Research Article
Long-Term Outcome of Patients Following Surgery for Metachronous Pulmonary Metastases of Highly Aggressive Non-Colorectal Cancers
Grotelüschen R*, Reeh M*, Vashist YK, Izbicki JR, Bockhorn M and Bachmann K
Department of General, Visceral and Thoracic Surgery, University Medical Centre Hamburg-Eppendorf, Germany
*Corresponding author: Matthias Reeh, Department of General, Visceral and Thoracic Surgery, University Medical Centre HamburgEppendorf, Martinistr. 52, 20246 Hamburg, Germany
Published: 14 Jul, 2016
Cite this article as: Grotelüschen R, Reeh M, Vashist YK, Izbicki JR, Bockhorn M, Bachmann K. Long-Term Outcome of Patients Following Surgery for Metachronous Pulmonary Metastases of Highly Aggressive Non-Colorectal Cancers. Clin Surg. 2016; 1: 1052.
Abstract
Background: Most epithelial tumors metastasize to the lung. In 20% of cases no involvement of other organs can be detected. In patients with resectable colorectal lung metastases, the benefit
of metastasectomy has been proven. No uniform guidelines exist, regarding entities such as
hepatocellular carcinoma, pancreatic carcinoma or esophageal carcinoma. This study evaluated the
long-term-outcome after resection of pulmonary metastases from highly aggressive, non-colorectal
cancer entities.
Methods: 49 patients were included who underwent complete pulmonary resection for lung
metastases at the University Medical Center Hamburg-Eppendorf from 2000 to 2009. Overallsurvival
rates were evaluated by Kaplan-Meier analysis.
Results: The median follow-up time was 96 months. The overall median survival after diagnosis of the
primary tumor was 59.9 months (mean survival 86.3 months). The overall 3-, 5- and 10 year survival
rate was 71.4% (35/49), 49% (24/49) and 26.5% (13/49). Median survival after metastasectomy was
22.6 months (mean 55 months). The 1-, 5- and 10- year survival rate after resection was 67.3%
(33/49), 36.7 % (18/49) and 14.3% (7/49). The disease-free survival was 63.3% (31/49), 34.7% (17/49)
and 12.2% (6/49) after 1, 3 and 5 years, respectively.
Conclusion: Even in patients with highly aggressive, non-colorectal cancer, pulmonary
metastasectomy can be performed with a low morbidity and mortality. It can improve overall
survival and provide a chance of long oncological outcome in selected patients. However, the
decision to perform a pulmonary metastasectomy in this setting requires a multidisciplinary setting.
Keywords: Pulmonary metastases; Lung resection
Introduction
The majority of epithelial tumors metastasize to the lung [1]. Although pulmonary metastases indicate an advanced disease with a limited life expectancy, improved surgical techniques and
changes in perioperative management result in an improved long-term survival after lung resection
[2]. Today, resection of metastases in the lung from a wide range of primary sites has become a
routine part of the daily clinical practice of a thoracic surgeon. Even in patients with advanced co
morbidities, surgical treatment can be performed with low morbidity and mortality rates [3]. Thus,
the spectrum of indications for pulmonary metastasectomy has been expanded.
20% of patients with lung metastasis have no detectable involvement of other organs and
complete removal of pulmonary metastases improves long-term survival with 5-year survival rates
of 30%. Surgical treatment is the treatment of choice in well selected patients [2]. Several studies
indicated that a small number of lesions, a long disease-free interval and no lymph node metastases
improve oncological long-term outcome [4]. In these patients, the primary tumor should be under
control, no other potentially curative treatment available and all metastases should be totally
resectable. On the contrary, diffuse or unresectable metastases are usually treated with palliative
chemotherapy. However, there are no prospective randomized trials proving the benefit of the
surgical treatment in patients with pulmonary, non-colorectal metastases. Especially, guidelines
are missing for the treatment of pulmonary metastases of highly aggressive tumor entities like
pancreatic or esophageal cancer.
This study evaluated the long-term outcome of patients following resection of pulmonary
metastases of aggressive, non-colorectal carcinomas.
Table 1
Patients and Methods
From 2000 to 2004, consecutively 49 patients with pulmonary
metastases originate from other than colorectal or renal cell
carcinomas with complete R0 resection at the University Medical
Center Hamburg-Eppendorf were included. Informed consent was
obtained from all patients. The study was approved by the medical
ethics committee of the Chamber of Physicians of Hamburg. Data
were collected retrospectively and followed up according to the
national guidelines. Based on the primary tumor, the following data
were collected: histology, date of diagnosis and surgical treatment,
tumor-classification. Based on the pulmonary metastases the
date of diagnosis and surgical treatment of metastases, surgical
approach and procedure, number of radiographically detected and
resected metastases, size of metastases, sequential and/or repeat
metastasectomies incl. approach and procedure, recurrence other
than pulmonary, perioperative therapy and the date of death were
recorded.
After primary tumor treatment, patients were evaluated at regular
intervals with chest CT-scans, either at our centre or by associated
oncologists. After the diagnosis of pulmonary metastases, patients
were treated by surgical resection in the absence of contraindications.
Operative approaches included lateral thoracotomy, sternotomy,
mini-thoracotomy and Video-Assisted Thoracoscopic Srgery (VATS)
depending on the number of metastases and the tumor location.
Pulmonary resection was performed as wedge resection, lobectomy
or bilobectomy. Patients, who developed pulmonary lesions during
the follow up were treated with repeated lung resection.
Statistical analysis
Overall survival and three-, five- and ten-year survival was
calculated from the date of diagnosis of the primary tumor. The time
to death was modelled using the Kaplan Meier method. Deaths which
resulted from the disease were treated as an endpoint for diseasespecific
survival; survival was treated as censored observations.
Subgroup analyses were performed on disease-free interval,
occurrence pattern, histology, surgical procedure, number and size
of metastases. In all statistical analyses, p-value ≤0.05 was considered
as significant.
Figure 1
Table 2
Figure 2
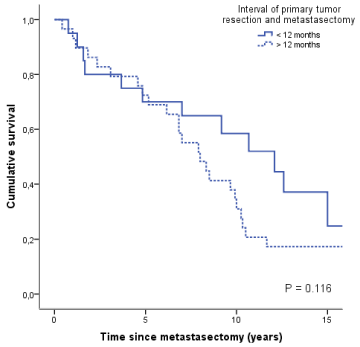
Figure 2
Survival as a function of the time interval between primary tumor
and pulmonary metastases.
Results
From 2000 to 2004, 49 patients, 34 (69%) male and 15 (31%)
female, with a median age of 55 years (range 5 -78 years) received
resection for pulmonary metastases from solid malignancies other
than colorectal or renal cell carcinoma. The primary tumors were
head and neck carcinoma (N=14), germ cell testicular carcinoma
(N=9), malignant melanoma (N=8), hepatocellular carcinoma (N=5),
pancreatic carcinoma (N=3), urothelial cell carcinoma (N=2), breast
carcinoma (N=1), nephroblastoma (N=1), adrenal gland carcinoma
(N=1), esophageal carcinoma (N=2), ovarian carcinoma (1),
porocarcinoma shoulder (1), anal carcinoma (1), (Table 1).
24 (49%) patients presented with unilateral and 25 (51%) with
bilateral metastases. The surgical approach was lateral thoracotomy (N=30), sternotomy (N=16) and VATS (N=2) and mini thoracotomy
(N=1). The resection was performed as a wedge resection (N=45),
lobectomy (N=3) and bilobectomy (N=1). Six patients underwent
sequential resection due to initially bilateral metastases. 11 patients
had a repeated resection after pulmonary tumor recurrence.
18 patients had a relapse of the primary tumor. Four of them had
a malignant melanoma, four a germ cell testicular carcinoma, three a
hepatocellular carcinoma, one pancreatic carcinoma, one urothelial
cell carcinoma, one ovarian carcinoma, two esophageal carcinoma,
one hypopharyngeal carcinoma and one differentiated thyroid
cancer. Median interval from diagnosis of primary tumor to the
diagnosis of lung metastases was 21 months (range 0-160 months).
The overall median survival after diagnosis of the primary tumor
was 59.9 months (mean survival 86.3 months). The overall 3-year
actuarial survival rate was 71.4% (35/49), the five-year survival rate
of 49.0% (24/49) and then 10-year survival rate after diagnosis of the
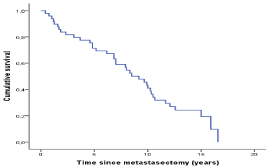
primary tumor was 26.5% (13/49). The cumulative survival is shown
in (Figure 1).
Median survival after metastasectomy was 22.6 months (mean 55
months). The 1-year survival rate after resection was 67.3% (33/49),
3-year survival rate after resection was 42.9% (21/49), the 5-year
survival rate was 36.7 % (18/49) and the 10-year survival rate after
first metastasectomy was 14.3% (7/49). The disease-free survival was
63.3% (31/49), 34.7% (17/49) and 12.2% (6/49) after 1, 3 and 5 years,
respectively. The mean survival rates related to the different tumor
entities are shown in (Table 2). Kaplan-Meier Survival Analysis
revealed no significant differences in histology, bilateral or unilateral
lung metastases, number or size of resected metastases or surgical
procedure (data not shown). Time interval between diagnosis of the
primary tumor and pulmonary metastases more than 12 months
showed longer overall survival in comparison to a time interval
smaller than 12 months (Figure 2). The follow up was calculated from
the first pulmonary metastasectomy, the median follow-up was 82
months.
For the 12 patients who were alive at the time of last followup,
the median post-metastasis follow-up was 57,5 months. The
primary malignancy of these patients was testicular carcinoma (N=5), malignant melanoma (N=2), hypopharyngeal carcinoma (N=2), maxillary sinus carcinoma (N=1), poro-carcinoma (N=1)
and thyroid gland carcinoma (N=1). 8 of these 12 patients had no
evidence of tumor recurrence at the last follow-up.
Discussion
Metastases cause 90% of cancer deaths. Only a few patients
with metastases survive more than one year after diagnosis [2]. In
patients presenting with diffuse or unresectable metastases, palliative
chemotherapy is usually the standard treatment. Accepted criterias
for resection are technical feasibility, controlled primary tumor
site, adequate cardiopulmonary function and complete removal of
metastases. Unresectable primary tumor and predicted incomplete
metastasectomy are contraindications to pulmonary metastasectomy
[4-6].
Standard treatment for isolated pulmonary metastases arising
from colorectal carcinoma is the surgical removal [7], but only a few
reports have addressed the value of surgical resection of pulmonary
metastases from other solid tumors. This study describes the clinical
courses of 49 patients operated on pulmonary metastases arising
from 20 different solid tumor entities.
In addition to the stage of disease, histology and tumor biology
are the major prognostic predictors and define the surgical approach
[3].
Patients suffering from head and neck cancer have a poor
outcome. Head and neck cancers often metastasize to distant organs
and most commonly to the lung [8,9]. Studies showed that pulmonary
metastasectomy improves survival of patients with squamous cell
carcinoma of the head and neck. Five-year survival rates are up to 40%
in the literature [10,11]. Shiono et al. [12] reviewed retrospectively
114 patients who were operated on pulmonary metastases. The
median survival time was 26 months, which is comparable to our data
(mean survival 24.5 months). Wedman et al. [8] demonstrated the
benefit of the surgical approach by reporting a 5-year survival rate of
59% compared to 4% in the non-metastasectomy group. But for other
histological types of head and neck cancers like thyroid cancer and
adenoid cystic carcinoma is less clear [9]. Winter et al. [13] described
a significant better outcome in patients who underwent pulmonary
metastasectomy when compared to chemotherapy regimes. Therefore,
Younes et al. [14] and Kaifi et al. [4] postulated that all patients with
head and neck cancers with no extrapulmonary metastasis should
undergo pulmonary resection to achieve long-term survival.
The lung is the most common site of metastasis in patients
with germ cell tumors. The surgical treatment of pulmonary
metastases in germ cell testicular cancer patients is due to the high
efficacy of anticancer chemotherapy confined to residual tumors.
Metastasectomy enables detection of viable tumor cells which
indicate direct further chemotherapy. This multimodal treatment
regime can achieve 5-year survival rates of up to 82% [10] and even
repeated resections are indicated [15]. We analysed the course of
disease of 9 patients, of which five patients survived with no signs of
relapse, mean survival after first metastasectomy was 49.5 months.
These results support the observation that a substantial number can
be successfully salvaged. Xia et al. [16] showed that surgical resection
of all residual masses is indicated in patients with metastatic germ cell
tumors of the lung and mediastinum.
In 40% of patients with malignant melanoma, pulmonary
metastasis is the first sign of metastatic disease [17]. Pulmonary
metastases from malignant melanoma are associated with poor
survival due to the propensity to metastasize systemically to other
sites in addition to the lung. The 5-year survival rate of patients
with pulmonary metastases is 4% [18]. The resection of isolated
pulmonary metastases can achieve 5-year survival rates of up to
27% [19,20]. In our study, two of eight patients were alive, one had
recurrent pulmonary metastases, and the other patient had no signs
of relapse. The mean survival after first metastasectomy was 37.6
months. In accordance to our results, Petersen et al. [21] support the
role of surgery for select subset of patients with isolated pulmonary
metastasis. Main prognostic factor for improved survival are complete
resection, long duration for formation of metastases, less than two
nodules and tumor negative thoracic lymph nodes [18].
The lungs are the most common extrahepatic metastatic site
[22]. Only few data are available about lung metastasectomy
for hepatocellular carcinoma [23,24]. Zhang et al. [25] analyzed
one hundred and five patients with lung metastases from HCC
retrospectively. The mean and median survival times were 8.7 and
5.9 months after lung metastases. The poor prognosis of this entity
is confirmed by our results, the mean survival was 12.6 months.
Patients with pancreatic cancer have a very limited life-expectancy.
Median survival is 8 to 12 months for patients with locally advanced
unresectable disease and only 3 to 6 months for those who present
with metastases. The 3 patients that have been analysed in this study
had a mean survival of 14.6 months. However, due to the small
number of patients a valid conclusion cannot be made. A report of
the International Registry of Lung Metastases from 1997 evaluated
the long-term survival after pulmonary metastasectomy in more
than 5000 patients [3]. 5- and 10-year survival rates were 36% and
26%, respectively, which is comparable to our series with a five-year
actuarial survival rate of 39,6% (19/48). Factors associated with a
better prognosis are a longer disease-free interval between treatment
of the primary tumor and diagnosis of pulmonary metastases,
complete resection of the metastases and the presence of a single
or limited number of metastases. While our retrospective analysis
confirmed the better prognosis for patients in whom the metastases
occur late (< 12 vs. > 12 months; Figure 2). No difference could be
seen depending on the number of metastases.
In our study, 18 of 49 (37%) patients had a relapse after initial
metastasectomy. The most frequent site of relapse following
pulmonary resection is the lung [26,27]. Following potentially
curative metastasectomy, additional lesions are identified in
50 percent of cases due to the subsequent growth of previously
undetectable lesions. Among the patients who underwent repeated
metastasectomy, the 5- and 10-year survival rates were 44% and 29%,
respectively. Repeat resection after several metastasectomies can
significantly improve overall survival. Kandioler et al. [28] analyzed
35 patients with recurrent pulmonary metastases. The authors could
show that patients with a long disease-interval between resection
for the primary tumor and the first pulmonary metastasectomy
have a significantly better 5-year survival rate. These results are in
accordance to other studies analysing the value of repeat pulmonary
metastasectomies [29,30]. In contrast, the median survival among
patients who were not resected following relapse in one series was 8
months [31].
Conclusion
Radical tumor resection continues to be the most effective therapy in patients with malignant tumors. In order to restrict surgery to those patients who are most likely to benefit in terms of symptom palliation or prolonging survival the decision to perform a pulmonary metastasectomy requires a multidisciplinary approach. In patients with limited pulmonary metastatic disease, pulmonary metastasectomy is a well accepted treatment with low morbidity and mortality rates. This treatment approach can improve overall survival and provide a chance of cure in selected patients which would probably not be possible by systemic therapy alone. Although the study population is limited and the tumor entities are heterogeneous with different tumor biology, this report may contribute to the discussion on the implication of pulmonary metastasectomy in patients suffering from aggressive, non-colorectal cancer entities.
References
- Brooks SA, Lomax-Browne HJ, Carter TM, Kinch CE, Hall DM. Molecular interactions in cancer cell metastasis. Acta Histochem. 2010; 112: 3-25.
- Younes RN, Haddad F, Ferreira F, Gross JL. [Surgical removal of pulmonary metastasis: prospective study in 182 patients]. Rev Assoc Med Bras. 1998; 44: 218-225.
- Pastorino U, Buyse M, Friedel G, Ginsberg RJ, Girard P, Goldstraw P, et al. Long-term results of lung metastasectomy: prognostic analyses based on 5206 cases. The International Registry of Lung Metastases. J Thorac Cardiovasc Surg. 1997; 113: 37-49.
- Kaifi JT, Gusani NJ, Deshaies I, Kimchi ET, Reed MF, Mahraj RP, et al. Indications and approach to surgical resection of lung metastases. J Surg Oncol. 2010; 102: 187-195.
- Erhunmwunsee L, D'Amico TA. Surgical management of pulmonary metastases. Ann Thorac Surg. 2009; 88: 2052-2060.
- Friedel G, Pastorino U, Ginsberg RJ, Goldstraw P, Johnston M, Pass H, et al. Results of lung metastasectomy from breast cancer: prognostic criteria on the basis of 467 cases of the International Registry of Lung Metastases. Eur J Cardiothorac Surg. 2002; 22: 335-344.
- Engstrom PF, Arnoletti JP, Benson AB 3rd, Chen YJ, Choti MA, Cooper HS, et al. NCCN Clinical Practice Guidelines in Oncology: colon cancer. J Natl Compr Canc Netw. 2009; 7: 778-831.
- Wedman J, Balm AJ, Hart AA, Loftus BM, Hilgers FJ, Gregor RT, et al. Value of resection of pulmonary metastases in head and neck cancer patients. Head Neck.1996; 18: 311-316.
- Locati LD, Guzzo M, Bossi P, Massone PP, Conti B, Fumagalli E, et al. Lung metastasectomy in adenoid cystic carcinoma (ACC) of salivary gland. Oral Oncol. 2005; 41: 890-894.
- Liu D, Abolhoda A, Burt ME, Martini N, Bains MS, Downey RJ, et al. Pulmonary metastasectomy for testicular germ cell tumors: a 28-year experience. Ann Thorac Surg. 1998; 66: 1709-1714.
- Finley RK, 3rd, Verazin GT, Driscoll DL, Blumenson LE, Takita H, Bakamjian V, et al. Results of surgical resection of pulmonary metastases of squamous cell carcinoma of the head and neck. Am J Surg. 1992; 164: 594-598.
- Shiono S, Kawamura M, Sato T, Okumura S, Nakajima J, Yoshino I, et al. Pulmonary metastasectomy for pulmonary metastases of head and neck squamous cell carcinomas. Ann Thorac Surg. 2009; 88: 856-860.
- Winter H, Meimarakis G, Hoffmann G, Hummel M, Rüttinger D, Zilbauer A, et al. Does surgical resection of pulmonary metastases of head and neck cancer improve survival? Ann Surg Oncol. 2008; 15: 2915-2926.
- Younes RN, Gross JL, Silva JF, Fernandez JA, Kowalski LP. Surgical treatment of lung metastases of head and neck tumors. Am J Surg. 1997; 174: 499-502.
- . Kesler KA, Wilson JL, Cosgrove JA, Brooks JA, Messiha A, Fineberg NS, et al. Surgical salvage therapy for malignant intrathoracic metastases from nonseminomatous germ cell cancer of testicular origin: analysis of a singleinstitution experience. J Thorac Cardiovasc Surg. 2005; 130: 408-415.
- Xiao H, Liu D, Bajorin DF, Burt M, Bosl GW. Medical and surgical management of pulmonary metastases from germ cell tumors. Chest Surg Clin N Am. 1998; 8: 131-143.
- Balch CM, Soong SJ, Murad TM, Smith JW, Maddox WA, Durant JR. A multifactorial analysis of melanoma. IV. Prognostic factors in 200 melanoma patients with distant metastases (stage III). J Clin Oncol. 1983; 1: 126-134.
- Harpole DH Jr, Johnson CM, Wolfe WG, George SL, Seigler HF. Analysis of 945 cases of pulmonary metastatic melanoma. J Thorac Cardiovasc Surg. 1992; 103: 743-748.
- Dalrymple-Hay MJ, Rome PD, Kennedy C, Fulham M, McCaughan BC. Pulmonary metastatic melanoma -- the survival benefit associated with positron emission tomography scanning. Eur J Cardiothorac Surg. 2002; 21: 611-614.
- Leo F, Cagini L, Rocmans P, Cappello M, Geel AN, Maggi G, et al. Lung metastases from melanoma: when is surgical treatment warranted? Br J Cancer. 2000; 83: 569-572.
- Petersen RP, Hanish SI, Haney JC, Miller CC 3rd, Burfeind WR Jr, Tyler DS, et al. Improved survival with pulmonary metastasectomy: an analysis of 1720 patients with pulmonary metastatic melanoma. J Thorac Cardiovasc Surg. 2007; 133: 104-110.
- Ikai I, Arii S, Kojiro M, Ichida T, Makuuchi M, Matsuyama Y, et al. Reevaluation of prognostic factors for survival after liver resection in patients with hepatocellular carcinoma in a Japanese nationwide survey. Cancer. 2004; 101: 796-802.
- Tomimaru Y, Sasaki Y, Yamada T, Eguchi H, Takami K, Ohigashi H, et al. The significance of surgical resection for pulmonary metastasis from hepatocellular carcinoma. Am J Surg. 2006; 192: 46-51.
- Nakagawa T, Kamiyama T, Nakanishi K, Yokoo H, Kamachi H, Matsushita M, et al. Pulmonary resection for metastases from hepatocellular carcinoma: factors influencing prognosis. J Thorac Cardiovasc Surg. 2006; 131: 1248-1254.
- Zhang SM, Zeng ZC, Tang ZY, Sun J, Cheng JM, Liu R, et al. Prognostic analysis of pulmonary metastases from hepatocellular carcinoma. Hepatol Int. 2008; 2: 237-243.
- Monteiro A, Arce N, Bernardo J, Eugénio L, Antunes MJ. Surgical resection of lung metastases from epithelial tumors. Ann Thorac Surg. 2004; 77: 431-437.
- Temple LK, Brennan MF. The role of pulmonary metastasectomy in soft issue sarcoma. Semin Thorac Cardiovasc Surg. 2002; 14: 35-44.
- Kandioler D, Kromer E, Tuchler H, End A, Müller MR, Wolner E, et al. Long-term results after repeated surgical removal of pulmonary metastases. Ann Thorac Surg. 1998; 65: 909-912.
- Pogrebniak HW, Roth JA, Steinberg SM, Rosenberg SA, Pass HI. Reoperative pulmonary resection in patients with metastatic soft tissue sarcoma. Ann Thorac Surg. 1991; 52: 197-203.
- Ogata Y, Matono K, Hayashi A, Takamor S, Miwa K, Sasatomi T, et al. Repeat pulmonary resection for isolated recurrent lung metastases yields results comparable to those after first pulmonary resection in colorectal cancer. World J Surg. 2005; 29: 363-368.
- Jaklitsch MT, Mery CM, Lukanich JM, Richards WG, Bueno R, Swanson SJ, et al. Sequential thoracic metastasectomy prolongs survival by reestablishing local control within the chest. J Thorac Cardiovasc Surg. 2001; 121: 657-667.