Review Article
Use of Extracorporeal Membrane Oxygenation (ECMO) for Management of Profound Dyspnea Caused by a Mediastinal Goiter
Hammond BM1, Shartel SA1, Fundakowski CE2, Keresztury MF1, Kaiser LR3 and Erkmen CP3*
1Department of Anesthesia, Temple University Health Systems, USA
2Department of Otolaryngology, Temple University Health Systems, USA
3Department of Thoracic Medicine and Surgery, Temple University Health Systems, USA
*Corresponding author: Cherie P Erkmen, Thoracic Medicine and Surgery, Temple University Health System, 3401 N. Broad Street Philadelphia, PA 19141, USA
Published: 08 Aug, 2016
Cite this article as: Hammond BM, Shartel SA, Fundakowski CE, Keresztury MF, Kaiser LR, Erkmen CP. Use of Extracorporeal Membrane Oxygenation (ECMO) for Management of Profound Dyspnea Caused by a Mediastinal Goiter. Clin Surg. 2016; 1: 1076.
Abstract
Perioperative management of a large mediastinal mass poses a substantial clinical challenge, particularly during induction of general anesthesia. Critical airway compression with inability to ventilate is a significant threat. We discuss a patient undergoing resection of a complex mediastinal mass who upon induction and attempted intubation had insufficient oxygenation and ventilation. We initiated veno-venous extracorporeal membrane oxygenation allowing for a controlled resection of the complex tumor. She recovered successfully with no complication. Our case exemplifies the importance of multidisciplinary coordination in the perioperative management of the patient with a complex mediastinal mass.
Introduction
The anesthetic management of the patient with a large mediastinal mass poses a significant clinical challenge. The induction of general anesthesia has the potential to evoke tumor-associated compression that may progress to acute respiratory decompensation and/or cardiovascular collapse [1-3]. Furthermore, airway compression can be deceptive; in an asymptomatic patient, it may manifest only during the induction period [4]. As such, a thorough, well-coordinated, multidisciplinary perioperative approach is essential, including appropriate pre-operative contingency planning.
Description of the Case Type
A 60 year old, African-American female with a history of a large thyroid goiter, containing
a significant intra-thoracic component, presented with acutely worsening dyspnea, orthopnea,
biphasic stridor, and hoarseness. She was evaluated in an outside hospital three months earlier, where
she declined surgery and underwent a tracheal stent placement to treat her progressive dyspnea.
Computed Tomography (CT) imaging of the thorax revealed a very large, heterogeneousthyroid
goiter with a significant posterior mediastinal component, extending distally between the trachea
and esophagus, as well as inferiorly to the carina (Figure 1). The trachea was tortuous and deviated
significantly to the right (Figure 2). The tracheal stent extended from the thoracic inlet to the carina,
with mass effect on the posterior aspect of the stent throughout its coarse, resulting in stent buckling
and fracture. There was tracheal luminal narrowing, greatest at the mid-aspect, with a minimum
diameter of approximately 8 mm x 5 mm. There was compression and narrowing of the right
and left mainstem bronchi. The distal airways were patent, although there was extensive bilateral
consolidative and ground glass opacities throughout the lung fields, most significant in the right
upper and lower lobes. The great vessels were noted to be normal in both course and caliber.
Surgical and anesthesia subspecialty teams developed a coordinated plan for induction,
intubation, possible initiation of Extracorporeal Membrane Oxygenation (ECMO), excision of the
tumor and removal of the fractured stent. All team members agreed that cardiothoracic anesthesia,
thoracic surgery, cardiovascular surgery, otolaryngology and perfusion specialists would be present
for evaluation and treatment upon entry to the operating room. Supplemental helium-oxygen
mixture, 6L/min, was administered via nasal cannula. The patient was hemodynamically stable and
oxygen saturation by pulse oximetry was 95%. She was placed in semi-Fowler’s position, as she
could not tolerate the supine position without significant hypoxia and distress. Standard monitoring
cardiac monitoring, pulse oxygen measurement and blood pressure monitoring was established.
Large-bore peripheral intravenous catheters and a right radial
arterial catheter were placed. In preparation for awake oral tracheal
intubation, intravenous midazolam and fentanyl were carefully
titrated to achieve sedation with persevered respiratory function.
Oxygen saturation measured by pulse oximetry remained 98%.
Topical anesthesia of the airway was achieved with administration
of atomized 4% lidocaine solution. Meanwhile, the cardiothoracic
surgical team prepped and draped the groin in preparation of venovenous
ECMO, but because of the semi-fowler positioning and the
patient’s body habitus, access to the femoral vessels was limited. The
perfusion team prepared for VV ECMO and the ECMO circuit was
primed.
We loaded a flexible video bronchoscope with an 8.0-mm wirereinforced
Endotracheal Tube (ETT). We chose the smallest sized ETT
that would accommodate our bronchoscope, keeping the option for
bronchoscopic intervention including balloon dilatation or stent
management available. The patient had sustained end-tidal CO2
, bilateral chest rise and breath sounds, though markedly diminished
on the right. Though the bronchoscope could not traverse the distal
trachea, a view of bilateral mainstem bronchi revealed profound
tracheobronchial narrowing, multiple fractures of the tracheal
stent eroding into the airway and a stent fragment occluding the
right mainstem bronchus, none of which was amenable to flexible
bronchoscopic intervention. Spontaneously breathing, the patient
received inhaled induction of anesthesia with sevoflurane in 100 %
oxygen. However, as the anesthetic depth increased, elevations in
peak airway pressure developed along with significant decreases in
tidal volume. Improvements in tidal volume could not be achieved
with manual-assisted synchronous ventilation. Shortly thereafter,
SpO2 began to decline.
With decreasing oxygenation and difficulty with ventilation,
cardiovascular collapse was eminent. At this point, the collaborative
decision was made to proceed with initiation of VV ECMO. The
patient was placed in the supine position, bilateral femoral venous
catheters were placed and VV ECMO was initiated at flow rate of 4 L/
min. The SpO2 improved rapidly. After ensuring hemodynamic and
respiratory stability, anesthetic depth was increased with sevoflurane.
Vecuronium was administered for neuromuscular blockade. Despite
elevated peak inspiratory pressures and decreased tidal volumes, the
patient had adequate arterial oxygen saturation.
The otolaryngology team subsequently performed a
transverse cervical incision, left paratracheal dissection, and left
hemithyroidectomy. Attempts to deliver the mediastinal component
of the mediastinal tumor through the cervical incision resulted in
complete obstruction of the airway. Despite VV ECMO, the patient
was not able to tolerate periods of apnea. A median sternotomy
allowed for removal of the 8 cm multilobulated mass extending into
the posterior mediastinum. An additional 8 cm subcarinal mass was
dissected, and removed in its entirety.
Unfortunately, the fractured and buckled stent continued to pose
obstruction to ventilation requiring a plan for removal. The patient
underwent extubation, and the thoracic surgeon performed rigid
bronchoscopy. A grasper was advanced through the bronchoscope
and the proximal edge of the stent was secured. The stent was extracted
piecemeal with minimal force. With removal of the mediastinal
component of the goiter and the fractured stent, the trachea assumed
a normal size and shape immediately. The rigid bronchoscope was
removed and the trachea was reintubated by direct laryngoscopy
without difficulty. Subsequent flexible bronchoscopy revealed diffuse
injury to the bronchial mucosa of the right mainstem bronchus as
well as to the trachea from carina to vocal cords (Figure 3). However,
there was no evidence of bleeding or perforation. Given the possibility
of tracheal or mainstem bronchial inflammation and airway laxity,
a deliberate and measured plan for weaning of the ventilator and
ECMO was initiated. Again this was a discussion between all the
invested surgical and anesthesiology teams.
On the morning of post-operative day 1, the patient was
successfully weaned from VV ECMO and the femoral vein cannulae
were removed. Following decannulation, the patient was extubated
with good respiratory function post-extubation. The remainder of her
hospital course was uneventful. She was discharged on post-operative
day 7, with instructions for follow-up with both ENT and thoracic
surgery. One year later, the patient is alive and well without any
lasting symptoms.
Pathology for the thyroid specimen was reported as multinodular
goiter with scarring/hyalinization, calcification, and focal Hurthle
cell metaplasia. The mediastinal component was described as
multinodular goiter with scarring, hyalinization and calcification.
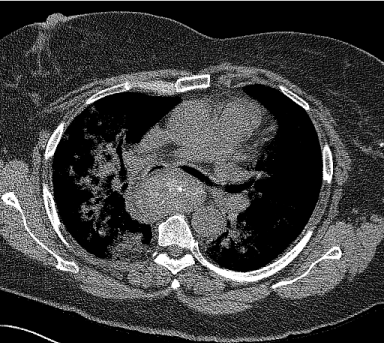
Figure 1
Figure 1
Axial image of Computed tomography depicting posterior mediastinal goiter causing narrowed right and left mainstem bronchi.Contrast CT shows thickening of ascending colon suspicious for malignancy.
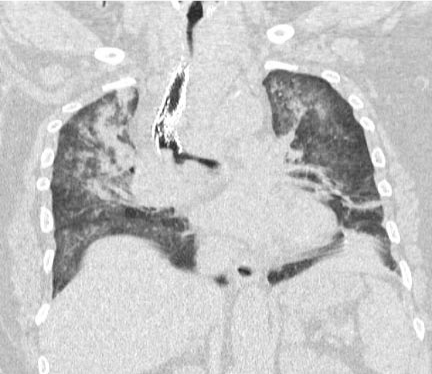
Figure 2
Figure 2
Computed tomography with coronal reconstruction depicting
tortuous trachea with significant deviation and luminal narrowing, as well
as posterior mass effect resulting in buckling and fracture of tracheal stent.
Bilateral lung fields have infiltrates and atelectasis.
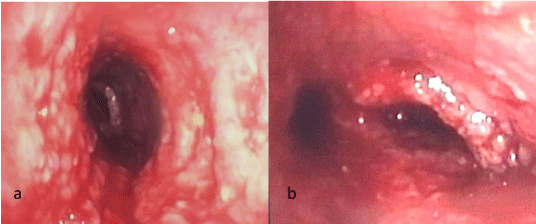
Figure 3
Figure 3
Bronchoscopic view of the trachea (a) and carina (b) following
removal of the compressive goiter and the stent. Though the tracheal
and bronchial mucosa was chronically irritated by the stent, there was no
evidence of transmural injury.
Discussion
The preoperative assessment is critical in identifying the potential
for perioperative respiratory complications. Signs and symptoms
elicited on history and physical exam that are predictive of these
complications include positional dyspnea/orthopnea, hoarseness,
stridor, wheezing, and cyanosis [5]. Bechard et al. [6] demonstrated
a positive correlation between signs and symptoms and perioperative
respiratory decompensation. CT imaging is a standard in preoperative
evaluation and is invaluable in gauging tumor location and degree
of tracheobronchial compression [7]. A cross-sectional tracheal
area of less than 50% of normal diameter can also be predictive of
postoperative pulmonary complications [7]. Finally, flow-volume
loops obtained by spirometry provide an estimation of dynamic
airway flows and obstruction [8]. Specifically, the presence of a midexpiratory
plateau when repositioning from upright to supine, is
seen with extrinsic intrathoracic airway obstruction [1]. However,
studies of flow-volume loops have shown poor correlation with the
degree of airway obstruction [9] and have suggested that upright and
supine spirometry may not offer any advantage over identifying signs
or symptoms and CT imaging [10]. In our case, the progression of
airway obstruction led to atelectasis and infiltrates of bilateral lung
fields, which would confound spirometry. Additionally, our patient’s
acute dyspnea would have prohibited accurate spirometry.
Traditionally, general anesthesia is avoided in patients at significant
risk for perioperative respiratory compromise. The induction of
general anesthesia escalates extrinsic airway compression by several
mechanisms. First, increased abdominal tone in conjunction with
decreased inspiratory muscle tone contributes to reductions in
lung volume of approximately 500 – 1500 mL. In addition, there is
relaxation of the bronchial smooth muscle, increasing the extent of
large airway compressibility and thereby the potential for extrinsic
compression. Finally, as spontaneous ventilation is diminished, there
is blunting of the negative intrathoracic pressure and transpulmonary
gradient that is generated by chest wall expansion and caudal
excursion of the diaphragm. Airway caliber will be comprised and the
effects of extrinsic compression amplified [1].
If general anesthesia is necessary, it is prudent to proceed with a
slow induction using short-acting, titratable agents, while maintaining
spontaneous ventilation and avoiding the use of neuromuscular
blockade [7]. Intravenous anesthetics such as dexmedetomidine and
ketamine may have utility in this context, as they have both analgesic
and sedative properties with the advantage of producing minimal
respiratory depression [11,12]. In addition, helium-oxygen mixtures
may be employed in order to promote laminar gas flow distal to
points of significant obstruction [8]. Neuromuscular blockade should
be avoided until the ability to ventilate with positive pressure is
confirmed. However, cardiopulmonary collapse despite maintenance
of spontaneous ventilation techniques has been reported in the
literature [13].
Airway management is a precarious task in patients with
a large mediastinal mass. Traditionally, the ideal modality for
securing the airway is awake tracheal intubation [7], using local
anesthesia and carefully titrated sedation. The location and degree of
tracheobronchial tumor burden dictate the position of the ETT. A
lesion located in the proximal airway may allow the safe passage of a
reinforced ETT distal to the obstruction. However, lesions involving
the distal trachea through the mainstem bronchi may hinder passage
of an ETT beyond the obstruction and necessitate the use of rigid
bronchoscopy [8]. The rigid bronchoscope can be advanced beyond
the obstruction and ventilation achieved. If the obstruction extends
beyond the mainstem bronchi, conventional intubation may not
provide adequate oxygenation or ventilation. Jet ventilation can be a
temporizing maneuver. However, jet ventilation can cause increased
airway pressure, overexpansion of the lungs, and decreased blood
return to the right ventricle thus exacerbating cardiovascular collapse.
We elected to employ VV ECMO as the patient began to
demonstrate progressive hypoxia. Case reports describe cannulation
for CPB prior to induction of general anesthesia [14,15]. Preinduction
cannulation in preparation for ECMO was considered, but not
performed for multiple reasons. First, we determined that the optimal
location for cannulation would be the femoral veins. Because of our
patient’s body habitus, the femoral vein anatomy was not easily
visible or palpable in the semi-Fowler position. The patient could not
tolerate any flattening at the waist to expose the groin. Positioning
the patient in any position beside semi-Fowler position to place
femoral vein cannulae would likely have caused tumor compression
of the airway, leading to acute airway obstruction and rapid hypoxia
before ECMO could be instituted. Safe and accurate placement of
the femoral cannulae was achievable after increasing anesthetic
depth and gradually repositioning the patient to the supine position.
Second, awake cannulation can be painful and anxiety provoking.
Though awake intubation can also cause anxiety, our judgment was
that awake cannulation would cause more pain, anxiety, hypoxia
and rapid demise than proceeding with awake intubation. Slinger
et al. [16] contest the utility of CPB as a ‘standby’ measure during
the induction of general anesthesia, as cannulation and institution
of CPB in an emergent circumstance is not without potential
obstacles and may require a substantial duration of time prior to
achieving adequate circulation and oxygenation. However, the
institution of ECMO has its own potential complications including
but not limited to vessel injury, bleeding and thromboembolism.
As mentioned previously, the patient’s body habitus and inability
to tolerate any position except the semi-Fowler position made
approach of cannulation prohibitive if not inevitably complicated or
ineffectual. We continuously evaluated the need for ECMO and the
optimal timing. The patient was successfully intubated and initially
successfully oxygenating and ventilating. We subsequently learned
that this was not sustainable. The rate of arterial oxygen desaturation
in our patient was not rapid and never reached dangerous levels.
The combination of partial ventilation and oxygenation from the
airway and the subsequent addition of ECMO allowed for optimal
management of the airway, thus averting cardiovascular collapse.
For this clinical scenario the institution ECMO after intubation was
the safest plan. In addition, the cardiothoracic surgical service at our
institution is very experienced in the placement of ECMO and was
able to cannulate the femoral veins in a timely fashion. Nonetheless,
in the setting of a mediastinal mass compressing the airway, femoral
vein cannulation or percutaneous placement of guidewires for ECMO
prior to induction should be considered, as it was in this case.
In summary, anesthetic management of the patient with a
mediastinal mass must be managed on an individualized basis.
As exemplified by our case, the clinical worth of a thorough, wellcoordinated,
multidisciplinary perioperative approach is invaluable.
Inadequate preoperative risk assessment and preparation, a poorly
executed anesthetic plan or lack of contingencies may ultimately
result in critical complications.
References
- Neuman GG, Weingarten AE, Abramowitz RM, Kushins LG, Abramson AL, Ladner W. The anesthetic management of the patient with an anterior mediastinal mass. Anesthesiology. 1984; 60: 144-147.
- Lin CM, Hsu JC. Anterior mediastinal tumour identified by intraoperative transesophageal echocardiography. Can J Anaesth. 2001; 48: 78-80.
- Takeda S, Miyoshi S, Omori K, Okumura M, Matsuda H. Surgical rescue for life-threatening hypoxemia caused by a mediastinal tumor. Ann Thorac Surg. 1999; 68: 2324-2326.
- Goh MH, Liu XY, Goh YS. Anterior mediastinal masses: an anaesthetic challenge. Anaesthesia. 1999; 54: 670-682.
- Fabbro M, Patel PA, Ramakrishna H, Valentine E, Ochroch EA, Agoustides JG. Challenging Perioperative Management of a Massive Anterior Mediastinal Mass in a Symptomatic Adult. J Cardiothac Vasc Anesth. 2014; 28: 819-825.
- Bechard P, Letourneau L, Lacasse Y, Cote D, Bussieres JS. Perioperative Cardiorespiratory Complications in Adults with Mediastinal Mass. Anesthesiology: incidence and risk factors. 2004; 100: 826-834.
- Erdos G, Tzanova I. Perioperative anaesthetic management of mediastinal mass in adults. Eur J Anaesthiol. 2009; 26: 627-632.
- Blank RS, de Souza DG. Anesthetic management of patients with an anterior mediastinal mass: Continuing Professional Development. J Can Anesth. 2011; 58: 853-867.
- Torchio R, Gulotta C, Perbondi A, Ciacco C, Guglielmo M, Orlandi F, et al. Orthopnea and tidal expiratory flow limitation in patients with euthyroid goiter. Chest. 2003; 124: 133-140.
- Hnatiuk OW, Corcoran PC, Sierra P. Spirometry in surgery for anterior mediastinal masses. Chest. 2001; 120: 1152-1156.
- Abdelmalak B, Marcanthony N, Abdelmalak J, Machuzak MS, Gildea TR, Doyle DJ, et al. Dexmedetomidine for anesthetic management of anterior mediastinal mass. J Anesth. 2010; 24: 607-610.
- Frawley G, Low J, Brown TC. Anaesthesia for an anterior mediastinal mass with ketamine and midazolam infusion. AnaesthIntens Care. 1995; 23: 610-612.
- Gardner JC, Royster RL. Airway Collapse with an Anterior Mediastinal Mass despite Spontaneous Ventilation in an Adult. Anesth Analg. 2011; 113: 239-242.
- Said SM, Telesz BJ, Makdisi G, Quevedo FJ, Suri RM, Allen MS, et al. Awake Cardiopulmonary Bypass to Prevent Hemodynamic Collapse and Loss of Airway in a Severely Symptomatic Patient With a Mediastinal Mass. Ann ThoracSurg. 2004; 98: e87-90.
- Tempe DK, Arya R, Dubey S, Khanna S, Tomar AS, Grover V, et al. Mediastinal mass resection: femorofemoral cardiopulmonary bypass before induction of anesthesia in the management of airway obstruction. J Cardiothorac Vasc Anesth. 2001; 15: 233-236.
- Slinger P, Karsil C. Management of the patient with a large anterior mediastinal mass: recurring myths. Curr Opin Anaesthesiol. 2007; 20: 1-3.