Research Article
Revised Cardiac Risk Index is an Effective Prognostic Tool for Vascular Postoperative Outcomes
Cho C, Lombardi JV, Gaughan J, Alexander JB, Trani JL, Carpenter JP and Caputo FJ*
Division of Vascular & Endovascular Surgery, Cooper University Hospital, USA
*Corresponding author: Francis J Caputo, Division of Vascular & Endovascular Surgery, Cooper University Hospital, 3 Cooper Plaza Suite 411 Camden, NJ 08103, USA
Published: 22 Jul, 2016
Cite this article as: Cho C, Lombardi JV, Gaughan J, Alexander JB, Trani JL, Carpenter JP, et al. Revised Cardiac Risk Index is an Effective Prognostic Tool for Vascular Postoperative Outcomes. Clin Surg. 2016; 1: 1050.
Abstract
Objectives: The Revised Cardiac Risk Index (RCRI) is a tool clinician’s use for preoperative risk stratification and predicting the likelihood of cardiac complications. This study looks at the role
of the RCRI for postoperative outcomes in vascular patients undergoing lower extremity bypass surgery.
Methods: National Surgical Quality Improvement Program (NSQIP) data from 2005-2011 of
patients undergoing lower extremity revascularization based on CPT codes were analyzed looking
at demographics, revised cardiac risk index variables (coronary heart disease, congestive heart
failure, creatinine >2, insulin dependent diabetes, high-risk surgery, cerebrovascular disease), and
outcomes including operative time, hospital length of stay (LOS), return to operating room, and
death. Data was analyzed using single and multivariable linear and logistical regression.
Results: 70,560 patients were included with 36% female and 64% male patients. Of these patients,
40,893 (58%) were over the age of 65 with 27,576 (39%) of the patients between the ages of 46 and 65.
Using linear regression, with each unit increase of the RCRI, operative time increases 5.4 minutes
(p<0.0001), and LOS increases 3.23 days (P<0.0001). Through logistic regression, with each unit
increase in the RCRI the risk of returning to OR increases 1.4 times (OR=1.449; 95%CI 1.41-1.49)
and the risk of death increases 1.97 times (OR =1.972; 95% CI 1.87-2.08). The risk of MI increased
1.6 times for each unit increase in RCRI (P<.0001).
Conclusion: Using a large retrospective national database, preoperative RCRI can be a useful
measure for predicting postoperative outcomes in patients undergoing lower extremity bypass surgery.
Introduction
Peripheral vascular disease is estimated to have a prevalence of 5.8% in the US population 40
years of age or older and to increase exponentially with age [1]. With the aging baby boomers,
the prevalence can only increase, likely requiring increased intervention from vascular surgeons.
However, patients with peripheral vascular disease seldom have disease limited to solely the
extremities. In fact, in the setting of PAD, one study found 68% patients to have coexistent Coronary Aartery Disease (CAD) and 42% to have coexistent stroke in the geriatric populations [2]. Other
studies have shown progressive Peripheral Arterial Disease (PAD) can lead to 2-3 times increased
Cerebral Vascular Disease (CVD) morbidity/mortality at 3-6 years [3]. With these comorbidities,
based on the REACH registry, patients with PAD have the highest 3 year event rate of MI/Stroke/
Death/Re-hospitalization at 40.4% [4].
These high risk patients usually have preoperative risk stratification before undergoing any
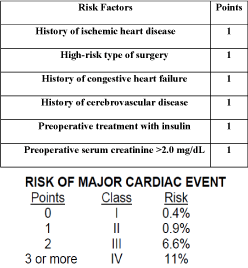
vascular intervention. An easy bedside clinical tool is the Revised Cardiac Risk Index (RCRI)
that can predict cardiac complications including MI, pulmonary edema, ventricular fibrillation
or primary cardiac arrest, and complete heart block [5] (Figure 1). By taking various risk factors for cardiovascular disease including diabetes, renal dysfunction, history of congestive heart
failure, history of cerebrovascular disease and ischemic heart disease, the index is used to predict
cardiovascular complications. Using the same principles and risk factors, this study applies the RCRI
to predict peripheral vascular complications in addition to cardiovascular problems. Additionally
we hypothesized the more risk factors a patient has, the higher the mortality and worsened outcome
the patient will have.
Methods
Data was collected from the American College of Surgeons National Surgical Quality
Improvement Program (ACS NSQIP), a validated, de-identified,
prospective database containing clinical variables from up to 315
clinical sites (in 2011). A trained Surgical Clinical Reviewer (SCR)
collected preoperative through 30 day postoperative data on
randomly assigned patients and entered data was entered online
in a HIPAA compliant database [6]. Data was collected through
patients undergoing procedures listed as specific Current Procedural
Terminology (CPT) codes. CPT codes were collected as follows:
infrainguinal bypass graft with vein; femoral-popliteal (CPT 35556),
femoral-anterior tibial, posterior tibial, peroneal artery or other
distal vessels (CPT 35566), popliteal-tibial, -peroneal artery or other
distal vessels (CPT 35571), and tibial-tibial; peroneal-tibial or tibial/
peroneal; trunk-tibial (CPT 35570). Patients also undergoing in-situ
vein bypass femoral-popliteal (CPT 35583), femoral-anterior tibial,
posterior tibial, or peroneal artery (CPT 35585), popliteal-tibial,
peroneal (CPT 35587) were selected as well. Patients who had bypass
graft with other than vein; femoral-popliteal (CPT 35656), femoralanterior
tibial, posterior tibial, or peroneal artery (CPT 35666), and
popliteal-tibial or -peroneal artery (CPT 35671) were also included.
As this was a national HIPAA regulated database study, Institutional
Review Bard (IRB) approval was not required nor was patient
consent required.
Demographic data was obtained from the selected patients. Data
was analyzed based on preoperative RCRI variables (congestive heart
failure, ischemic heart disease, cerebrovascular disease, diabetes,
creatinine > 2), operative time, 30 day outcomes including length of
stay, mortality, MI, and return to operating room.
Statistical analysis
Variables were summarized for the study population and
presented as means with standard deviations for continuous variables
and percentages for categorical variables. Single and multiple variable
linear and logistic regressions were used to determine the relationship
of the RCRI with surgical outcomes. Regression coefficients and odds
ratio are presented with 95% confidence intervals. All analyses were
carried out using SAS v9.4 (SAS Institute, Cary, NC.)
Figure 1
Results
From the database, 70,560 patients were identified with
demographic data demonstrated in (Table 1). The majority of
patients were over 66 years old (55.9%) with 39.1% in the 46-65 year
old age bracket. Patients were predominantly male (64.3%) compared
to female (35.7%) (p<.0001). Most all of the patients had RCRI scores
of 3 or less, with a score of 1 being most prevalent (64.3%).
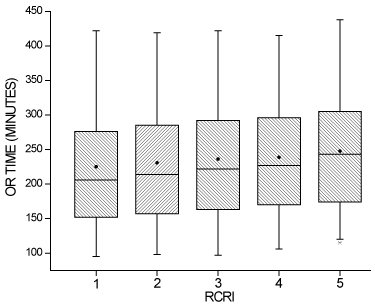
Mean average operative time was 227.62 minutes ±104.59
minutes (Table 2). Based on linear regression, each unit increase
in RCRI resulted in an increase in operative time of 5.4 minutes
(P<.0001) (Figure 2). The main contributing risk factors were diabetes
and creatinine >2, although CHF and CVD were also significant
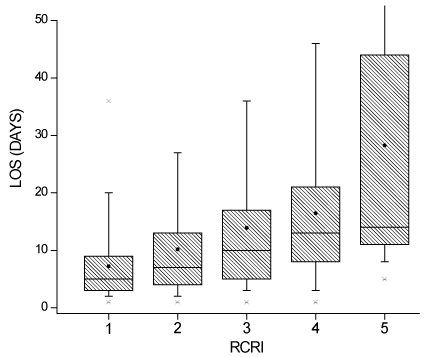
contributors. Length of stay was recorded for 99.8% of the patients.
The mean hospital length of stay was 8.7 days ± 10.4 days. For each
unit increase in RCRI, length of stay increased 3.23 days (P<.0001)
(Figure 3).
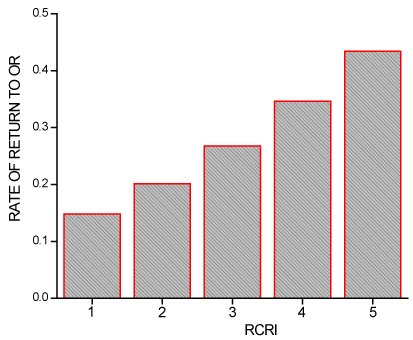
Using single variable analysis, the 17.4% (Table 3) of patients
requiring return to the operating room were 1.45 (95%CI 1.41-
1.49) times more likely to return to the OR for each unit increase
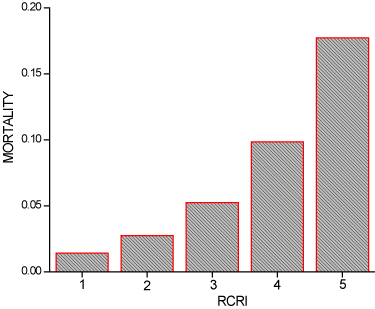
in RCRI (P<.0001) as noted in Figure 4. Overall mortality was 2.2%
with an odds ratio of 1.97 (95% CI 1.87-2.08) for each unit increase
in RCRI (P<.0001) (Figure 5). Contributing to RCRI were mainly
CHF, ischemic heart disease, and Cr > 2; however, CVD was also a
significant contributor. Unsurprisingly, the risk of MI increased 1.6
times for each unit increase in RCRI (P<.0001). When adjusted for
age and gender using multiple variable analysis, the relationship of
RCRI to outcomes was similar (data not shown).
Table 1
Figure 2
Figure 3
Table 2
Table 2
Outcomes and respective linear regression slopes with 95% confidence interval for each unit increase in RCRI.
Table 3
Table 3
Outcomes and respective odds ratios with 95% confidence interval for each unit increase in RCRI.
Figure 4
Discussion
The Revised Cardiac Risk Index is a well used bedside tool for
preoperative risk stratification for cardiovascular complications
with its easily obtainable variables. The objective of this study was to
determine if RCRI could predict lower extremity vascular outcomes.
We hypothesized that as RCRI increased, patients would have worse
outcomes.
Operative time was increased by 5.4 minutes for every unit
increase in RCRI. Increased operative time has been shown to
increase surgical site infections [7-9]. This lengthens hospital stay
by 9.7 days as well as increases cost by $20,842 per admission [10].
Increased operative time is also associated with increased blood loss,
operative difficulty, repeat operation, and increased tissue ischemia
in an already high risk patient. Increase in operative time also has
economic repercussions. One study [11] found a $5-$20 increase per
minute for operative time.
It is not surprising that the hospital length of stay increased 3.23
days for each unit increase in RCRI. With increased risk factors,
patients likely had more complications whether it be renal, cardiac,
graft complications, surgical site infections, etc. A slight increase in
creatinine can increase length of stay by 3.5 days and increase hospital
costs by $7500 [12].
For each unit increase in RCRI, patients had a 1.45 times greater
chance of returning to OR. The reoperation rate in this study was
17.4%, a little higher than the 11.2% found in another study [13].This
can increase the morbidity and mortality (from 2.0% to 8.8% [12]) for
patients, as well as increase length of stay and hospital costs [14-16].
Lastly our study found a 2.2% mortality with an odds ratio 1.97
for each unit increase in RCRI. The mortality rate is consistent with
other studies reporting 2.6%-5.3% [17-19]. The odds ratio is quite
startling for just an additional risk factor in RCRI. In one study [20].
CHF and hemodialysis dependent renal failure were found to be
independent risk factors for mortality.
This study has multiple limitations. Since it is a NSQIP study, it
is limited to 30 day postoperative data. With NSQIP it is not possible
to explore specifics of individual patient details about reasons for
returning to the OR. Since the data is based on CPT codes, it is
not possible to know if a primary or reoperation was performed.
However, the variables selected leave little for interpretive ambiguity,
e.g. mortality. Additionally because the database is large, an extensive
sample was available.
Figure 5
Conclusion
The Revised Cardiac Risk Index was designed to predict cardiac complications, but also appears to be a good tool to be used in infrainguinal bypasses. The higher the index, the worse the outcomes. Due to the increased mortality, chances of returning to OR for another procedure, informed consent and a better discussion can be made with patients before choosing to undergo these procedures.
References
- Allison MA, Ho E, Denenberg JO, Langer RD, Newman AB, Fabsitz RR, et al. Ethnic-specific prevalence of peripheral arterial disease in the United States. 2007 Am J Prev Med. 2007; 32: 328-333.
- Ness, J, Aronow WS. Prevalence of coexistence of coronary artery disease, ischemic stroke, and peripheral arterial disease in older persons, mean age 80 years, in an academic hospital based geriatrics practice. J Am Geriatr Soc. 1999; 47: 1255-1256.
- Criqui MH, Ninomiya JK, Wingard DL, Ji M, Fronek A. Progression of peripheral arterial disease predicts cardiovascular disease morbidity and mortality. J Am Coll Cardiol. 2008; 52: 1736-1742.
- Alberts MJ, Bhatt DL, Mas JL, Ohman EM, Hirsch AT, Röther J, et al. Three-year follow-up and event rates in the international reduction of atherothrombosis for continued health registry. Eur Heart J. 2009; 30: 2318-2326.
- Lee TH, Marcantonio ER, Mangione CM, Thomas EJ, Polanczyk CA, Cook EF, et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation. 1999; 100: 1043-1049.
- NSQIP
- Haridas M, Malangoni MA. Predictive factors for surgical site infection in general surgery. Surgery. 2008; 144: 496-503.
- Chang JK, Calligaro KD, Ryan S, Runyan D, Dougherty MJ, Stern JJ. Risk factors associated with infection of lower extremity revascularization: Analysis of 365 procedures performed at a teaching hospital. Ann Vasc Surg. 2003; 17: 91-96.
- Leong G, Wilson J, Charlett A. Duration of operation as a risk factor for surgical site infection: comparison of English and US data. J Hosp Infect. 2006; 63: 255-262.
- Homer-Vanniasinkam S. Surgical site and vascular infections: treatment and prophylaxis. Int J Infect Dis. 2007; 11: S17-S22.
- Babineau TJ, Becker J, Gibbons G, Sentovich S, Hess D, Robertson S, et al. The “cost” of operative training for surgical residents. Arch Surg. 2004; 139: 366-370.
- Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol. 2005; 16: 3365-3370.
- Ploeg AJ, Lange CP, Lardenoye JW, Breslau PJ. The incidence of unplanned returns to the operating room after peripheral arterial bypass surgery and its value as indicator of quality of care. Vasc Endovascular Surg. 2008; 42: 19-24.
- Ng RL, Davies AH, Magee TR, Tennant S, Horrocks M, Baird RN. Early reoperation rates after arterial surgery. Eur J Vasc Surg. 1994; 8: 78-82.
- Davies AH, Pope I, Collin J, Morris PJ. Early reoperation after major vascular surgery: a four-year prospective analysis. Br J Surg. 1992; 79: 76-78.
- Birkmeyer JD, Hamby LS, Birkmeyer CM, Decker MV, Karon NM, Dow RW. Is unplanned return to the operating room a useful quality indicator in general surgery? Arch Surg. 2001; 136: 405-411.
- Schanzer A, Hevelone N, Owens CD, Beckman JA, Belkin M, Conte MS. Statins are independently associated with reduced mortality in patients undergoing infrainguinal bypass graft surgery for critical limb ischemia. J Vasc Surg. 2008; 47: 774-781.
- LaMuraglia GM, Conrad MF, Chung T, Hutter M, Watkins MT, Cambria RP. Significant perioperative morbidity accompanies contemporary infrainguinal bypass surgery: an NSQIP report. J Vasc Surg. 2009; 50: 299-304.
- Moitra VK, Flynn BC, Mazzeffi M, Bodian C, Bronheim D, Ellis JE. Indication for surgery, the revised cardiac risk index, and 1-year mortality. Ann Vasc Surg. 2011; 25: 902-908.
- Henke PK, Blackburn S, Proctor MC, Stevens J, Mukherjee D, Rajagopalin S, et al. Patients undergoing infrainguinal bypass to treat atherosclerotic vascular disease are underprescribed cardioprotective medications: effect on graft patency, limb salvage, and mortality. J Vasc Surg. 2004; 39: 357-365.