Case Report
Sub-Acute Bowel Obstruction Preventing Sudden Cardiac Death: A Case of Synchronous but Unrelated Left Atrial Myxoma and Colorectal Adenocarcinoma
Kar A1,2*, Sudarsanam A1, Papaspyros S2 and Prasad S2
1Department of Cardiothoracic Surgery, St Bartholomew’s Hospital, UK
2Department of Cardiothoracic Surgery, Royal Infirmary of Edinburgh, UK
*Corresponding author: Ashok Kar, Department of Cardiothoracic Surgery, St Bartholomew’s Hospital West Smithfield, London, EC1A 7BE, UK
Published: 08 Aug, 2016
Cite this article as: Kar A, Sudarsanam A, Papaspyros S, Prasad S. Sub-Acute Bowel Obstruction Preventing Sudden Cardiac Death: A Case of Synchronous but Unrelated Left Atrial Myxoma and Colorectal Adenocarcinoma. Clin Surg. 2016; 1: 1075.
Abstract
Primary cardiac tumours are an extremely rare entity, which may be completely asymptomatic or
present with a wide range of clinical symptoms and signs such as congestive heart failure, murmurs,
stroke or arrhythmias.
We discuss the case of a 53 year old with synchronous but unrelated left atrial myxoma and colorectal
adenocarcinoma. To our knowledge, this is the first report in the literature of a patient with an atrial
myxoma incidentally discovered as part of staging following a diagnosis of colorectal cancer.
Introduction
Although left atrial myxomas are the most commonly encountered benign neoplasm by the
cardiac surgeon, they remain relatively rare. The commonest neoplasms of the heart are metastatic
(autopsy frequency 1.5-21%). Primary cardiac tumours have an autopsy frequency of 0.001%–
0.28%. Three-quarters of these are benign and three-quarters of these are myxomas [1]. Myxomas
most commonly occur in the left atrium (90%) and are generally attached to the atrial septum in or
adjacent to the fossa ovalis. The incidence is greatest in those aged 30-60 years, with a notable female
preponderance [2].
Carney’s complex, a neuroendocrine-cardiac syndrome is a common cause of familial recurrent
cardiac myxomas. Other clinical features include pigmented skin lesions, Schwannomas and
multiple recurrent mucocutaneousmyxomas. Endocrine neoplasms and/or overactivity may also
be present [3].
The presentation of myxomas is variable. Asymptomatic myxomas may be picked up incidentally
on routine imaging e.g. echocardiography, Computed Tomography (CT). Others may present with
dyspnoea, palpitations, heart murmurs, syncope, pulmonary hypertension, pulmonary oedema or
constitutional symptoms. Notably, it often presents late with myxomatous thrombo-embolisation
manifesting as strokes or end-organ dysfunction [2,4].
Diagnosis of cardiac myxomasis usually made using Transthoracic Echocardiography (TTE),
which has a sensitivity of 95.2% [2]. However, trans-oesophageal echocardiography although more
invasive, remains the gold standard for diagnosing cardiac myxomas with a sensitivity approaching
100% [5]. In a large cohort study with 112 patients, less than 2% of left atrial myxomas were
diagnosed using CT or MRI [4].
We believe this is the first reported atrial myxoma in the literature, which was diagnosed
incidentally on cross-sectional imaging during staging for colorectal cancer. Various similar rare
associations have been reported in the literature including cardiac metastases from colorectal
cancer [6,7], synchronous atrial myxomas with a small bowel myxoma [8] and adenosquamous lung carcinoma [9].
Case Presentation
A 53 year old woman presented to the A&E department with symptoms and signs of sub-acute
bowel obstruction. Her past medical history significantly included hypertension and a previous
appendicectomy. She reported a family history of lung cancer (mother). She also demonstrated no
evidence of Carney complex phenotype on clinical assessment.
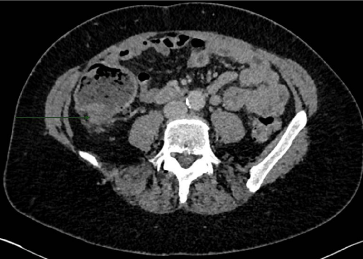
A contrast CT Abdomen/Pelvis identified a dilated caecum and
small bowel with thickening of the ascending colon suspicious for
malignancy (Figure 1). Colonoscopy demonstrated an ulcerated,
obstructing stricture of the ascending colon which appeared malignant.
She subsequently underwent an urgent right hemi-colectomy to
relieve the obstruction and tissue was sent for histopathological
diagnosis. Histology confirmed Dukes C1 (pT4b N2 - 8/24 positive
lymph nodes) moderately differentiated adenocarcinoma extending
onto the peritoneal surface.
Following MDT discussion, adjuvant chemotherapy was offered
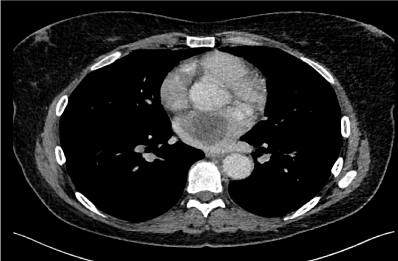
to the patient. A staging PET – CT/CT Thorax was undertaken which
revealed a 6 cm abnormal mass involving the left atrium (Figure
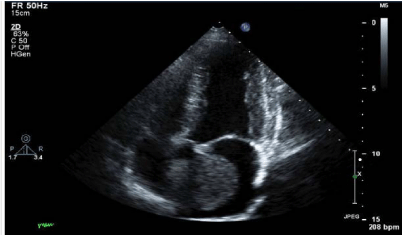
2). A Trans-Oesophageal Echocardiography (TOE) was organised
to characterise this mass further. This confirmed the likelihood of
a left atrial myxoma (Figure 3). The patient reported no significant
symptoms associated with this other than occasional shortness of
breath on exertion which was steadily worsening over the previous
six months. Clinically she had no murmurs or signs of congestive
cardiac failure.
In view of the cardiac and embolic risks associated with this
large myxoma, a decision was made to proceed with urgent surgical
resection. Access was gained via midline sternotomy and the
pericardium lifted to obtain optimal exposure of the heart in the
mediastinum.
Cardiopulmonary bypass was established via bi-cavalcannulation.
Cardioplegia was delivered using an aortic root vent. A standard aortic
cannulation was performed and the myocardial protection strategy
involved cooling to 32 degrees Celsius. Cold blood cardioplegia was
delivered at twenty minute intervals.
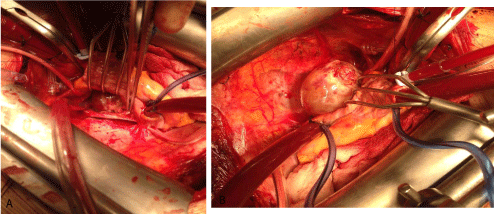
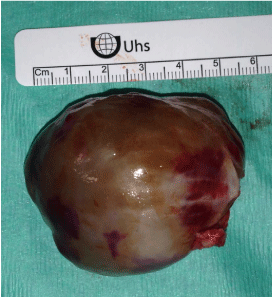
The left atrium was carefully incised and dissected allowing
the tumour to be identified and exposed. The tumour was carefully
removed en-bloc to prevent embolisation leaving an intact left atrial
wall and septum. Notably it was tethered to the posterior left atrial
wall on a broad stalk, with minimal involvement of the inter-atrial
septum (Figures 4a and 4b).
Following complete excision (Figure 5), primary closure of the
left atrium was undertaken and the patient re-warmed. The patient
was weaned off cardiopulmonary bypass following standard decannulation.
Haemostasis was achieved thereafter and the chest
closed using sternal wires.
The patient was transferred to the Intensive Care Unit (ICU) postoperatively
in normal sinus rhythm without any inotropic support.
She was extubated after a period of observation and subsequently
stepped down to the general ward before being discharged home on
day five post operatively without any complications.
Subsequent tumour histopathology confirmed an atrial myxoma.
No issues were identified at out-patient follow-up 6 weeks post
operatively. Chemotherapy was scheduled as planned for the
colorectal adenocarcinoma.
Figure 1
Figure 2
Figure 3
Figure 4
Figure 5
Discussion
Surgical resection remains the mainstay of treatment with cardiac
myxomas and operative outcomes are excellent with recurrence rates
of about 5% [1]. Recurrence rates are significantly higher in patients
with Carney’s complex (22%) and this highlights the importance of
genetic screening in picking up high risk patients [4].
There is an increasing role for genetic screening in patients
diagnosed with cardiac myxomas. Mutations in the PRKAR1 – Alpha
gene sequence have been known to account for Carney’s complex
which results in recurrent atrial myxomas, amongst several other
clinical features. These patients also have a predisposition to develop
tumours of the ovary, pituitary, adrenal, and thyroid glands [3,11].
The use of other novel genetic techniques – genotyping, SNPs, gene
profiling of patients along with immunocytochemistry analysis of
tissue may also help ascertain whether certain patients with myxoma
are pre-disposed to extra—cardiac tumours.
When evaluating the symptoms related to atrial myxoma in
this case, it is important to emphasise that the 6 month history
of progressively worsening shortness of breath was not elicited
nor thought of as relevant by the patient during the initial clinical
assessment. In hindsight, following the incidental diagnosis of an
atrial myxoma, this would have been a clinically important symptom.
This case report therefore shows how fortunate our patient was
to have cross-sectional imaging as part of her staging work-up for
colorectal cancer. For instance, had our patient not presented with subacute
bowel obstruction secondary to a colorectal adenocarcinoma,
she may have presented at a later point in life with potentially life
threatening or life altering clinical sequelae associated with the atrial
myxoma e.g. stroke (secondary to embolic phenomena), arrhythmias/
sudden death or heart failure.
Synchronous cardiac myxomas with other neoplasms have
been reported in the literature [8,9]. However there is only one
other case report that describes asynchronous cardiac myxoma and
colorectal cancer and the patient presented with a combination of
gastrointestinal and cardiac symptoms and signs [10].In contrast, our
patient’s diagnosis of an atrial myxoma was purely incidental with
the cardiac pathology being discovered after extra-cardiac symptoms
were investigated and treated, rather than concurrently or as the
initial diagnosis.
Other case reports describe colorectal cancer with metastatic
disease to the heart and notably metastatic cardiac neoplasms are far
commoner than primary cardiac neoplasms [2,6,7].
However with the latter, this case report highlights whether or
not patients presenting with cardiac myxomas should be screened for
extra-cardiac tumours, as there is little clear evidence for this in the
literature, and therefore may well be an area of research for the future.
Finally another interesting issue relates to the follow-up of patients
with cardiac myxoma. Currently in the UK, patients are discharged
back to their referring cardiologist from the cardiothoracic surgical
clinic after being usually seen within 4-6 weeks post-operatively. It
may well be prudent that these patients be discussed at a regional
MDT with oncology input to identify whether they would benefit
from further imaging or investigations as part of extra-cardiac
screening.
Their remains a lot of interesting, unanswered questions in cardiac
myxoma management. This is partly because of the low incidence of
this disease entity. Future studies should focus on links with extracardiac
tumours and associated screening, and understanding
underlying genetic mechanisms to guide optimal post-operative
follow up and treatment.
References
- Maraj S, Pressman GS, Figueredo VM. Primary Cardiac Tumours. Int J Cardiol. 2009; 133: 152–156.
- Sudarsanam A, Briffa N. ‘Doctor, there is a lump in my heart’ – A rare case of a ventricular myxoma. J Surg Case Rep. 2011; 2011: 1.
- Shetty Roy AN, Radin M, Sarabi D, Shaoulian E. Familial recurrent atrial myxoma: Carney’s complex. Clin Cardio. 2011; 34: 83–86.
- Pinede L, Duhaut P, Loire R. Clinical presentation of left atrial cardiac myxoma.A series of 112 consecutive cases. Medicine (Baltimore). 2001; 80: 159–172.
- Engberding R, Daniel WG, Erbel R, Kasper W, Lestuzzi C, Curtius JM, et al. Diagnosis of heart tumours by transesophageal echocardiography: A multicentre study in 154 patients. Eur Heart J. 1993; 14: 1223–1228.
- Choi PW, Kim CN, Chang SH, Chang WI, Kim CY, Choi HM. Cardiac metastasis from colorectal cancer: A case report. World J Gastroenterol. 2009; 15: 2675-2678.
- Pizzicannella J, Ricci V, Gorla R, Spinapolice E, Esposito A. Isolated cardiac metastasis from colorectal cancer in a 35-year-old man. Case Rep Med. 2012; 2012: 751761.
- Wang Y, Sharkey FE. Myxoma of the small bowel in a 47-year-old woman with a left atrial myxoma. Arch Pathol Lab Med. 2003; 127: 481-484.
- Hong-cheng L, Xiao-feng C, Yu-ming Y, Ge-ning J. Synchronous left atrial myxoma and adenosquamous lung carcinoma. Chin Med J. 2013; 126: 2992-2993.
- Nuño IN, Kang TY, Arroyo H, Starnes VA. Synchronous cardiac myxoma and colorectal cancer. Tex Heart Inst J. 2001; 28: 215-217.
- Boikos SA, Stratakis CA. Carney complex: pathology and molecular genetics. Neuroendocrinology. 2006; 83: 189-199.