional study of reverse transcriptase polymerase chain reaction for molecular staging of melanoma. J Clin Oncol. 2006; 24: 2849-2857.
- di Meo N, Stinco G, Gatti A, Errichetti E, Bonin S, Albano A, et al. Giant melanoma of the abdomen: case report and revision of the published cases. Dermatology Online Journal. 2014; 15: 20.