Research Article
Management of Large Cirsoid Aneurysms of the Scalp using Tissue Expanders, Intravascular Occlusion, and En Bloc Excision
Nagy E, Setta HS and Elshahat A*
Department of Plastic Surgery, Ain Shams University, Egypt
*Corresponding author: Ahmed Elshahat, Eldemerdash Hospital, 56 Ramsis Street, Abbassia 11566, Cairo, Egypt
Published: 11 Jul, 2016
Cite this article as: Nagy E, Setta HS, Elshahat A. Management of Large Cirsoid Aneurysms of the Scalp using Tissue Expanders, Intravascular Occlusion, and En Bloc Excision. Clin Surg. 2016; 1: 1049.
Abstract
Arteriovenous malformations of the scalp consist of abnormally connecting arterial feeding vessels and draining veins that are devoid of a normal capillary bed within the subcutaneous fatty layer of
the scalp. The name "cirsoid" in cirsoid aneurysm is derived from the Greek word kirsos meaning
varix or varicose vein. The en bloc excision of scalp tissues affected by aneurysm is preferable to
selective ligation of the feeding and draining vessels. Because the management of cirsoid aneurysm
is an elective procedure, it is best to use tissue expanders to create sufficient scalp flaps to reconstruct
the site of the excised lesion in the first stage. Preoperative embolization greatly reduces blood loss
during resection. The aim of this work is to present the successful management of cirsoid aneurysms
of the scalp using tissue expanders, endovascular occlusion, and en bloc excision. Five patients who
had presented cirsoid aneurysms of the scalp (two temporoparietal, two frontal, and one occipital)
were managed successfully using three stages of intervention. The first stage was the application
of one or two tissue expanders, in which expanders were applied under the normal (non-affected)
scalp in the subgaleal plane; expansion was then performed weekly for 3-4 months. The second
stage involved endovascular occlusion through endovascular neuroradiology. The third stage was
performed the day after occlusion and included en bloc excision, the delivery of tissue expanders,
and reconstruction of the site of excision using scalp flaps. The postoperative period was uneventful.
Six months to three years of following up showed no recurrence. We conclude that the three-stage
management of large cirsoid aneurysms of the scalp (application of tissue expanders, endovascular
occlusion, then en bloc excision and reconstruction) provides excellent results.
Keywords: Cirsoid aneurysm; Vascular malocclusion; Tissue expansion; Endovascular occlusion; Scalp
Introduction
Cirsoid aneurysms are rare arteriovenous fistulas of the scalp whose etiology is usually congenital, although traumatic fistulas have also been reported [1-3]. The condition is called “cirsoid” (from the Greek kirsos, meaning "varix," or varicose vein) because of the characteristic variceal dilatation of
the draining veins. The various names that are used to describe vascular malformations of the scalp
include aneurysm cirsoide, aneurysma serpentinum, aneurysm racemosum, plexiform angioma,
arteriovenous fistula and Arterio Venous Mmalformation (AVM) [1,2,4].
Cirsoid aneurysms of the scalp were first described in 1833 by Brecht [5]. Only 10%–20% of these
arteriovenous fistulas develop following penetrating trauma to the scalp. Traumatic arteriovenous
malformation of the scalp develops months or even years after the scalp trauma. While congenital
arteriovenous malformation of the scalp (the remaining 80%-90%) might be present at birth, for
most patients it is asymptomatic until adulthood [6-8].
In 90% of patients, the superficial temporal artery is the main supply to the fistula, with only one
dominant feeding artery in 71% of those patients [9]. In the remaining 29% of cases usually involve
both the superficial temporal and occipital arteries [5,9].
The most important distinction regarding AVM is between single-hole arteriovenous fistula
(usually traumatic) and more complex lesions, which are thought to be (congenital). These more
complex lesions are characterized by a network of abnormal channels (nidi) between the arterial
feeder(s) and the draining vein or veins (i.e., an AVM). They also may have components with no
intervening nidi (i.e., fistulas) [10].
The management of scalp AVM is difficult because of the scalp's
high shunt flow, intracranial communication, complex vascular
anatomy, and cosmetic problems. The indication of treatment
includes cosmetic relief of the pulsatile or non-pulsatile mass and the
prevention of hemorrhage and other symptoms such as headache and
tinnitus [5,11-14 ].
It is well known in the literature that the only effective method of
preventing the evolution of AVM of the scalp is to exclude the lesion
completely from circulation [15]. The management of AVMs of the
head and neck includes observation, ligation of the feeding arteries,
embolization, and surgical resection [2,7,9,16,17],or a combination of
these elements [18,19]. Ligation of the feeding arteries was one of the
earliest treatment methods for this condition, although recurrence
nearly always ensues because of the development of collateral vessels
[1,2,17]. The collateral vessels that develop following ligation of the
feeding arteries might parasitize blood flow from the brain, which can
lead to ischemic complications [20].
Endovascular treatment may be applied in order to decrease
the hemorrhage and to facilitate surgical treatment or the direct
treatment of AVMs [4,9,21,22]. The pre-operative embolization of
both feeders and nidus before surgery is safer than embolization of
the feeders alone for reducing the risk of excessive hemorrhage [4].
Three different approaches have been described in the literature for
accessing the fistula; femoral transarterial catheterization, femoral
transvenous catheterization and direct percutaneous catheterization
of the feeding arteries or draining veins [13,18,23-26].
Surgical excision is the most common and successful method of
dealing with vascular scalp lesion [6,24,27,28]. Surgical treatment
is particularly indicated for preventing bleeding, the resolution of
cosmetic problems, and for the treatment of the accompanying issues
of tinnitus and headache [4,21,29]. Various techniques have been
used to control hemorrhage during surgery, including percutaneous
sutures of the feeding vessels [6]. Interlocking sutures along the
line of incision, and the use of a scalp tourniquet and an intestinal
clamp over the base of the flap [30]. A step-wise incision with careful
pressure control is useful for controlling scalp bleeding [11].
The excision of large arteriovenous malformation of the
scalp leaves a large defect that requires reconstruction; the Ideal
reconstruction is the use of hair-bearing skin flaps. Marotta et al. [31]
expanded the scalp adjacent to the arteriovenous malformation to
prepare a hair-bearing skin flap that they used for reconstruction.
The lack of descriptions of this technique in the literature, together
with its absence in plastic surgery journals, encouraged us to revisit
this technique. The aim of this work is thus to present a complete
excision using a combined approach of preoperative endovascular
occlusion followed by radical excision and coverage of the defect via
expanded adjacent scalp.
Table 1
Patients and Methods
This is a retrospective case series study on five patients with cirsoid
aneurysms who were treated from March 2012 to December 2015.
Diagnosis was based on both a clinical picture of pulsatile swelling in
the scalp with bruit and thrill and on the intravascular dye injection
(angiography). None of our patients presented with hemorrhage, and
none had neurological deficits. The patients' lesions were located in
the temperoparietal region in two patients, in the frontal region in
two patients and in the occipital region in one patient. No intracranial
lesion was demonstrated in any patient. (Table 1) shows the data of the
five patients included in this study.
In an attempt to completely eradicate the AVM, one or two large,
rectangular tissue expanders were then placed in the less-affected
scalp area in the subgaleal plane at least 3 cm from the edge of the
AVM. All incisions for the expander placement were perpendicular
to the expansion axis and were distant from the AVM. Ports were
buried in all patients; the use of suction drains for a few days was
the routine for all cases. In the second stage and before excision,
aggressive preoperative coiling (occlusion) of the malformation was
performed by the intervention neuroradiology department in our
hospital. One day after endovascular occlusion, complete excision
of the malformation was then performed. Interlocking sutures along
the line of incision were used in all patients prior to surgical excision
to decrease blood loss. The expanded scalp was then transposed for
coverage of the entire defect.
Results
All patients were treated according to the proposed protocol.
Expansion was successful in all patients despite the seropurulent
collection that was detected in one patient around the expanders;
this was managed successfully by exteriorization of the ports. The
operating time during the application of the tissue expanders ranged
from 30 to 45 minutes in the five cases, while en bloc exsion and
reconstruction ranged from 90 to 120 minutes. Blood loss ranged
from 350 to 1000ml in the en bloc excision. Blood transfusion of 500
ml whole blood was the routine in all five cases.
All the preoperative symptoms and signs were eliminated. The
expanded flaps survived completely, and all the patients had excellent
postoperative cosmetic appearance; No postoperative complications
related to the surgery had occurred. The patients were followed up
with for a period ranged from 6 to 36 months; no recurrences occurred
during the follow-up period. Figures 1-6 show three patients after
application of the tissue expanders and before excision of the lesions
(1,3,5) and after excision of the cirsoid aneurysms and advancement
of the expanded flaps (2,4,6).
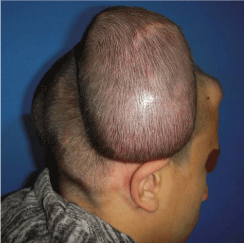
Figure 1
Figure 1
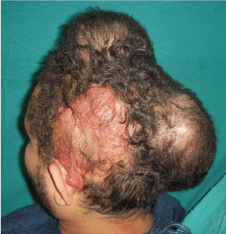
A photo shows large cirsoid aneurysm in scalp (left temporoparietal
region) of 26 year old male patient with two inflated tissue expanders
underneath the adjacent sound scalp.
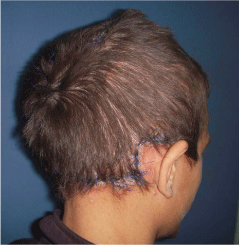
Figure 2
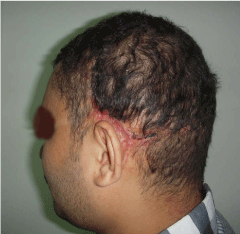
Figure 2
A photo of the same patient in figure 1 after en block excision of the
cirsoid aneurysm and advancement of expanded scalp flaps.
Figure 3
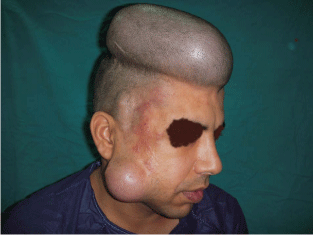
Figure 3
A photo shows cirsoid aneurysm in the right frontal region of a 30
year old male patient with two inflated tissue expanders in the adjacent scalp
and cheek.
Figure 4
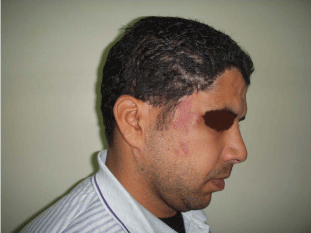
Figure 4
A photo shows the same patient in figure 3 after en block excision
of the cirsoid aneurysm and advancement of expanded flaps to reconstruct
the right frontal region and anterior hair line.
Figure 5
Figure 5
A photo shows cirsoid aneurysm in the right occipital and
postauricular regions in a 19 year old male patient with inflated tissue
expander underneath the adjacent sound scalp.
Figure 6
Figure 6
A photo shows the same patient in figure 5 after en block excision
of the cirsoid aneurysm and advancement of the expanded scalp flap.
Discussion
Cirsoid aneurysms are rare despite the intense vascularity of the
scalp and the relatively high frequency of trauma to this region [3].
Most of the reports in the literature consist of individual case reports;
very few studies consist of sufficient number of patients [7,13,18].
One study from the literature mentioned that these lesions usually
begin as a small, subcutaneous lump on the head that over a period
of time evolves into a grotesque, deforming mass [13]. A bruit or a
throbbing headache might be the chief complaint in some patients;
other presenting symptoms include pulsatile tinnitus or (rarely)
hemorrhage from the mass [25]. Large lesions may occasionally be
associated with scalp necrosis. Congestive heart failure has occurred
with large fistulas [18]. The same sequelae were found in our patients,
except for the absence of hemorrhage or congestive heart failure.
Definitive treatment for the condition requires controlled en
bloc excision, in which the entire malformation is excised [15,32].
In the five patients included in this study coiling (occlusion) of the
arteriovenous malformations was done before the en bloc excision to
limit intraoperative bleeding; interlocking sutures were also used for
the same purpose. Blood loss still took place despite these precautions,
so blood transfusion was the routine.
Many techniques have been used to cover scalp defects; some of
these include direct closure in small AVM defects, skin and galealpericranial
flaps, compound musculocutaneous flaps, as well as freetissue
transfer [34,35]. These techniques were not applicable in the
five patients included in this study, however, because the lesions
were large and the defects that were left after en bloc excision were
even larger. Although Free-tissue transfer can cover large defects, it
requires normal vessels for anastomosis and the skin provided is non
hair bearing. Donor-site morbidity is another disadvantage of the use
of free flaps
Similarly to the technique Marotta et al. [31] used, planned tissue
expansion of the adjacent scalp allowed for large hair-bearing flaps to
cover the resultant defects after en bloc excision.
Periexpander seropurulent collection in one patient was managed
by exteriorization of the buried ports for drainage in order to
salvage the infected expanders (as mentioned by Elshahat [36]); this
management was successful, and the expansion was completed to the
planned-for volume. A double back-cut of the expanded flaps [37]
was performed to maximize the gain from the expanded flaps; this
allowed for full coverage of the resultant defects.
One point of criticism is that bearing a tissue expander on the
head for 3-4 months will likely cause patients social- and workrelated
difficulties, so economic and social factors should be taken
into account when indicating such a procedure. On the other hand,
the non-treated patients suffer repeated attacks of headache and
subsequently have repeated need for sick leave in addition to the
psychological effects of the cosmetic disfigurement from the lesion.
Therefore, bearing a tissue expander on the head for 3-4 months is
made tolerable by the elimination of these symptoms.
Conclusion
Careful planning for the management of large cirsoid aneurysm is crucial to assure uneventful outcomes. Three-stage management with application of tissue expanders and endovascular occlusion shortly before en bloc excision and reconstruction resulted in excellent results with no recurrence, no morbidity and an appealing cosmetic outcome.
References
- Khodadad G. Familial cirsoid aneurysm of the scalp. J Neurol Neurosurg Psychiatry. 1971; 34: 664-667.
- Khodadad G. Arteriovenous fistula of the scalp. Ann Surg. 1973; 177: 79-85.
- Badejo L, Rockwood P. Case report: Traumatic arteriovenous fistula of the scalp. J Neurosurg. 1987; 66: 773-774.
- Massimi L, De Bonis P, Esposito G, Novegno F, Pettorini B, Tamburrini G, et al. Vertex scalp mass as presenting sign of a complex intracranial vascular malformation. J Neurosurg Pediatr. 2009; 3: 307-310.
- Muthukumar N, Rajagopal V, Manoharan A, Durairaj N. Surgical management of cirsoid aneurysms. Acta Neurochir (Wien). 2002; 144: 349-356.
- Shenoy SN, Raja A. Scalp arteriovenous malformations. Neurol India. 2004; 52: 478-481.
- Fisher-Jeffes ND, Domingo Z, Madden M, de Villiers JC. Arteriovenous malformations of the scalp. Neurosurgery. 1995; 36: 656-660.
- Li F, Zhu S, Liu Y, Chen Y, Chi L, Chen G, et al. Traumatic arteriovenous fistula of the superficial temporal artery. J Clin Neurosci. 2007; 14: 595-600.
- Gurkanlar D, Gonul M, Solmaz I, Gonul E. Cirsoid aneurysms of the scalp. Neurosurg. Rev 2006; 29: 208-212.
- Robbins SL, Cotran RS, Kumar V. Pathological basis of disease. 3rd edn. WB Saunders Philadelphia P 505.
- Domingo Z, Fisher-Jeffes ND, de Villiers JC. Surgical management of arteriovenous malformations of the scalp. In: Shmidek HN, editor. Operative Neurosurgical Techniques: Indications, Methods and Results. 4th edn. Philadelphia: Saunders Company; 2000. p. 1331-1338.
- Weinzweig N, Chin G, Polley J, Charbel F, Shownkeen H, Debrun G. Arteriovenous malformation of the forehead, anterior scalp and nasal dorsum. Plast Reconstr Surg. 2000; 105: 2433-2439.
- Nagasaka S, Fukushima T, Goto K, Ohjimi H, Iwabuchi S, Maehara F. Treatment of scalp arteriovenous malformation. Neurosurgery. 1996; 38: 671-677.
- Shepard RH. Proceedings: Cirsoid arteriovenous malformations of the scalp. J Neurol Neurosurg Psychiatry. 1975; 38: 827-828.
- Schultz RC, Hermosillo CX. Congenital arteriovenous malformation of the face and scalp. Plast Reconstr Surg .1980; 65: 496-501.
- Berenstein A, Scott J, Choi IS, Persky M. Percutaneous embolization of arteriovenous fistulas of the external carotid artery. AJNR Am J Neuroradiol. 1986; 7: 937-942.
- Dandy WE. Arteriovenous aneurysms of the scalp and face. Arch Surg. 1946; 52: 1-32.
- Barnwell SL, Halbach VV, Dowd CF, Higashida RT, Hieshima GB. Endovascular treatment of scalp arteriovenous fistulas associated with a large varix. Radiology. 1989; 173: 533-539.
- Elkin DC. Cirsoid aneurysm of the scalp; report of four cases. Trans South Surg Assoc. 1946; 57: 122-131.
- Malan E, Azzolini A. Congenital arteriovenous malformations of the face and scalp. J Cardiovasc Surg.1986; 8: 109-140.
- Gupta AK, Purkayastha S, Bodhey NK, Kapilamoorthy TR, Krishnamoorthy T, Kesavadas C, et al. Endovascular treatment of scalp cirsoid aneurysms. Neurol India. 2008; 56: 167-172.
- Tiwary SK, Khanna R, Khanna AK. Craniofacial cirsoid aneurysm: 2-stage treatment. J Oral Maxillofac Surg. 2007; 65: 523-525.
- Kasdon DL, Altemus LR, Stein BM. Embolization of the traumatic arteriovenous fistula of the scalp with radiopaque gelfoam pledgets. Case report and technical note. J Neurosurg. 1976; 44: 753-756.
- Hendrix LE, Meyer GA, Erickson SJ. Cirsoid aneurysm treatment by percutaneous injection of sodium tetradecyl sulfate. Surg Neurol. 1996; 46: 557-560.
- Heilman CB, Kwan ES, Klucznik RP, Cohen AR. Elimination of a cirsoid aneurysm of the scalp by direct percutaneous embolization with thrombogenic coils. Case report. J Neurosurg.1990; 73: 296-300.
- Matsushige T, Kiya K, Satoh H, Mizoue T, Kagawa K, Araki H. Arteriovenous malformation of the scalp: Case report and review of the literature. Surg Neurol. 2004; 62: 253-259.
- El shazly AA, Saoud KM. Results of surgical excision of cirsoid aneurysm of the scalp without preoperative interventions. Asian J Neurosurg. 2012; 7: 191-196.
- Senoglu M, Yasim A, Gokce M, Senoglu N. Nontraumatic scalp arteriovenous fistula in an adult: Technical report on an illustrative case. Surg Neurol. 2008; 70: 194-197.
- Burrus TM, Miller GM, Flynn LP, Fulgham JR, Lanzino G. NeuroImages. Symptomatic left temporal arteriovenous traumatic fistula. Neurology. 2009; 73: 570.
- Hochberg J, Ardenghy M, Pait TG. Scalp tourniquet. Br J Plast Surg. 1994; 47: 194-198.
- Marotta TR, Berenstein A, Zide B. The combined role of embolization and tissue expansion in management of arteriovenous malformation of the scalp. AJNR Am J Neuroradiol. 1994; 15: 1240-1246.
- Hurwitz DJ, Kerber CW. Hemodynamic considerations in the treatment of arteriovenous malformations of the face and scalp. Plast Reconstr Surg. 1981; 67: 421-434.
- Shultz RC, Hermosillo CX. Congenital arteriovenous malformation of the face and scalp. Plast Reconstr Surg. 1980; 65: 496-501.
- Horowitz JH, Persing JA, Nichter LS, Morgan RF, Edgerton MT. Galealpericranial flaps in head and neck reconstruction: anatomy and application. Am J Surg. 1984; 148: 489-497.
- Fonseca JL. Use of pericranial flap in scalp wounds with exposed bone. Plast ReconstrSurg. 1983; 72: 786-790.
- Elshahat A. Exteriorization of buried port to salvage infected tissue expander. Eplasty. 2009; 9: e37.
- Zide BM, Karp NS. Maximizing gain from rectangular tissue expanders. Plast Reconstr Surg. 1992; 90: 500-504.